Hereditary Transthyretin Amyloidosis Market Summary
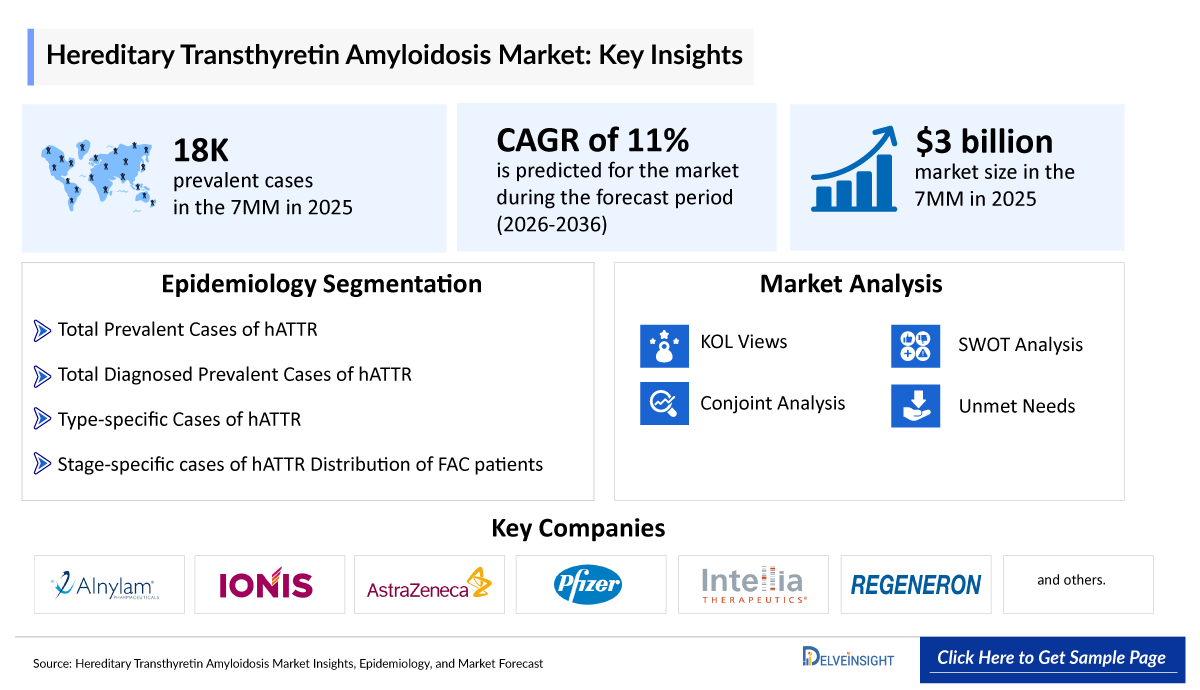
- The hereditary transthyretin amyloidosis (hATTR) market size is projected to reach approximately USD 3,000 million in 2025 and is expected to grow at a robust CAGR of 11% during 2026–2036.
- According to estimates, the United States accounted for the largest hATTR market size, reaching approximately USD 2,000 million in 2025.
- The leading hATTR companies developing therapies in the treatment market include - Alnylam Pharmaceuticals, Ionis Pharmaceuticals, AstraZeneca, Pfizer, Intellia Therapeutics, Regeneron, and others.
Hereditary Transthyretin Amyloidosis (hATTR) Market & Epidemiology Insights
- hATTR is caused by a change or mutation in the transthyretin (TTR) gene, which is inherited (i.e., runs in families). This change makes the TTR protein unstable, so it folds the wrong way and attaches and builds up in different organs and tissues as amyloid.
- hATTR can present as polyneuropathy (hATTR-PN), primarily affecting the peripheral nervous system and leading to progressive motor decline with fatal outcomes within about ten years if untreated. Transthyretin amyloidosis–related cardiomyopathy, whether hereditary (hATTR-CM) or wild-type (ATTRwt-CM), primarily affects the heart, presenting as restrictive cardiomyopathy that can progress to heart failure and may result in death within about five years if untreated.
- The rarity and clinical heterogeneity of hATTR amyloidosis complicate diagnosis, which is confirmed by Congo red–stained biopsy of affected tissue, with less invasive methods such as salivary gland, gastric mucosa, or fat aspiration biopsies increasingly utilised.
- The hATTR mutation has a 50% inheritance risk, but variable penetrance means symptoms may not appear; therefore, close monitoring is essential, as asymptomatic carriers can still pass the disease to their children.
- The typical age for patients with the T80A (formerly known as T60A) mutation to start showing symptoms is usually between 45 and 78, most often after the age of 60.
- Management focuses on disease-modifying therapies (TTR stabilisers and gene-silencing agents), along with symptomatic care and, in select cases, organ transplantation to slow progression and improve outcomes.
- The current treatment landscape includes US FDA-approved drugs such as vutrisiran (AMVUTTRA), patisiran (ONPATTRO), inotersen (TEGSEDI), tafamidis (VYNDAMAX), and eplontersen (WAINUA), among others.
- There is no FDA-approved curative therapy for hATTR; current treatments focus on stabilising or reducing abnormal TTR production.
- Available therapies, including TTR stabilisers and gene silencers, are effective in slowing disease progression, particularly when started early. However, they do not reverse existing nerve or cardiac damage, highlighting a key unmet need.
- Akcea Therapeutics discontinued the commercial availability of Tegsedi (inotersen) in the United States on September 27, 2024, due to low market utilisation. This decision was not caused by safety or manufacturing issues. Patients must transition to other treatments under the guidance of a healthcare provider, with support available until early 2025.
- Several potential therapies are being investigated for the management of hATTR. The anticipated launch of promising candidates like nexiguran ziclumeran (nex-z), nucresiran, and others during the forecast period (2026–2036) is expected to bring about a significant shift in the market dynamics of hATTR.
Hereditary Transthyretin Amyloidosis (hATTR) Market Size and Forecast in the 7MM
- 2025 Projected hATTR Market Size: USD 3,000 million
- 2036 Projected hATTR Market Size: USD XXX million
- hATTR Growth Rate (2026–2036): ~11% CAGR
Request a sample to unlock the CAGR for "Hereditary Transthyretin Amyloidosis (hATTR) Market Forecast"
Key Factors Driving the Hereditary Transthyretin Amyloidosis (hATTR) Market
DelveInsight's ‘Hereditary Transthyretin Amyloidosis (hATTR) Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the hATTR, historical and forecasted epidemiology, as well as the hATTR therapeutics market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The Hereditary Transthyretin Amyloidosis (hATTR) market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates hATTR patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Hereditary Transthyretin Amyloidosis (hATTR) and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the hATTR Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
hATTR Market CAGR (Study period/Forecast period) |
~11% (2026–2036) |
|
hATTR Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
hATTR Companies |
|
|
hATTR Therapies |
|
|
hATTR Market |
Segmented by
|
|
Analysis |
|
Hereditary Transthyretin Amyloidosis (hATTR) Disease Understanding
Hereditary Transthyretin Amyloidosis (hATTR) Overview and Diagnosis
Hereditary Transthyretin Amyloidosis (hATTR) is a rare, inherited disorder caused by mutations in the transthyretin (TTR) gene, leading to misfolded protein deposits (amyloid) in tissues. It primarily affects the peripheral nerves (hATTR-PN) and the heart (hATTR-CM), resulting in progressive neuropathy and cardiomyopathy. The disease shows variable penetrance and clinical heterogeneity, often causing delays in diagnosis. If untreated, hATTR can lead to severe disability and early mortality. Current treatments focus on stabilising or reducing TTR protein production to slow disease progression.
Hereditary Transthyretin Amyloidosis (hATTR) Diagnosis
Diagnosis is often challenging due to its clinical heterogeneity and overlap with other neuropathies and cardiomyopathies. It is confirmed through genetic testing to identify pathogenic TTR gene mutations, along with tissue biopsy demonstrating amyloid deposits using Congo red staining with apple-green birefringence under polarised light. Common biopsy sites include the abdominal fat pad, salivary gland, gastric mucosa, or affected nerve tissue, offering both invasive and less invasive diagnostic options. In cases of cardiac involvement, echocardiography, cardiac MRI, and nuclear scintigraphy (e.g., bone-avid tracers) help detect transthyretin cardiac amyloid deposition. Early and accurate diagnosis is critical to initiate therapy before irreversible neurological or cardiac damage occurs.
Further details are provided in the report.
Hereditary Transthyretin Amyloidosis (hATTR) Treatment
Treatment focuses on reducing, stabilising, or silencing transthyretin (TTR) protein production to slow disease progression. Approved therapies include TTR stabilisers (tafamidis) and gene-silencing agents (patisiran, inotersen, vutrisiran, eplontersen) that reduce abnormal TTR levels. These treatments are most effective when initiated early and primarily aim to delay neurological and cardiac decline rather than reverse existing damage. Supportive care is also used to manage symptoms and improve quality of life.
Further details related to country-based variations are provided in the report.
Hereditary Transthyretin Amyloidosis (hATTR) Unmet Needs
The section “unmet needs of Hereditary Transthyretin Amyloidosis (hATTR)” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- No curative therapy available; current treatments only slow disease progression
- Irreversible organ damage (nerve and cardiac) at diagnosis limits treatment benefit
- Delayed and difficult diagnosis due to non-specific symptoms and disease heterogeneity
- Need for therapies that effectively treat both neuropathy and cardiomyopathy together
- High treatment cost and limited accessibility in many regions, and others…..
Comprehensive unmet needs insights in Hereditary Transthyretin Amyloidosis (hATTR) and their strategic implications are provided in the full report.
Hereditary Transthyretin Amyloidosis (hATTR) Epidemiology
The hATTR epidemiology section provides insights about the historical and current hATTR patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the hATTR market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings from Hereditary Transthyretin Amyloidosis (hATTR) Epidemiological Analysis and Forecast
- As per DelveInsight’s analysis, the US accounted for nearly 65% of the total diagnosed prevalent cases of hATTR in the 7MM in 2025, which is expected to increase further by 2036.
- The secondary analysis indicates that hATTR generally affects both males and females equally, with no clear gender predominance. However, a possible parent-of-origin effect is suggested, as maternal inheritance may increase disease risk. In contrast, late-onset familial cases tend to show a higher prevalence among males.
- A Japan-based study revealed that late-onset and nonendemic cases of hATTR are more common than previously recognised. Disease severity by ambulatory status showed 59% in stage 1, 19% in stage 2, and 14% in stage 3.
- In 2025, France recorded the highest diagnosed prevalence of hATTR among other EU4 countries and the United Kingdom.
- In the USA, the highest proportion of hATTR cases is seen in Familial Amyloid Polyneuropathy (FAP) at 45%, followed by mixed hATTR, while the lowest proportion is observed in Familial Amyloid Cardiomyopathy (FAC).
- In the USA, the New York Heart Association (NYHA) classification of FAC shows that the majority of patients fall under Class II (60%), followed by Class III, while the lowest proportion is seen in Class IV.
Hereditary Transthyretin Amyloidosis Epidemiology Segmentation
- Total Prevalent Cases of hATTR
- Total Diagnosed Prevalent Cases of hATTR
- Type-specific Diagnosed Prevalent Cases of hATTR
- Stage-specific Diagnosed Prevalent Cases of FAP
- Distribution of FAC Patients by New York Heart Association (NYHA) Criteria
Recent Developments in the Hereditary Transthyretin Amyloidosis (hATTR) Treatment Marktet
- In March 2026, Intellia Therapeutics presentation, the Nex-Z ATTR program outlines a planned resumption of patient enrollment in the Phase III MAGNITUDE trial in ATTR-CM, resumption of patient enrollment in the Phase III MAGNITUDE-2 trial in ATTRv-PN, and expected completion of enrollment in MAGNITUDE-2 in the second half of 2026.
- In February 2026, according to the Q4 and Full Year 2025 financial results of Alnylam Pharmaceuticals, the company plans to launch Nucresiran, a next-generation RNA silencer for Transthyretin Amyloidosis, with a potential launch timeline of 2028 for polyneuropathy and 2030 for cardiomyopathy indications.
- As per the Ionis Pharmaceuticals Q4 2025 presentation, eplontersen is highlighted as a key asset, with an NDA submission expected in 2026 and a potential launch anticipated in 2027 for hATTR-CM.
- In August 2025, Pfizer announced that it would discontinue VYNDAQEL (tafamidis meglumine) in the United States effective December 31, 2025, while confirming that VYNDAMAX (tafamidis) would continue to remain available for all eligible patients with a prescription.
- In March 2025, Alnylam Pharmaceuticals announced that the US FDA approval of the supplemental New Drug Application (sNDA) for its RNAi therapeutic, AMVUTTRA (vutrisiran), for the treatment of the cardiomyopathy of wild-type or hATTR-CM in adults to reduce cardiovascular mortality, cardiovascular hospitalisations, and urgent heart failure visits. The approval expands the indication for AMVUTTRA, which now becomes the first and only therapeutic approved by the FDA for the treatment of ATTR-CM and the polyneuropathy of hATTR-PN in adults.
Numbers are subject to change with report updation, clinical information updates, etc.
Hereditary Transthyretin Amyloidosis (hATTR) Drug Chapters & Competitive Analysis
The hATTR drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III hATTR clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the hATTR treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the hATTR therapeutics market.
Approved Therapies for Hereditary Transthyretin Amyloidosis (hATTR)
Vutrisiran (AMVUTTRA): Alnylam Pharmaceuticals
Vutrisiran (AMVUTTRA) is a SC administered transthyretin-directed small interfering ribonucleic acid (siRNA) therapeutic (also called RNA interference, or RNAi therapeutic) being developed by Alnylam Pharmaceuticals, Inc. for the treatment of ATTR amyloidosis, including hATTR amyloidosis and wild-type ATTR (wtATTR) amyloidosis. Vutrisiran was approved in June 2022 in the USA for the treatment of the polyneuropathy of hATTR amyloidosis in adults and received a positive opinion in the EU in July 2022 for the treatment of hATTR amyloidosis in adult patients with stage 1 or stage 2 polyneuropathy. Vutrisiran is also under regulatory review for the treatment of the polyneuropathy of hATTR amyloidosis in adults in Japan. This article summarises the milestones in the development of vutrisiran leading to this first approval in hATTR amyloidosis.
Eplontersen (WAINUA/WAINZUA): Ionis Pharmaceuticals and AstraZeneca
WAINUA/WAINZUA (eplontersen) is a ligand-conjugated antisense oligonucleotide (LICA medicine designed to reduce the production of transthyretin, or TTR protein. WAINUA has been approved in the US for the treatment of the polyneuropathy of hereditary transthyretin-mediated amyloidosis in adults. Eplontersen is an antisense oligonucleotide-GalNAc conjugate that causes degradation of mutant and wild-type TTR mRNA through binding to the TTR mRNA, which results in a reduction of serum TTR protein and TTR protein deposits in tissues.
hATTR Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Vutrisiran (AMVUTTRA) |
Alnylam Pharmaceuticals |
hATTR-PN/CM |
RNAi (siRNA) |
SC |
TTR gene silencing |
US: 2022; EU: 2022; JP: 2022 |
|
Patisiran (ONPATTRO) |
Alnylam Pharmaceuticals |
hATTR-PN |
RNAi (siRNA) |
IV infusion |
TTR gene silencing |
US: 2018; EU: 2018; JP: 2019 |
|
Inotersen (TEGSEDI) |
Ionis Pharmaceuticals |
hATTR-PN |
Antisense oligonucleotide |
SC |
TTR mRNA inhibition |
EU: 2018 |
|
Eplontersen (WAINUA) |
Ionis Pharmaceuticals and AstraZeneca |
hATTR-PN |
Antisense oligonucleotide |
SC |
TTR mRNA inhibition |
US: 2023; EU: 2025 |
|
Tafamidis (VYNDAMAX) |
Pfizer |
hATTR-CM |
Small molecule |
Oral |
TTR stabilizer |
US: 2019; EU: 2019; JP: 2019 |
Note: Detailed marketed therapies assessment of therapies will be provided in the final report.
Hereditary Transthyretin Amyloidosis (hATTR) Pipeline Analysis
Nexiguran ziclumeran (nex-z): Intellia Therapeutics and Regeneron
Nexiguran ziclumeran (nex-z), also known as NTLA-2001, is an investigational in vivo CRISPR-based gene editing therapy being developed by Intellia Therapeutics as a one-time treatment for transthyretin (ATTR) amyloidosis. It is designed to inactivate the TTR gene, which encodes the TTR protein. Intellia is leading the development and commercialisation of nex-z as part of a collaboration with Regeneron. The drug is currently active in Phase III for transthyretin (ATTR) amyloidosis.
NEX-Z has received Orphan Drug (ODD) and Regenerative Medicine Advanced Therapy (RMAT) designations from the US FDA and has also been granted ODD by the European Commission, highlighting regulatory support for its development as a potential therapy for rare diseases.
Nucresiran: Alnylam Pharmaceuticals
Nucresiran is an investigational RNA interference (RNAi) therapy being developed by Alnylam Pharmaceuticals for the treatment of Transthyretin Amyloidosis. It aims to reduce hepatic production of transthyretin protein and is currently being evaluated in clinical trials for ATTR cardiomyopathy and polyneuropathy.
It is currently in Phase III clinical development for ATTR-CM and hATTR-PN.
Comparison of Emerging hATTR Drugs Under Development | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
Nexiguran ziclumeran (nex-z) |
Intellia Therapeutics and Regeneron |
III |
hATTR-PN and hATTR-CM |
IV infusion |
Targets the TTR gene |
Gene Therapy |
Information is available in the full report |
|
Nucresiran |
Alnylam Pharmaceuticals |
III |
hATTR-PN and hATTR-CM (ATTR Amyloidosis) |
SC |
Targeting and silencing messenger RNA (mRNA) |
Small interfering RNA (siRNA) |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Note: A detailed emerging therapies assessment will be provided in the final report
Hereditary Transthyretin Amyloidosis (hATTR) Key Players, Market Leaders, and Emerging Companies
- Alnylam Pharmaceuticals
- Ionis Pharmaceuticals
- AstraZeneca
- Pfizer
- Intellia Therapeutics
- Regeneron, and others
Hereditary Transthyretin Amyloidosis (hATTR) Market Outlook
Although there is currently no curative therapy for hATTR, early diagnosis and timely intervention play a critical role in improving patient outcomes and slowing disease progression. Current disease-modifying treatments include TTR stabilisers such as tafamidis, which prevent TTR protein misfolding and subsequent amyloid fibril formation, thereby reducing progression of organ damage, particularly in cardiomyopathy. Gene-silencing therapies, including patisiran, inotersen, eplontersen, and vutrisiran, act by reducing hepatic production of both mutant and wild-type TTR protein, leading to a significant decrease in circulating amyloidogenic protein levels and slowing neuropathy progression. While no approved therapies specifically target amyloid fibril clearance, this remains an important emerging area of research with several investigational approaches aimed at enhancing amyloid removal.
Importantly, the emerging pipeline is increasingly focused on next-generation disease-modifying and potentially curative approaches, including CRISPR-based gene-editing therapies and novel RNA-targeting platforms designed to deliver deeper and more durable TTR suppression.
Liver transplantation, once considered a treatment option to eliminate mutant TTR production, is now rarely used due to the availability of effective RNA-based therapies. In addition, comprehensive symptomatic management remains essential, addressing neuropathic pain, autonomic dysfunction, cardiac complications, and overall functional impairment to improve quality of life. The next major evolution in the treatment landscape is gene-editing therapy, exemplified by Nexiguran ziclumeran, a CRISPR-Cas9–based approach designed to permanently reduce TTR production and potentially offer a one-time curative strategy for hATTR amyloidosis.
Overall, the launch of first-in-class therapies, improved diagnostic approaches, and increasing disease awareness are expected to drive steady growth in the 7MM hATTR market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- According to the estimates, the largest market size of hATTR was captured by the United States, i.e., ~USD 2,000 million in 2025.
- The hATTR market is shifting toward targeted therapies, especially RNA-based and gene-editing treatments, driven by improved efficacy, safety, and broader use across earlier disease stages.
- Regional market dynamics show the US leading in revenue, while Japan and EU markets demonstrate steady growth, reflecting increasing diagnosis rates, broader pediatric access, and rising adoption of novel mechanisms.
Numbers are subject to change with report updation, clinical information updates, etc. Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Hereditary Transthyretin Amyloidosis (hATTR) (2022–2036 Forecast)
The hATTR market comprises targeted small molecules and biologics, alongside emerging gene-silencing and gene-editing therapies, each addressing transthyretin (TTR) production, stabilisation, or clearance pathways driving disease progression.
- Targeted small molecules: Tafamidis (VYNDAMAX) stabilises the TTR protein to prevent misfolding and amyloid formation, particularly in hATTR cardiomyopathy, supporting its role as a key oral disease-modifying therapy.
- RNA-based therapies (gene silencing): Agents such as patisiran, inotersen, eplontersen, and vutrisiran reduce hepatic production of TTR via siRNA or antisense oligonucleotide mechanisms, forming the backbone of modern treatment for hATTR polyneuropathy and cardiomyopathy.
- Emerging pipeline therapies: Next-generation approaches include CRISPR-Cas9 gene-editing therapy (Nexiguran ziclumeran) and novel siRNA candidates such as nucresiran, aiming for deeper and potentially long-lasting suppression of TTR.
Overall, the hATTR landscape is transitioning from TTR stabilisation toward durable gene-silencing and curative gene-editing strategies, with biologics and RNA-based therapies driving the current standard of care while pipeline agents define future growth.
Further details will be provided in the report….
Hereditary Transthyretin Amyloidosis (hATTR) Drug Uptake
This section focuses on the uptake rate of potential hATTR drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the hATTR market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The hATTR drug uptake is steadily increasing, driven by improved diagnosis, genetic testing, and growing use of disease-modifying therapies. RNA-based therapies such as vutrisiran, patisiran, inotersen, and eplontersen are witnessing the fastest uptake due to strong efficacy and expanding use in both hATTR-PN and ATTR-CM, while the TTR stabiliser tafamidis continues to hold a significant share, particularly in cardiomyopathy patients. Uptake of antisense oligonucleotides remains moderate due to safety monitoring requirements and competition from RNAi agents. Meanwhile, emerging gene-editing therapies, including CRISPR-based approaches like Nexiguran ziclumeran, are still in early clinical stages but are expected to drive future transformative growth as potential one-time curative options. Overall, the market is transitioning from stabiliser-led therapy to RNA-silencing dominance, with long-acting and curative therapies expected to define the next growth phase.
Further detailed analysis of emerging therapies' drug uptake in the report…
hATTR Market Access and Reimbursement Scenario
-
The United States
The US Reimbursement for hATTR Therapies | |
|
Drug |
Access Program |
|
Vutrisiran (AMVUTTRA) |
Alnylam Assist Copay Program |
|
Patisiran (ONPATTRO) |
Alnylam Assist |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report….
Hereditary Transthyretin Amyloidosis (hATTR) Therapies Price Scenario & Trends
Pricing and analogue assessment of hATTR therapies highlights evolving price dynamics structures. This section summarises the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and the understanding of how pricing influences market access, adherence, and long-term uptake.
Further details are provided in the final report….
Industry Experts and Physician Views for Hereditary Transthyretin Amyloidosis (hATTR)
To keep up with hATTR market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the hATTR emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in hATTR, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centres such as the University of North Carolina at Chapel Hill, the Berlin Institute of Health at Charité, and the University of Nottingham, etc. were contacted. Their opinion helps understand and validate current and emerging hATTR therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritisation in hATTR.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Genetic testing should be considered in anyone suspected of having ATTR amyloidosis and is crucial for diagnosis. It helps determine if the patient has the hereditary form or the acquired (wild-type) form” |
|
Germany |
“Affected patients develop proximal weakness and myogenic changes, which may not always be easy to distinguish clinically. Systemic amyloid progressively impairs multiple organs, leading to disability and death.” |
hATTR Report Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Hereditary Transthyretin Amyloidosis (hATTR), strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyses emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyse the effectiveness of therapy.
The team of analysts analyses promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the hATTR Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Hereditary Transthyretin Amyloidosis (hATTR), explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborate profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Hereditary Transthyretin Amyloidosis (hATTR) market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Hereditary Transthyretin Amyloidosis (hATTR) market.
hATTR Market Report Insights
- Hereditary Transthyretin Amyloidosis (hATTR) Patient Population Forecast
- Hereditary Transthyretin Amyloidosis (hATTR) Therapeutics Market Size
- Hereditary Transthyretin Amyloidosis (hATTR) Pipeline Analysis
- Hereditary Transthyretin Amyloidosis (hATTR) Market Size and Trends
- Hereditary Transthyretin Amyloidosis (hATTR) Market Opportunity (Current and forecasted)
hATTR Market Report Key Strengths
- hATTR Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Hereditary Transthyretin Amyloidosis (hATTR) Market Outlook (North America, Europe, Asia-Pacific)
- hATTR Patient Burden Trends (by geography)
- Hereditary Transthyretin Amyloidosis (hATTR) Treatment Addressable Market (TAM)
- Hereditary Transthyretin Amyloidosis (hATTR) Competitive Landscape
- Hereditary Transthyretin Amyloidosis (hATTR) Major Companies Insights
- Hereditary Transthyretin Amyloidosis (hATTR) Price Trends and Analogue Assessment
- Hereditary Transthyretin Amyloidosis (hATTR) Therapies Drug Adoption/Uptake
- Hereditary Transthyretin Amyloidosis (hATTR) Therapies Peak Patient Share analysis
hATTR Market Report Assessment
- Hereditary Transthyretin Amyloidosis (hATTR) Current Treatment Practices
- Hereditary Transthyretin Amyloidosis (hATTR) Unmet Needs
- Hereditary Transthyretin Amyloidosis (hATTR) Clinical Development Analysis
- Hereditary Transthyretin Amyloidosis (hATTR) Emerging Drugs Product Profiles
- Hereditary Transthyretin Amyloidosis (hATTR) Market Attractiveness
- Hereditary Transthyretin Amyloidosis (hATTR) Qualitative Analysis (SWOT and Conjoint Analysis)
- hATTR Market Drivers
- hATTR hATTR Barriers
FAQs Related to hATTR Market Report:
hATTR Market Insights
- What was the Hereditary Transthyretin Amyloidosis (hATTR) market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Hereditary Transthyretin Amyloidosis (hATTR)?
- What are the disease risks, burdens, and unmet needs of Hereditary Transthyretin Amyloidosis (hATTR)? What will be the growth opportunities across the 7MM concerning the with Hereditary Transthyretin Amyloidosis (hATTR) patient population?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Hereditary Transthyretin Amyloidosis (hATTR)? What are the current guidelines for treating Hereditary Transthyretin Amyloidosis (hATTR) in the US, Europe, and Japan?
Reasons to Buy hATTR Market Forecast Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Hereditary Transthyretin Amyloidosis (hATTR) market.
- Bottom-up forecasting builds from the affected population to product forecasts, delivering a robust, data-driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)-enabled report summarises and simplifies complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data-driven decisions.

-pipeline.png&w=256&q=75)


