Alzheimer's Disease Market Summary
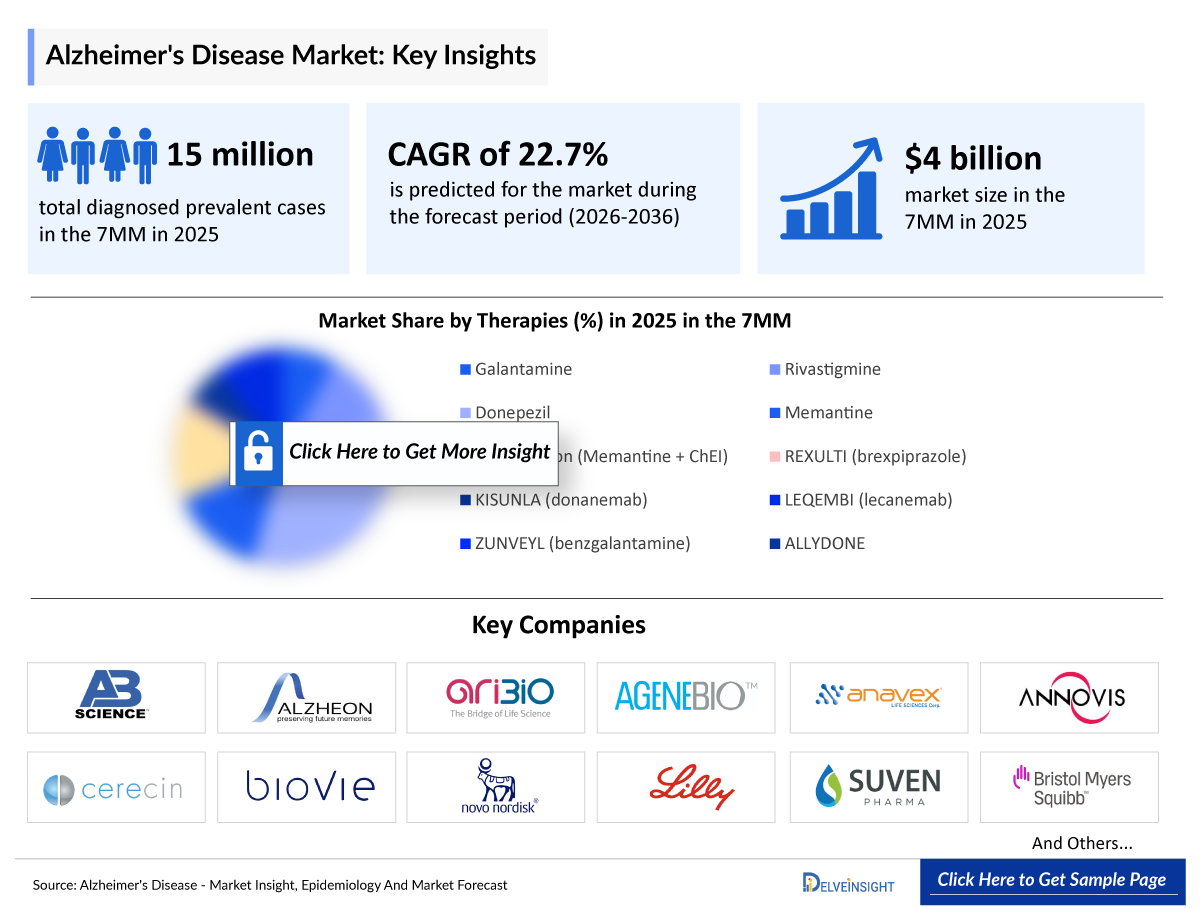
- According to DelveInsight’s analysis, the Alzheimer’s disease market in the 7MM (the US, EU4, UK, and Japan) is valued at approximately USD 4 billion in 2025. Driven by the clinical take-off of disease-modifying therapies, the market is projected to expand at a robust CAGR of 22.7%, with 7MM.
- The United States continues to hold the largest market share, with a revenue of more than USD 2 billion in 2025. DelveInsight estimates this number will rise significantly by 2036, with the total 7MM diagnosed prevalent cases growing due to the aging "baby boomer" generation.
Alzheimer’s Disease Insights and Trends
- The therapeutic landscape is undergoing a paradigm shift from symptomatic drugs (like galantamine and memantine) to Anti-Amyloid Monoclonal Antibodies. Key players include lecanemab (LEQEMBI) and donanemab (KISUNLA).
- In 2025, the treatment market was led by the cholinesterase inhibitor donepezil, generating approximately USD 1,500 million across the 7MM.
- In EU4 and the UK, the total market size was calculated to be nearly USD 1 billion in 2025, accounting for 24% of the total 7MM revenue. Germany maintains the highest disease burden in the region, accounting for USD 280 million, followed by France with USD 250 million, while the UK accounted for the least with nearly USD 120 million.
- Advances in diagnostic pathways, including increasing use of biomarker-based tools such as PET imaging and emerging blood-based assays, are enabling earlier disease identification and facilitating timely treatment initiation.
- Japan faces a unique challenge with high rates of Alzheimer’s-associated agitation. The market size in Japan was calculated to be around USD 80 million in 2025, which is expected to increase at a CAGR of 14.3% during forecast period.
- The emerging pipeline toward 2036 is focused on "cocktail therapies" and new assets from companies like Eli Lilly, BioVie, and Alzheon. Hydromethylthionine mesylate (HMTM), one of the emerging therapies, is anticipated to enter the US market in 2026.
- A robust and mechanistically diverse late-stage pipeline, encompassing metabolic, anti-amyloid, tau-targeted, and neuroinflammation-modulating therapies, is expected to drive innovation and sustain long-term market expansion.
- Safety, tolerability, and adherence challenges, particularly in elderly patients with multiple comorbidities, can influence long-term treatment persistence and real-world effectiveness.
Alzheimer’s Disease Market size and forecast
- 2025 Alzheimer’s Disease Market Size in 7MM: USD 4 billion
- 2036 Projected Alzheimer’s Disease Market Size in the 7MM: USD 40 billion
- Alzheimer’s Disease Growth Rate (2026–2036) in the 7MM: 22.7% CAGR
DelveInsight’s ‘Alzheimer’s Disease – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of Alzheimer’s Disease, historical and forecasted epidemiology, as well as the market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The Alzheimer’s Disease market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Alzheimer ’s disease patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2026 –2036) across the 7MM regions. The report highlights key unmet medical needs in Alzheimer’s disease and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Alzheimer’s Disease Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Alzheimer’s Disease Market CAGR (Forecast period) |
22.7% (2026-2036) |
|
Alzheimer’s Disease Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Alzheimer’s Disease Companies |
|
|
Alzheimer’s Disease Therapies |
|
|
Alzheimer’s Disease Market |
Segmented by
|
|
Analysis |
|
Alzheimer’s Disease Understanding and Treatment Algorithm
Alzheimer’s Disease Overview and Diagnosis
Alzheimer’s disease is an age-related neurodegenerative disease that is the most prevalent source of dementia in elders, causing significant of cases of dementia. Though primarily afflicting those aged 65 and over, Alzheimer’s disease is being increasingly observed in younger people starting from around age 40. It is progressive, leading to irreversible loss of neurons in the cerebral cortex and hippocampus. The prevalence, cost of care, impact on individuals and caregivers, and lack of mechanism-based treatments make Alzheimer’s disease one of the most challenging diseases.
The diagnosis of Alzheimer’s Disease has transitioned from a clinical diagnosis of exclusion to a proactive, biomarker-integrated framework. In the United States, diagnostic practices primarily follow the NIA-AA criteria, which categorize the disease into three distinct stages: Preclinical (asymptomatic with biomarker evidence), Mild Cognitive Impairment (MCI), and Alzheimer’s Dementia. While these criteria are foundational in research, clinical practice is increasingly adopting the DSM-5 standards, which require documented evidence of significant cognitive decline in domains such as memory or language that interferes with daily independence.
Further details are provided in the report..
Current Alzheimer’s Disease Treatment Landscape
The overall Alzheimer’s Disease treatment landscape is rapidly evolving, driven by the advancement of anti-amyloid monoclonal antibodies, cholinesterase inhibitors, and emerging tau-targeted cellular therapies. Key standard innovations in current practice include Lecanemab (LEQEMBI) and donanemab-azbt (KISUNLA) which are humanized monoclonal antibodies designed to clear amyloid-beta plaques, and benzgalantamine (ZUNVEYL) a next-gene ration acetylcholinesterase inhibitor used to manage the cognitive decline characteristic of the disease. In addition, next-generation oral amyloid inhibitors such as valiltramiprosate (ALZ-801) are designed to optimize pharmacokinetics and improve tolerability while maintaining the proven amyloid-depletion mechanism. Collectively, these therapies along with advanced tau-based approaches such as HMTM (TRx0237) are expected to transform Alzheimer’s management by improving safety, durability of response, and patient quality of life.
Further details related to country-based variations are provided in the report.
Alzheimer’s Disease Unmet Needs
The section “unmet needs of Alzheimer’s disease” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Post-transplant Relapse as the Leading Cause of Failure
- Incomplete Control of Acute and Chronic Alzheimer
- Limited Applicability in the Aging Alzheimer Population
- Delayed Immune Reconstitution and Infection Burden
- Long-term Toxicities and Secondary Malignancies
and others…..
Note: Comprehensive unmet needs insights in Alzheimer’s Disease and their strategic implications are provided in the full report.
Alzheimer’s Disease Epidemiology
Key Findings from Alzheimer’s Disease Epidemiological Analysis and Forecast
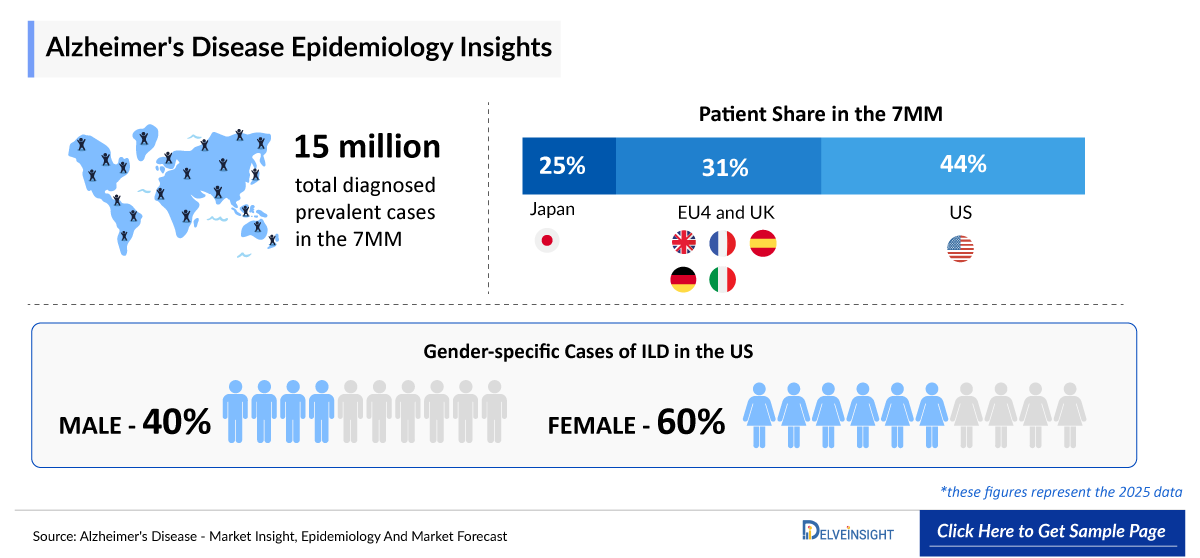
- The Total diagnosed prevalent cases of Alzheimer’s disease across the 7MM are estimated at nearly ~15 million in 2025. Among the 7MM, the United States accounts for approximately 44% of the total cases.
- In EU4 and the UK account for 31% of the total diagnosed prevalent cases. Japan accounts for 25% of the total diagnosed prevalent cases in 2025.
- The US accounted for around 7 million diagnosed prevalent cases of Alzheimer’s disease in 2025.
- In 2025, among EU4 and the UK countries, Germany accounted for the highest diagnosed prevalent cases of Alzheimer’s disease, followed by France. At the same time, the UK accounted for the fewest diagnosed prevalent cases of Alzheimer’s disease.
- In Japan, there were around ~4 million diagnosed prevalent cases of Alzheimer’s disease in 2025, expected to increase at a CAGR of 1.6% during the study period.
- In 2025, the gender-specific diagnosed prevalent cases of Alzheimer’s disease accounted for around 67% of females and 33% of males in the US.
- In EU4 and the UK, the age group 75–84 had the highest case of Alzheimer’s disease (~2 million), <65 age group had the fewest cases (~130,000) in 2025.
- In Japan, based on the severity of Alzheimer’s disease, MCI ranked first with ~3 million cases, followed by mild dementia (~710,000), moderate dementia (~370,000), and severe dementia (~200,000) in 2025. The cases are expected to increase during the forecast period.
- In 2025, The US accounted for 2,083,188 diagnosed prevalent cases of Psychosis Alzheimer’s disease and EU4 and the UK accounted for 3,056,647 diagnosed prevalent cases of agitation in Alzheimer’s disease.
Alzheimer’s Disease Drug Analysis & Competitive Landscape
The Alzheimer’s Disease drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I - Phase III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Alzheimer’s Disease treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Alzheimer’s Disease therapeutics market.
Approved Therapies for Alzheimer’s Disease
ZUNVEYL (benzgalantamine): Alpha Cognition
ZUNVEYL (benzgalantamine), formerly known as ALPHA-1062, is a prodrug of the Alzheimer’s treatment galantamine and an acetylcholinesterase inhibitors (AChEI) developed by Alpha Cognition. It is indicated for the treatment of mild to moderate dementia of the Alzheimer’s type in adults.
Note: Detailed marketed therapies assessment will be provided in the final report.
Alzheimer’s Disease Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
ZUNVEYL (benzgalantamine) |
Alpha Cognition |
Mild-to-moderate Alzheimer’s disease |
Small molecule |
Acetylcholinesterase inhibitors |
Oral |
US: 2024 |
|
LEQEMBI (lecanemab) |
Biogen/Eisai |
MCI or mild dementia |
Monoclonal antibody |
Amyloid beta-protein inhibitor |
IV infusion |
US: 2023 JP: 2023 EU: 2025 |
|
KISUNLA (donanemab-azbt) |
Eli Lilly |
MCI or mild dementia |
Monoclonal antibody |
Anti-amyloid antibody |
IV infusion |
US: 2024 EU: 2025 JP: 2024 |
|
REXULTI (brexpiprazole) |
Otsuka Pharma/ Lundbeck |
Agitation associated with dementia due to Alzheimer's disease |
Small molecule |
Dopamine partial agonist |
Oral |
US: 2023 JP: 2024 |
|
Drug E |
XX |
XX |
XX |
XX |
XX |
XX |
Alzheimer’s Disease Pipeline Analysis
Masupirdine (SUVN-502): Suven Life Sciences
Masupirdine (SUVN-502) is an investigational small-molecule therapy developed by Suven Life Sciences for the treatment of cognitive impairment and neuropsychiatric symptoms, including agitation, associated with Alzheimer's disease. It is a highly selective serotonin 5-HT6 receptor antagonist, designed to enhance neurotransmitter release and improve cognitive and behavioral functions. Masupirdine is currently being evaluated in a global Phase III clinical trial targeting agitation in Alzheimer’s disease, where effective treatment options remain limited. The Phase III trial is anticipated to read out in Q1/Q2 2025, with company expectations focusing on findings by 2027.
In November 2025, Suven Life Sciences reported that its global Phase III clinical trial of masupirdine (SUVN-502) achieved 50% patient enrollment for agitation in Alzheimer's disease, with the multicenter, randomized, double-blind, placebo-controlled study ongoing across North America and Europe.
Competitive Landscape of Alzheimerls Disease Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Masupirdine (SUVN-502) |
Suven Life Sciences |
III |
Agitation in dementia of Alzheimer’s type, moderate Alzheimer’s disease |
Oral |
5-HT6 receptor antagonist |
2028 |
|
Trontinemab (RG6102) |
Roche |
III |
Early symptomatic Alzheimer's disease (MCI to mild dementia Alzheimer’s disease), mild to moderate Alzheimer’s disease |
Oral |
Aβ-protein inhibitors |
Information is available in the full |
|
Buntanetap (ANVS401) |
Annovis Bio |
III |
Mild to moderate Alzheimer's disease/ early Alzheimer’s disease |
Oral |
Translational Inhibitor of Neurotoxic Aggregating Proteins (TINAPs) |
Information is available in the full report |
|
Drug 4 |
XX |
XX |
XX |
XX |
XX |
XX |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Alzheimer’s Disease Key Players, Market Leaders and Emerging Companies
- Bristol Myers Squibb
- Eli Lilly
- Biogen/Eisai
- Alpha Cognition
- Otsuka Pharma/ Lundbeck
- and others
Recent Alzheimer’s Disease Drug Updates
- In March 2025, Alpha Cognition announced the commercial launch of ZUNVEYL in the United State.
- In July 2024, the US FDA approved ZUNVEYL for the treatment of mild-to-moderate Alzheimer’s disease. The approval was obtained through the 505(b)(2) regulatory pathway, which allows reliance on existing safety and efficacy data from previously approved galantamine products.
- In August 2025, the US FDA approved Lecanemab-irmb (LEQEMBI IQLIK) subcutaneous injection for maintenance dosing for the treatment of patients with early Alzheimer’s disease. The approval enables a once-weekly at-home subcutaneous administration option following the initial 18-month intravenous treatment phase with LEQEMBI (lecanemab), making it the first anti-amyloid therapy to offer a self-administered maintenance regimen designed to support continued treatment in patients with MCI or mild dementia due to Alzheimer’s disease.
- In April 2025, Eisai and Biogen reported that the EC granted marketing authorization in the European Union for Lecanemab-irmb (LEQEMBI IQLIK) for Alzheimer’s disease. The approval marks the first therapy targeting an underlying cause of Alzheimer’s disease to receive authorization in the EU. LEQEMBI is indicated for the treatment of adult patients with MCI or mild dementia due to Alzheimer’s disease (early Alzheimer’s disease) who are apolipoprotein E ε4 non-carriers or heterozygotes with confirmed amyloid pathology.
Alzheimer’s Disease Market Outlook
Alzheimer’s disease is an age-related neurodegenerative disease that is the most prevalent source of dementia in elders, causing significant of cases of dementia. Though primarily afflicting those aged 65 and over, Alzheimer’s disease is being increasingly observed in younger people starting from around age 40. It is progressive, leading to irreversible loss of neurons in the cerebral cortex and hippocampus. The prevalence, cost of care, impact on individuals and caregivers, and lack of mechanism-based treatments make Alzheimer’s disease one of the most challenging diseases.
Key marketed therapies shaping current management
Benzgalantamine (ZUNVEYL)
ZUNVEYL (benzgalantamine), formerly known as ALPHA-1062, is a prodrug of the Alzheimer’s treatment galantamine and an acetylcholinesterase inhibitors (AChEI) developed by Alpha Cognition. It is indicated for the treatment of mild to moderate dementia of the Alzheimer’s type in adults.
Lecanemab (LEQEMBI)
LEQEMBI is an accelerated FDA-approved drug for the treatment of Alzheimer’s disease, specifically for patients in the Mild Cognitive Impairment (MCI) or mild dementia stages of the condition. This humanized IgG1 monoclonal Antibody (mAb) targets both soluble and insoluble forms of Amyloid β (Aβ) and was developed to slow disease progression by addressing Aβ pathology.
Donanemab (KISUNLA)
KISUNLA (donanemab-azbt) is an amyloid-targeting therapy designed for individuals with MCI and early symptomatic Alzheimer’s disease confirmed by amyloid pathology. This treatment focuses on eliminating amyloid plaques, which contribute to the progression of Alzheimer’s. Donanemab, a humanized IgG1 mAb, specifically targets Aβ proteins responsible for forming these pathology.
Overall, the launch of first-in-class therapies, improved testing, and rising disease awareness are expected to drive steady growth in the 7MM Alzheimer’s disease market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
Key Findings from Alzheimer’s disease Market Forecast Report
- Among the 7MM, the US accounted for the largest market size of Alzheimer’s disease i.e., ~USD 2 billion in 2025.
- In 2036, among all the therapies for Alzheimer’s disease, the highest revenue is estimated to be generated by disease-modifying therapies in the 7MM, with the total market valuation reaching approximately USD 40 billion.
- Japan’s Alzheimer’s disease market remains resilient and increasingly competitive, reaching USD 800 million in 2025. This market is expected to grow at a CAGR of 14.3% during the forecast period.
Further details will be provided in the report….
US administrative claims data
Overall, the launch of first-in-class therapies, improved genetic testing, and rising disease awareness are expected to drive steady growth in the 7MM Alzheimer’s Disease market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
Among the 7MM, the US is projected to account for the largest market size for Alzheimer’s, estimated at USD 2 billion in 2025.This growth is underpinned by an aging "baby boomer" population and a significant gender disparity, with females representing nearly two-thirds of the total patient burden.
In 2036, among all the therapies for Alzheimer’s in the 7MM, the highest revenue is estimated to be generated by anti-amyloid monoclonal antibodies such as Lecanemab (LEQEMBI) and Donanemab (KISUNLA). The market is expected to shift from specialized infusion centers to more accessible subcutaneous maintenance dosing, closely followed by the anticipated uptake of emerging oral small molecules like AR1001 and ALZ-801.
The most meaningful recent shift in the market has been the rapid emergence of precision diagnostics and "cocktail therapies". While traditional symptomatic agents (e.g., galantamine, memantine) currently hold a share, they show stabilized growth, whereas newer entries targeting Tau proteins and neuroinflammation are expected to dominate the long-term revenue landscape. These advancements aim to reduce the logistical burden of IV infusions and address the high unmet need for better tolerability and disease-altering potential.
Further details will be provided in the report…
Alzheimer’s Disease Drug Uptake
This section focuses on the uptake rate of potential Alzheimer’s disease drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Alzheimer’s disease drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The treatment patterns for Alzheimer’s disease in the United States and Europe were examined, highlighting a shift from symptomatic to disease-modifying therapies. Real-world evidence indicates that approximately two-thirds of newly diagnosed patients receive symptomatic treatment. According to recent studies, 75% of treated patients in certain registries were prescribed AChEIs (donepezil, rivastigmine, galantamine), while 14.7% received a combination of AChEI and memantine, and 10.3% received memantine alone. In the UK, symptomatic drug uptake varies by severity, with 54.1% in mild cases and 71.8% in moderate cases receiving treatment.
Lecanemab (LEQEMBI) and Donanemab (KISUNLA) developed by Eisai/Biogen and Eli Lilly respectively, are IV-administered anti-amyloid monoclonal antibodies (mAbs) that have redefined the landscape. LEQEMBI has emerged as a key biologic for early AD, demonstrating the ability to slow clinical decline. To improve convenience, a subcutaneous formulation Lecanemab (LEQEMBI) is expected to simplify administration. These therapies are positioned as lead assets, moving the market toward biomarker-confirmed, biologically targeted disease management.
Detailed insights of emerging therapies' drug uptake is included in the report…
Market Access and Reimbursement of Approved therapies in Alzheimer’s Disease
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
EU4 and the UK Reimbursement of Therapies Approved for Alzheimer’s Disease | |
|
Drug/Therapy |
IQWiG Assessment |
|
Lecanemab (LEQEMBI) |
In February 2026, in patients with clinically diagnosed MCI due to Alzheimer’s disease, additional benefits have not been proven. |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Alzheimer’s Disease Price Scenario & Trends
Pricing and analogue assessment of Alzheimer’s Disease therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Alzheimer’s Disease Approved Drugs
Brexpiprazole (REXULTI) is utilized for the treatment of agitation associated with dementia due to Alzheimer’s disease. Based on a structured secondary analysis integrating dosing assumptions and treatment duration, an annual cost of therapy was estimated for the US and Japan. With a US WAC of approximately USD 1,515 and a typical treatment duration of up to 12 weeks, the total per-patient treatment cost is estimated at USD 4,500. For Japan, using a comparative pricing approach based on regional price-adjustment ratios of approximately 10% of the US cost, the estimated treatment cost is assumed to be USD 450.
Industry Experts and Physician Views for Alzheimer’s Disease
To keep up with Alzheimer’s Disease market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the Alzheimer’s Disease emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Alzheimer’s Disease, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as the Memorial Sloan Kettering Cancer Center, University of California San Francisco, State University of New York Upstate Medical University, etc. were contacted.
Their opinion helps understand and validate current and emerging Alzheimer’s Disease therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Alzheimer’s Disease.
Latest KOL Views on Alzheimer’s Disease
Region |
Key Opinion Leaders (KOLs) & Subject Matter Experts (SMEs) |
|
United States |
“Alzheimer’s is a progressive neurodegenerative disorder characterized by the accumulation of amyloid-beta plaques and tau tangles. The focus has shifted toward early biomarker detection (PET scans, CSF, and blood-based tests) and the clinical implementation of disease-modifying therapies (DMTs). While cognitive decline remains the primary concern, the burden on caregivers and the need for equitable access to infusion-based treatments are critical unmet needs in the aging US population.” |
|
Germany |
“Alzheimer’s is viewed through a lens of comprehensive geriatric care and rigorous diagnostic standards. Experts emphasize the pathophysiology of neuroinflammation alongside protein misfolding. There is a strong focus on multimodal intervention strategies, combining pharmacological treatments with structured psychosocial support. German KOLs advocate for high-quality longitudinal monitoring to manage comorbidities like cardiovascular health, which significantly influence the progression of dementia.” |
Alzheimer’s Disease Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Alzheimer’s Disease, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Alzheimer’s Disease Market Report
- The Alzheimer’s Disease market report covers a segment of key events, an executive summary, a descriptive overview of Alzheimer’s Disease, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Alzheimer’s Disease market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Alzheimer’s Disease market report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Alzheimer’s Disease market.
Alzheimer’s Disease Market Report Insights
- Alzheimer’s Disease Patient population forecast
- Alzheimer’s Disease therapeutics market size
- Alzheimer’s Disease pipeline analysis
- Alzheimer’s Disease market size and trends
- Alzheimer’s Disease market opportunity (Current and forecasted)
Alzheimer’s Disease Market Report Key Strengths
- Epidemiology‑based (EPI‑based) bottom‑up forecasting
- Artificial Intelligence (AI) -enabled market research report
- 11-year forecast
- Alzheimer’s Disease market outlook (North America, Europe, Asia-Pacific)
- Alzheimer’s Disease Burden trends (by geography)
- Alzheimer’s Disease Treatment addressable Market (TAM)
- Alzheimer’s Disease Competitive Landscape
- Alzheimer’s Disease major companies Insights
- Alzheimer’s Disease Price trends and analogue assessment
- Alzheimer’s Disease Therapies Drug Adoption/Uptake
- Alzheimer’s Disease Therapies Peak Patient Share analysis
Alzheimer’s Disease Market Report Assessment
- Alzheimer’s Disease Current treatment practices
- Alzheimer’s Disease Unmet needs
- Alzheimer’s Disease Clinical development Analysis
- Alzheimer’s Disease emerging drugs product profiles
- Alzheimer’s Disease Market attractiveness
- Alzheimer’s Disease Qualitative analysis (SWOT and conjoint analysis)
Frequently Asked Questions from Alzheimer’s Disease Market
Alzheimer’s Disease Market Insights
- What was the Alzheimer’s Disease market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Alzheimer’s Disease?
- What are the disease risks, burdens, and unmet needs of Alzheimer’s Disease? What will be the growth opportunities across the 7MM concerning the patient population with Alzheimer’s Disease?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Alzheimer’s Disease? What are the current guidelines for treating Alzheimer’s Disease in the US, Europe, and Japan?
Reasons to Buy the Alzheimer’s Disease Market Report
- The Alzheimer’s Disease market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Alzheimer’s Disease market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing Alzheimer’s Disease market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming Alzheimer’s Disease copanies in the Alzheimer’s Disease market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.