Search by Categories
- Insights
- Rare Disease
- Oncology
- Cell and Gene Therapy
- Medical Devices
- Consulting
- News & Analysis
-
Therapeutic Areas

Our expertise in the industry

The FDA recently approved Amgen’s Wezlana (ustekinumab-auub) for various inflammatory conditions, marking the first biosimilar approval referencing the popular J&J drug Stelara (ustekinumab). Wezlana, mirroring its reference product, is approved for treating multiple inflammatory diseases in adults, including m...
Find More
Roche’s Vabysmo secures a new FDA nod for retinal vein occlusion treatment, intensifying the competition with Regeneron and Bayer’s Eylea in the eye medication market. This approval for Vabysmo in RVO adds another shared medical indication for the competing eye medications. Vabysmo’s successful FDA approval for RVO...
Find More
Competitive inroads by Regeneron’s Eylea in the retinal vein occlusion treatment landscape changed the market once dominated by Roche.Moving forward, RVO’s robust pipeline, along with several VEGF inhibitors, creates trouble for the big ones; Lucentis and Eylea. Retinal vein occlusion is the second most common s...
Find More
TherapeuticsMD bags FDA approval for hormone therapy The FDA has nodded hormone therapy of TherapeuticsMD for hot flashes that are associated with menopause. Bijuva is an oral treatment that contains molecularly identical (bio-identical) versions of the hormones estradiol and progesterone for menopausal women with a...
Find More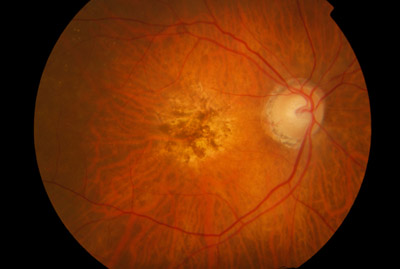
Wet AMD is a chronic eye disease that causes blurred vision or a blind spot in a person’s visual field. It is characterized by abnormal blood vessels that leak blood and fluids into the macula, a region of the eye that is responsible for sharp, central vision. Age-related macular degeneration (AMD) is a common eye c...
Find More
The American Society of Clinical Oncology (ASCO) is one of the largest and most respected conferences in the field of oncology. Held annually, this conference brings together researchers, physicians, and other healthcare professionals from around the world to discuss the latest advances in cancer research, diagnosis, and treatment.