Multiple Myeloma Market
Multiple Myeloma Insights and Trends
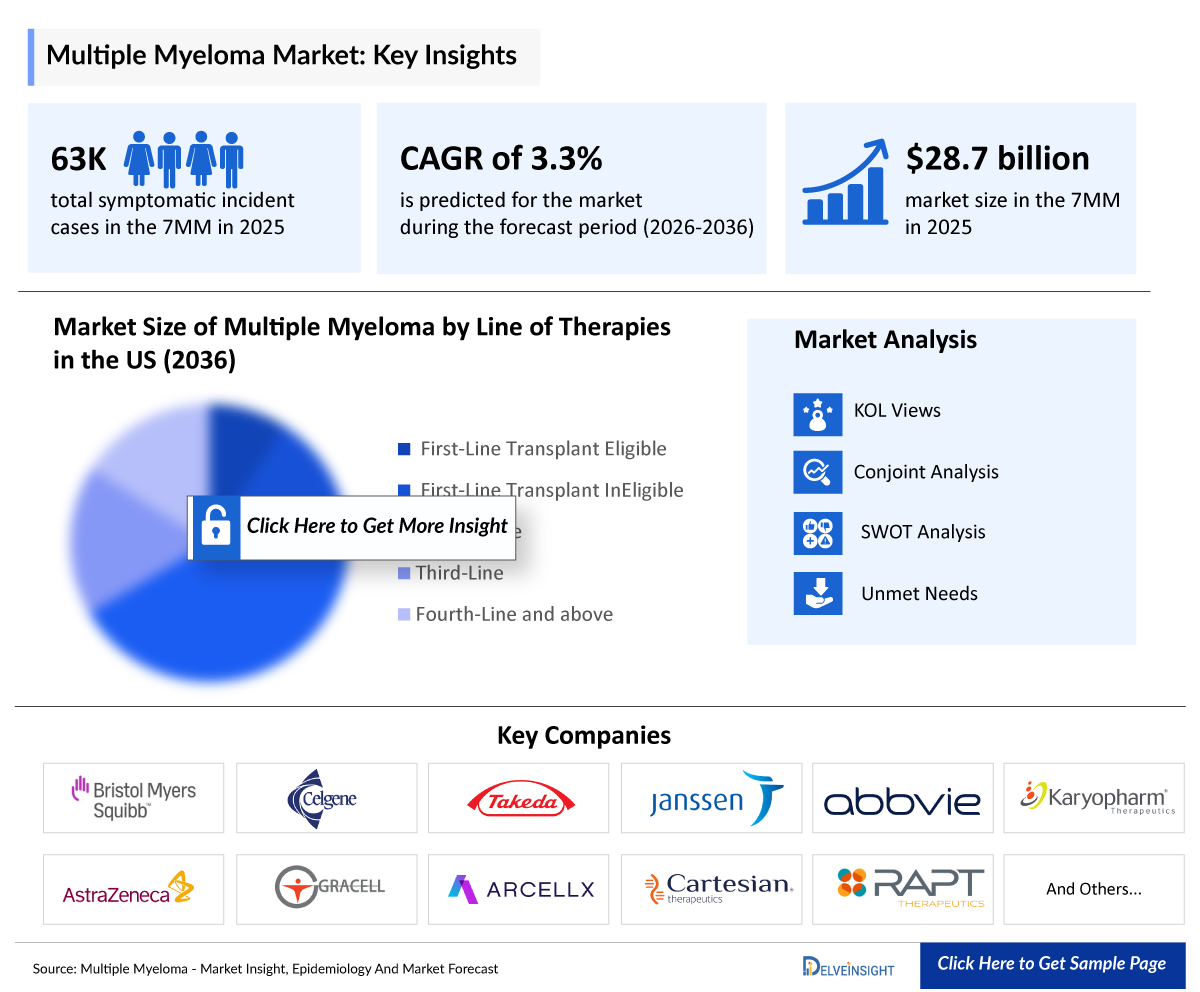
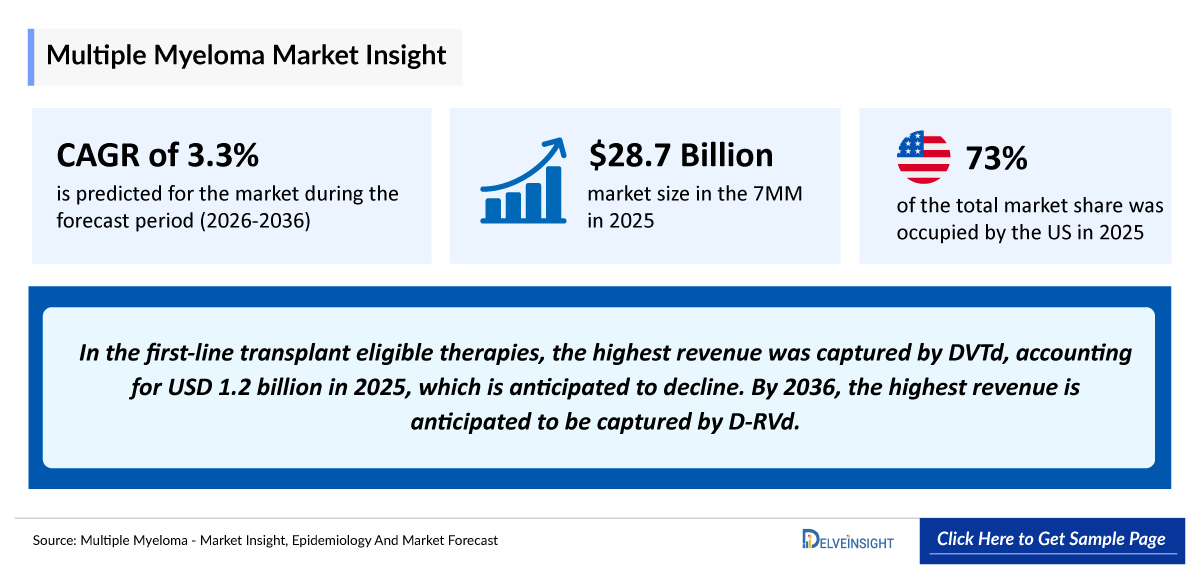
- According to DelveInsight’s analysis, multiple myeloma market size was found to be USD 28,722 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- Despite therapeutic advances, disease relapse remains common due to clonal heterogeneity and evolving resistance mechanisms, maintaining the need for continuous innovation in treatment sequencing.
- MRD (minimal residual disease) assessment is emerging as a critical biomarker, with increasing interest in MRD-guided treatment strategies to optimize therapy duration and improve long-term disease control.
- Treatment strategies for multiple myeloma include stem cell transplantation, chemotherapy, targeted therapies, corticosteroids, proteasome inhibitors, immunomodulatory agents, monoclonal antibodies, surgery, and radiation therapy. Advances in these therapeutic approaches and the approval of novel agents have significantly contributed to improved survival outcomes for patients with multiple myeloma.
- The treatment landscape for multiple myeloma has expanded significantly with multiple drug classes targeting distinct disease pathways. Key therapies include immunomodulatory drugs (IMiDs) such as Lenalidomide (REVLIMID) and Pomalidomide (POMALYST), anti-CD38 monoclonal antibodies including Daratumumab (DARZALEX) and Isatuximab (SARCLISA), SLAMF7-targeting antibodies such as Elotuzumab (EMPLICITI), and proteasome inhibitors including Bortezomib (VELCADE), Carfilzomib (KYPROLIS), and Ixazomib (NINLARO). While these therapies have significantly improved survival outcomes over the past decade, disease relapse remains common after initial treatment.
- Triplet therapy remains the backbone of multiple myeloma management, typically combining an IMiD, a proteasome inhibitor, and a corticosteroid (commonly dexamethasone). More recently, quadruplet regimens incorporating an anti-CD38 monoclonal antibody have gained prominence, reflecting a shift toward antibody-based combination therapies in earlier treatment lines to achieve deeper and more durable responses.
- Assessment of prior treatment impact in multiple myeloma remains challenging due to the lack of a standardized definition of lines of therapy (LoT).
- Trispecific antibodies are emerging as a next-generation immunotherapy approach in the multiple myeloma landscape. Several trispecific candidates are currently being explored in early-stage clinical research.
- Additionally, emerging assets include AZD0120, anito-cel, etentamig, iberdomide, and others.
Multiple Myeloma Market size and forecast
- 2025 Multiple Myeloma Market Size: USD 28,722 million
- 2036 Projected Multiple Myeloma Market Size: USD 43,578 million
- Multiple Myeloma Growth Rate (2026–2036): 3.3% CAGR
DelveInsight's ‘Multiple Myeloma Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the multiple myeloma, historical and forecasted epidemiology, as well as the multiple myeloma market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The multiple myeloma market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, multiple myeloma patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in multiple myeloma and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Geography Covered
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Multiple Myeloma Market CAGR (Study period/Forecast period) |
5.7% (2022–2036) |
|
Multiple Myeloma Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Multiple Myeloma Companies |
|
|
Multiple Myeloma Therapies |
|
|
Multiple Myeloma Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Multiple Myeloma Market
Rising Disease Incidence and Aging Population
The increasing prevalence of multiple myeloma, particularly among the elderly population, is a primary driver of market growth. As incidence rates rise globally due to aging demographics and improved survival, the demand for effective therapies continues to expand.
Advancements in Treatment Modalities
The evolution of novel therapies including proteasome inhibitors, immunomodulatory drugs, monoclonal antibodies, CAR-T cell therapies, and bispecific antibodies has significantly transformed treatment paradigms and improved patient outcomes
Improved Diagnosis and Awareness
Enhanced diagnostic capabilities and increasing awareness initiatives are enabling earlier detection and treatment, thereby increasing the treated patient pool and supporting market growth.
Multiple Myeloma Understanding and Treatment Algorithm
Multiple Myeloma Overview and Diagnosis
Multiple myeloma is the second most prevalent hematological malignancy worldwide, with a median onset of 60 years. This incurable malignancy develops from accumulating terminally differentiated monoclonal plasma cells in the bone marrow. Multiple myeloma is a malignant disorder characterized by uncontrolled proliferation of clonal plasma cells, causing various complications leading to organ dysfunction and eventually death. MM has a record for 10% of hematological malignancies. It grows from a premalignant condition called monoclonal gammopathy of undetermined implication (MGUS). This is found in 3% of those over 50 and progresses to myeloma at a rate of 1% per year. Common symptoms include bone pain (particularly in the spine or ribs), fatigue due to anemia, recurrent infections, and complications such as hypercalcemia and kidney dysfunction.
Diagnosis is based on a combination of blood and urine tests to detect monoclonal proteins, bone marrow biopsy to confirm clonal plasma cells, and imaging techniques such as X-ray, CT, or MRI to identify bone lesions.
Further details are provided in the report.
Current Multiple Myeloma Treatment Landscape
Treatment of multiple myeloma depends on the stage of the disease, patient eligibility for transplantation, and prior therapies. For newly diagnosed patients eligible for transplant, induction therapy with a combination of proteasome inhibitors, immunomodulatory drugs, and corticosteroids is typically followed by Autologous Stem Cell Transplantation (ASCT) and maintenance therapy to prolong remission. Patients who are not eligible for transplantation generally receive combination regimens consisting of proteasome inhibitors, immunomodulatory agents, monoclonal antibodies, and steroids.
For patients with relapsed or refractory disease, treatment options include next-generation immunotherapies such as monoclonal antibodies, bispecific antibodies, and CAR-T Cell Therapy, which target specific antigens on malignant plasma cells and enhance immune-mediated tumor destruction. Recent advances in genomic and molecular profiling have also improved the understanding of high-risk cytogenetic abnormalities, enabling more personalized treatment strategies.
Further details related to country-based variations are provided in the report.
Multiple Myeloma Unmet Needs
The section “unmet needs of multiple myeloma” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Delayed Diagnosis and Suboptimal Early Detection
- High Early Mortality Despite Therapeutic Advances
- Lack of Curative Therapies and Limited Long-term Remission
- Drug Resistance and Relapse and others…..
Note: Comprehensive unmet needs insights in multiple myeloma and their strategic implications are provided in the full report.
Multiple Myeloma Epidemiology
Key Findings from Multiple Myeloma Epidemiological Analysis and Forecast
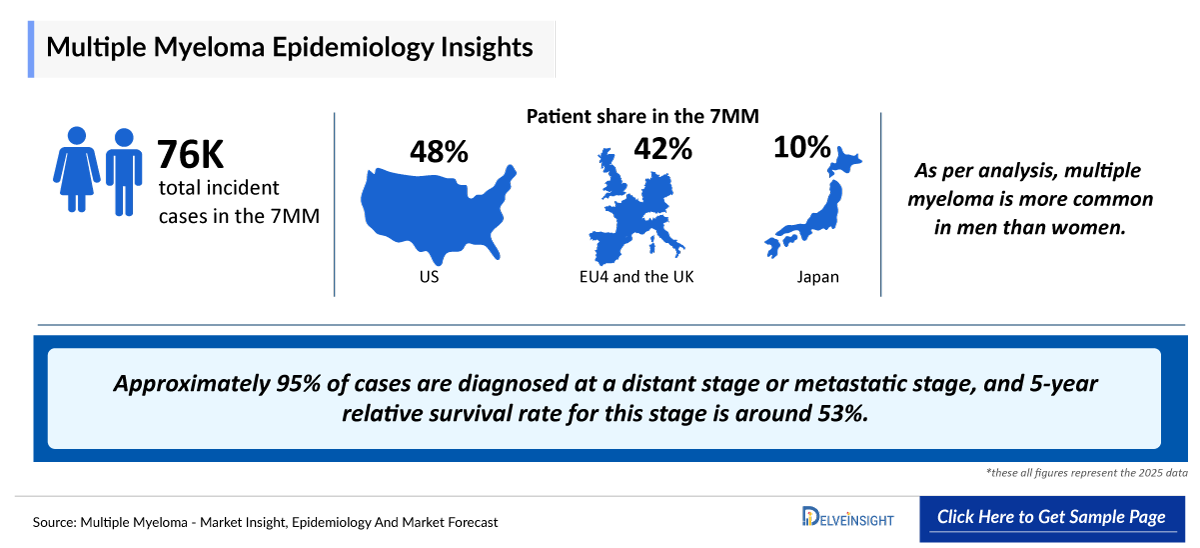
- In the 7MM, the US recorded the highest number of Incident cases of multiple myeloma accounting for 34,097 cases in 2025, which are expected to continue increasing through 2036.
- Among the EU4 and the UK, Germany accounted for the highest number of multiple myeloma cases, followed by the UK and Germany, whereas Spain accounted for the lowest number of cases in 2025.
- In the US, the 65+ age group accounted for ~73% of multiple myeloma cases in 2025, while patients 55–64 years represented ~17%, and <54 years accounted for only ~10%.
- Incident cases of multiple myeloma in the US represent a significant and gradually increasing disease burden, largely driven by an aging population and improved survival outcomes resulting from advances in therapeutic options.
- The presence of a large precursor population, including MGUS and smoldering multiple myeloma, highlights a considerable latent pool of individuals at risk of progression. Over time, a proportion of these patients transition to symptomatic disease, which contributes to the sustained growth of the diagnosed multiple myeloma patient population.
Multiple Myeloma Drug Analysis & Competitive Landscape
The multiple myeloma drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the multiple myeloma treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the multiple myeloma market.
Approved Therapies for Multiple Myeloma
LYNOZYFIC (linvoseltamab): Regeneron Pharmaceuticals
LYNOZYFIC is a bispecific B-cell Maturation Antigen (BCMA)-directed CD3 TCE indicated for the treatment of adult patients with relapsed or refractory multiple myeloma who have received at least four prior lines of therapy, including a proteasome inhibitor, an IMiD agent, and an anti-CD38 monoclonal antibody.
Regeneron Corporate Presentation of January 2026: Four registrational trials underway and four more trials to be expected to be initiated in 2026, these are LINKER-MM5 (initiated), LINKER-MM7, LINKER-MM8, and LINKER-SMM2. Also, Pivotal data anticipated starting in 2027.
SARCLISA (isatuximab-irfc): Sanofi
SARCLISA (isatuximab) is a CD38 monoclonal antibody that binds to a specific epitope on the CD38 receptor on multiple myeloma cells, inducing distinct antitumor activity. It is designed to work through multiple mechanisms of action, including programmed tumor cell death (apoptosis) and immunomodulatory activity.
According to the company’s presentation, SARCLISA is being evaluated in a subcutaneous formulation for RRMM, with anticipated approval in the United States, Europe, and Japan in H1 2026.
|
Multiple Myeloma Marketed/Approved Therapies | ||||||||
|
Drugs |
Company Name |
Combination |
MoA |
US |
EU4 & the UK |
JP |
Patient Segment |
Line of Therapy |
|
LYNOZYFIC |
Regeneron Pharmaceuticals |
Monotherapy |
BCMA × CD3 |
2025 |
2025 |
- |
RRMM, NDMM, HR-SMM, HR-MGUS, and NHR-SMM |
4L+ |
|
POMALYST |
Bristol-Myers Squibb (Celgene) |
Dexamethasone |
IMiDS |
2013 (RRMM) |
2013 (RRMM) |
2013 (RRMM) |
RRMM |
2L+ |
|
Bortezomib + dexamethasone |
- |
2019 (RRMM) |
- |
RRMM |
2L+ | |||
|
VELCADE |
Takeda Pharmaceutical and Janssen Pharmaceutical |
Monotherapy |
PI |
2003 (RRMM) |
2004 (RRMM) |
2006 |
RRMM |
3L+ |
|
Oral melphalan + prednisone |
2008 (NTE-NDMM) |
2008 (NTE-NDMM) |
2011 (NTE-NDMM) |
NDMM |
1L | |||
|
Monotherapy |
2012 (in all approved indication) |
2012 (in all approved indication) |
2012 (in all approved indication) |
- |
- | |||
|
± Dexamethasone |
2014 |
2013 |
- |
RRMM |
2L+ | |||
|
VELCADE as induction therapy (a first therapeutic option) + dexamethasone or thalidomide + dexamethasone |
NA |
2013 (TE-NDMM) |
- |
NDMM |
1L | |||
|
NINLARO |
Takeda Pharmaceutical |
Lenalidomide + dexamethasone |
PI |
2015 (RRMM) |
2016 (RRMM) |
2017 (RRMM) |
RRMM |
2L+ |
|
Monotherapy |
NA |
NA |
2020 (TE-NDMM) |
NDMM |
1L | |||
|
Monotherapy |
NA |
NA |
2021 (NTE-NDMM) |
NDMM |
1L | |||
|
Monotherapy |
- |
- |
2024 (0.5 mg) |
Multiple myeloma with a lower dose formulation of NINLARO |
- | |||
|
2025* |
- |
- |
- |
5L+ | ||||
Multiple Myeloma Pipeline Analysis
Anito-cel (anitocabtagene autoleucel; formerly CART ddBCMA): Arcellx/Gilead (Kite)
Anito-cel is Arcellx's BCMA-specific CAR-modified T-cell therapy utilizing the company’s novel BCMA-targeting binding domain for treating patients with RRMM. As per the company’s pipeline, anito-cel is currently being evaluated in a confirmatory Phase III trial (iMMagine-3) in the 2L+ RRMM setting and in a pivotal Phase II trial (iMMagine-1) in the 4L+ RRMM setting, with regulatory approval being pursued in collaboration with Kite Pharma; furthermore, according to the company’s presentation, initiation of the Phase III iMMagine-4 trial in NDMM is anticipated in the near term, supported by safety data from the GEM-AnitoFIRST study.
In February, the company stated in a news release that the US FDA accepted the BLA for anito-cel for the treatment of adult patients with RRMM in the fourth-line setting. The submission is supported by data from the Phase I study (NCT04155749) and the pivotal Phase II iMMagine-1 trial, with a PDUFA target action date of December 23, 2026.
Etentamig (ABBV-383/TNB-383B): AbbVie (TeneoOne)
ABBV-383 is a monoclonal, B-cell maturation antigen (BCMA)-targeted, IgG4 bispecific antibody.
- Regulatory submission: AbbVie anticipated the regulatory submission of etentamig monotherapy in 3L MM in 2027.
- Date readout: Registrational data readout of Phase III 3L MM (ORR) and of Phase I 2L MM Combinations in 2026 Phase I 1L MM.
|
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
AZD0120 |
AstraZeneca (Gracell Biotechnologies) |
III |
RRMM |
IV infusion |
BCMA x CD19 |
2026 (4L+); 2029 (2L+) |
|
VENCLEXTA (venetoclax, ABT199 and RG7601) |
AbbVie and Roche (Genentech) |
III |
t(11;14)-positive RRMM and RRMM |
Oral |
Bcl-2 inhibitor |
Information is available in the full report |
|
Anito-cel (CART-ddBCMA) |
Arcellx/ Gilead (Kite) |
III |
RRMM, NDMM |
IV Infusion |
Targets BCMA |
Information is available in the full report |
|
Etentamig (ABBV-383) |
AbbVie/ TeneoOne |
III |
RRMM |
IV infusion |
BCMA × CD3 T-cell engaging |
Information is available in the full report |
|
BMS-986393 (CC-95266) |
Bristol-Myers Squibb (Juno Therapeutics, a Subsidiary of Celgene) |
III |
RRMM, NDMM |
Oral |
Cereblon E3 ligase modulator |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Multiple Myeloma Key Players, Market Leaders and Emerging Companies
- AstraZeneca (Gracell Biotechnologies)
- Arcellx/ Gilead (Kite)
- AbbVie
- Bristol-Myers Squibb (Juno Therapeutics, a Subsidiary of Celgene)
- Cartesian Therapeutics
- RAPA Therapeutics
- C4 Therapeutics
- Exicure (GPCR Therapeutics)
- CellCentric, and others
Multiple Myeloma Drug Updates
- In February 2026, AstraZeneca announced that the Phase II (DURGA-3) trial data is projected to be read out in H1 2026, followed by results from the Phase I (DURGA-2) study in H2 2026 and the Phase I/II (DURGA-1) trial in 2027.
- In January 2026, Exicure announced that they presented a poster at the 2026 Transplantation & Cellular Therapy Meetings of the American Society for Transplantation and Cellular Therapy (ASTCT) and Center for International Blood and Marrow Transplant Research (CIBMTR), highlighting results from Exicure’s open-label, multicenter Phase II trial (NCT05561751) evaluating burixafor in patients with multiple myeloma undergoing autologous hematopoietic cell transplant (AHCT).
- In December 2025, CellCentric announced new data from dose optimization cohorts of its Phase II clinical trial of inobrodib in combination with pomalidomide and dexamethasone in heavily pretreated patients with RRMM. Presented during a poster session at the ASH annual meeting, the data further validate a compelling efficacy and safety profile for the all-oral InoPd regimen, including in patients with high-risk disease and prior exposure to TCEs and BCMA-targeted therapies.
Multiple Myeloma Market Outlook
The standard treatment for multiple myeloma typically involves combination regimens, historically consisting of triplet therapy (three drugs). However, more recently, quadruplet regimens incorporating an anti-CD38 monoclonal antibody in addition to a PI, immunomodulatory agent, and corticosteroid are increasingly becoming the SoC for newly diagnosed patients.
At present, the treatment landscape includes a diverse range of therapeutic options, such as PIs, IMiDs, CELMoDs, histone deacetylase inhibitors, monoclonal antibodies, antibody-drug conjugates, chemotherapy, corticosteroids, nuclear export inhibitors, CAR T-cell therapy, and bispecific antibodies.
Among the IMiDs, lenalidomide historically dominated the multiple myeloma market across the seven major markets and has been widely used across multiple lines of therapy, either as monotherapy or in combination regimens. However, following the expiration of key patents and the entry of generic versions beginning in 2022, the market landscape for lenalidomide has become increasingly competitive, although it continues to remain an important backbone therapy in multiple myeloma treatment.
Monoclonal antibodies have become an important component of therapy for multiple myeloma by targeting antigens expressed on malignant plasma cells and enhancing immune-mediated tumor destruction. Currently approved antibodies mainly target CD38 and SLAMF7. Anti-CD38 antibodies include DARZALEX (daratumumab) and SARCLISA (isatuximab-irfc), while EMPLICITI (elotuzumab) targets the SLAMF7 receptor. The introduction of these therapies has significantly expanded treatment options across both newly diagnosed and relapsed or refractory disease settings.
Despite recent therapeutic advances in multiple myeloma, several unmet needs remain in the management of the disease. These include challenges such as treatment resistance, frequent disease relapse, and the limited durability of response in some patients receiving currently available therapies. Additionally, there is a need for further prospective studies to determine optimal treatment sequencing and combination strategies, particularly for patients with advanced or relapsed disease. Continued research and the development of novel therapies are expected to improve long-term disease control and enhance patient outcomes in multiple myeloma.
- In 2025, the total market size of multiple myeloma in first-line transplant eligible therapies was USD 2,945 million in the US. The highest revenue was captured by DVTd accounting for USD 1,295 million in 2025.
- CARVYKTI's launch remains the strongest CAR-T launch to date. About 60-70% of CARVYKTI utilization was occurring in earlier line settings.
- In frontline multiple myeloma clinical studies, DARZALEX was successful and is now considered standard of therapy. DARZALEX has outperformed rivals' expectations in terms of effectiveness and safety, and expected rule the multiple myeloma market.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Multiple Myeloma (2022–2036 Forecast)
The Multiple Myeloma (MM) treatment landscape comprises monoclonal antibodies, small molecules, cellular therapies, and bispecific antibodies, each targeting different mechanisms involved in myeloma cell survival and immune evasion.
Bispecific antibodies: Agents such as TECVAYLI (teclistamab) and ELREXFIO (elranatamab) represent emerging T-cell–redirecting therapies in relapsed or refractory MM. These therapies target B-cell maturation antigen (BCMA) on myeloma cells and CD3 on T cells, enabling immune-mediated destruction of malignant plasma cells. Other investigational bispecific antibodies targeting alternative antigens such as GPRC5D are also being explored to address resistance to BCMA-directed therapies.
Small molecules: Targeted therapies such as NINLARO (ixazomib), an oral proteasome inhibitor, and POMALYST (pomalidomide), an immunomodulatory agent, play a key role in combination regimens for patients with newly diagnosed and relapsed disease. These agents interfere with protein degradation pathways and immune modulation, thereby inhibiting the growth and survival of myeloma cells.
Cellular therapies: Advanced cell-based treatments such as CARVYKTI (ciltacabtagene autoleucel) and ABECMA (idecabtagene vicleucel) are BCMA-directed CAR-T cell therapies that have demonstrated significant clinical activity in heavily pretreated patients with multiple myeloma. These therapies involve engineering a patient’s T cells to recognize and attack malignant plasma cells, offering deep and durable responses in relapsed or refractory settings.
Multiple Myeloma Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the multiple myeloma drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Among the first-line transplant eligible therapies, Daratumumab (SC) + Bortezomib + Lenalidomide + Dexa (D-RVd), Lenalidomide ± Bortezomib + dex, and DARAZLEX based combinations, are expected to capture the largest market share.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Multiple Myeloma
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
The United States
|
US Reimbursement of Therapies Approved for Multiple Myeloma | |
|
Drug/Therapy |
Access Program |
|
REVLIMID |
|
|
KYPROLIS |
|
|
NINLARO |
|
Multiple Myeloma therapies Price Scenario & Trends
Pricing and analogue assessment of multiple myeloma therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Multiple Myeloma Approved Drugs
ELREXFIO is administered as SC as step-up Dose 1 of 12 mg on Day 1, step-up Dose 2 of 32 mg on Day 4, followed by the first treatment dose of 76 mg on Day 8, and then 76 mg weekly thereafter through Week 24. The estimated annual treatment cost is approximately USD 382,560.
Industry Experts and Physician Views for Multiple Myeloma
To keep up with multiple myeloma market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the multiple myeloma emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in multiple myeloma, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the Multiple Myeloma Research Foundation (MMRF), University of Texas MD Anderson Cancer Center, and Iwate Medical University, etc. were contacted. Their opinion helps understand and validate current and emerging multiple myeloma therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in multiple myeloma.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Although options for the treatment of multiple myeloma have expanded significantly in recent years, the disease remains incurable, and therefore, patients are in need of new treatment options. The approval of talquetamab provides patients with a new treatment approach for relapsed or refractory disease that is a welcome addition to the myeloma community.” |
|
Spain |
“As patients with multiple myeloma become exposed to the three main classes of therapy earlier in treatment and still experience relapsed and/or refractory disease, it is critical that we continue to add innovative treatment options to our arsenal that can potentially provide long-term disease control. The expanded approval of ide-cel represents the key to progress in bringing a personalized therapy that delivers significantly improved, durable outcomes to patients with triple-class exposed RRMM after two prior therapies.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of multiple myeloma, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy. The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed.
In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of multiple myeloma, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the multiple myeloma market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM multiple myeloma market.
Report Insights
- Multiple Myeloma Patient population forecast
- Multiple Myeloma Market Opportunity (Current and forecasted)
- Multiple Myeloma Pipeline Analysis
- Multiple Myeloma Market Size and Trends
Report Key Strengths
- Epidemiology-based (Epi-based) bottom-up forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- Multiple Myeloma market outlook (North America, Europe, Asia-Pacific)
- Patient Burden trends (by geography)
- Multiple Myeloma Treatment addressable Market (TAM)
- Multiple Myeloma Competitive Landscape
- Multiple Myeloma major companies Insights
- Multiple Myeloma price trends and analogue assessment
- Multiple Myeloma therapies and Drug Adoption/Uptake
- Multiple Myeloma therapies Peak Patient Share Analysis
Report Assessment
- Multiple Myeloma Current treatment practices
- Multiple Myeloma Unmet needs
- Multiple Myeloma Clinical development Analysis
- Multiple Myeloma emerging drugs product profiles
- Multiple Myeloma Market attractiveness
- Multiple Myeloma Qualitative analysis (SWOT and conjoint analysis)
FAQs
Market Insights
- What was the multiple myeloma market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of multiple myeloma?
- What are the disease risks, burdens, and unmet needs of multiple myeloma? What will be the growth opportunities across the 7MM concerning the patient population with multiple myeloma?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of multiple myeloma? What are the current guidelines for treating multiple myeloma in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the multiple myeloma market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.