Pulmonary Aterial Hypertension Market Summary
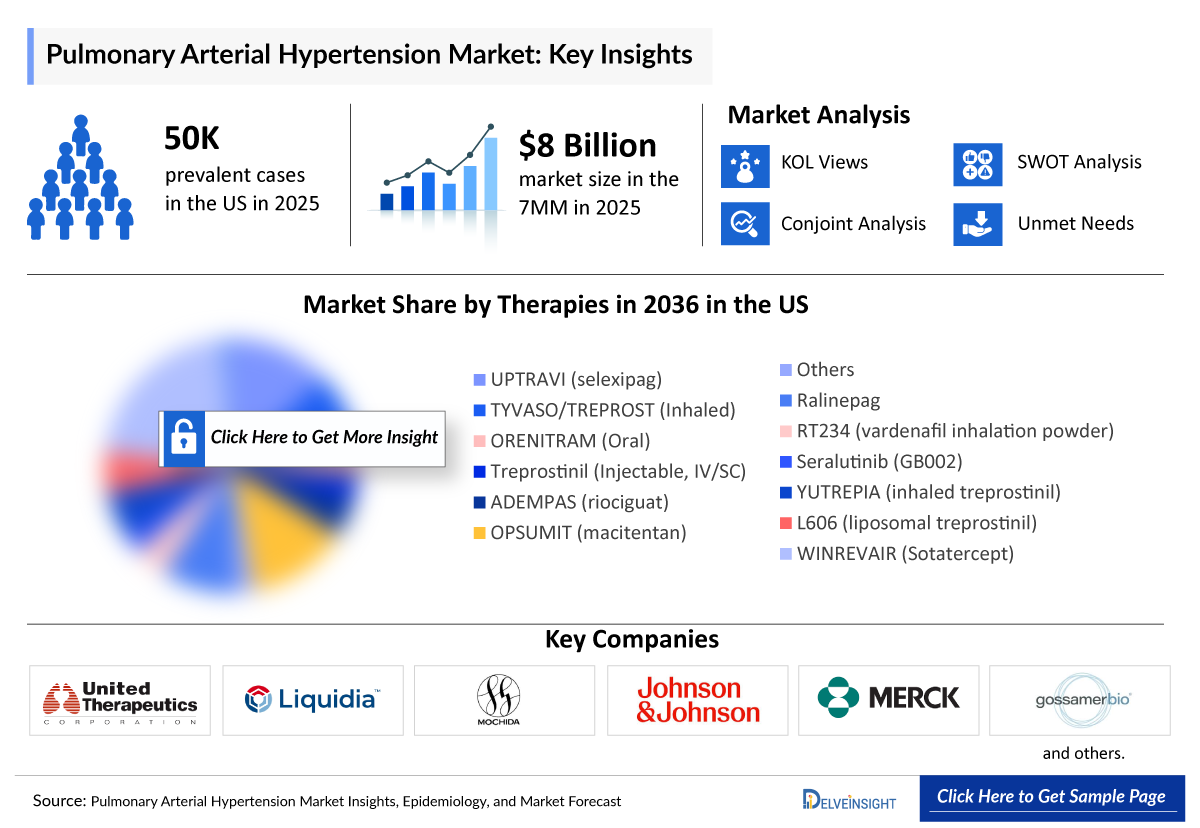
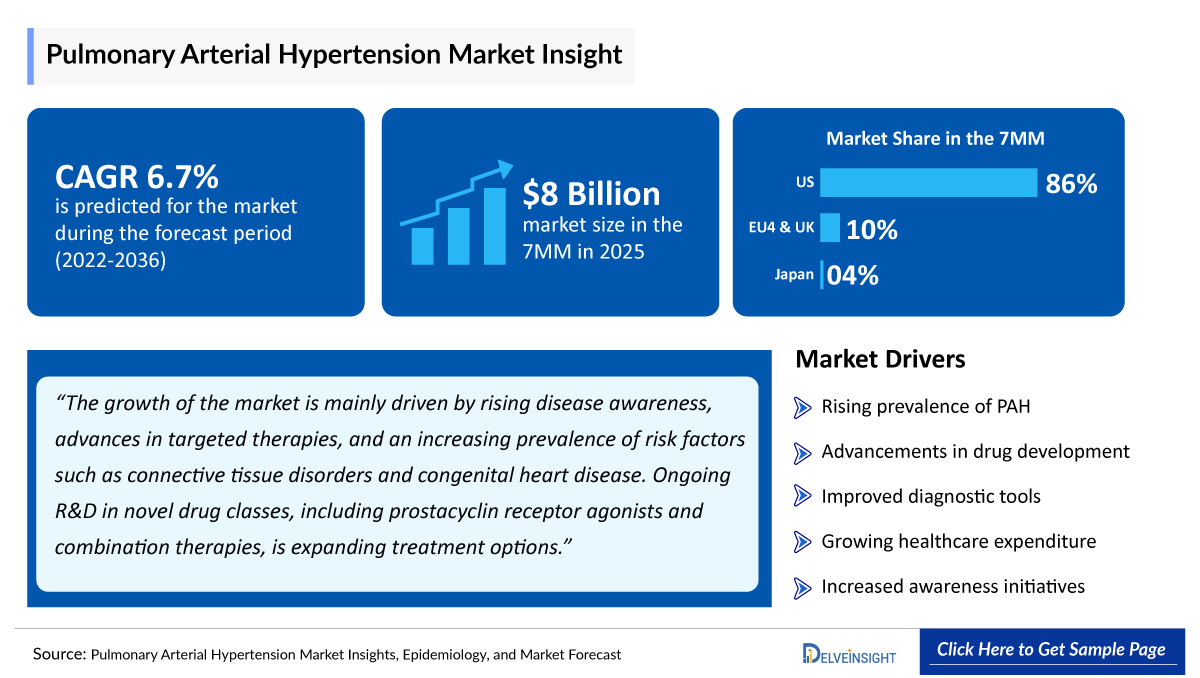
- According to DelveInsight’s analysis, the Pulmonary Arterial Hypertension Market Size was ~USD 8 billion in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- The leading Pulmonary Arterial Hypertension Companies such as Merck, Bristol Myers Squibb, United Therapeutics, Liquidia Technologies, Tenax Therapeutics, Respira Therapeutics, Gossamer Bio, Pharmosa Biopharm, Aerovate Therapeutics, Insmed, Novartis, Cumberland Pharmaceuticals, Sumitomo Pharma (Enzyvant Therapeutics), Cereno Scientific, AstraZeneca, and others.
Pulmonary Arterial Hypertension Market Insights and Trends
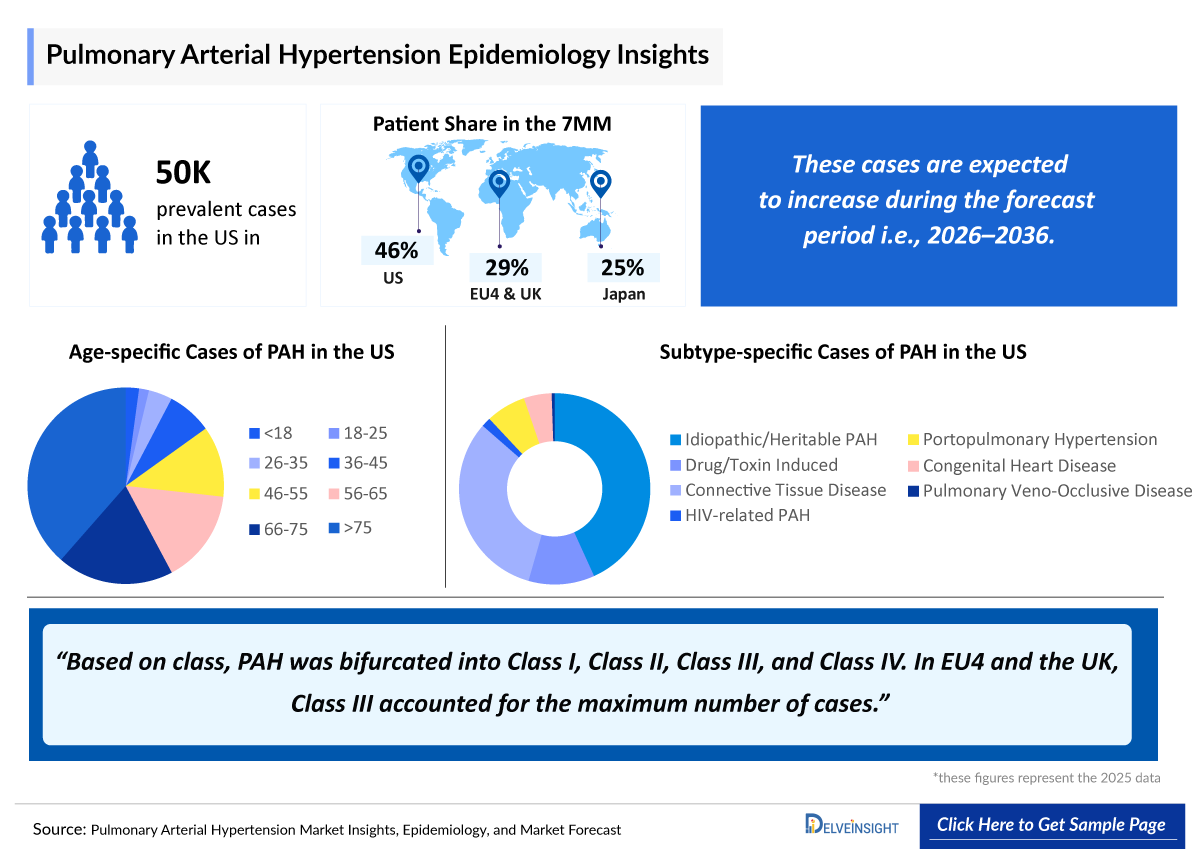
- Pulmonary Arterial Hypertension reported approximately 50,000 prevalent cases and around 44,000 diagnosed prevalent cases in the US in 2025.
- Epidemiological understanding of Pulmonary Arterial Hypertension remains limited due to outdated, regionally variable data, inconsistent diagnostic methodologies, and frequent underdiagnosis driven by non-specific early symptoms.
- The main purpose of the Pulmonary Arterial Hypertension Treatment is to improve patients’ symptoms and slow the rate of clinical deterioration. General measures, supportive therapy, pharmacological treatment, and surgical treatment are used for treating PAH.
- The Pulmonary Arterial Hypertension Treatment Landscape is anchored by multiple approved therapies across functional classes, including selexipag (UPTRAVI), treprostinil (TYVASO), and riociguat (ADEMPAS), among others. Current standard of care is driven by targeted agents, PDE5 inhibitors, soluble guanylate cyclase stimulators, endothelin receptor antagonists (ERAs), and prostacyclin pathway therapies—which collectively modulate the key pathogenic pathways.
- Various Pulmonary Arterial Hypertension Therapies that are expected to enter the market during the forecast period include Ralinepag (United Therapeutics), RT234 (vardenafil inhalation powder) (Respira Therapeutics), seralutinib (GB002) (Gossamer Bio), and L606 (liposomal treprostinil) (Pharmosa Biopharm/Liquidia).
- Pulmonary Arterial Hypertension is a rare, highly complex, and progressive disorder that is incurable and often leads to premature death. Early diagnosis is required for the best outcomes; however, the clinical presentation of PAH is ambiguous and usually overlaps with numerous other illnesses, often leading to a delay in diagnosis or misdiagnosis. In the past decades, an increased understanding of the pathobiology of PAH has led to changes in its definition, but a lack of awareness adds to the difficulty.
Pulmonary Arterial Hypertension Market size and forecast
- 2025 PAH Market Size in the 7MM: ~USD 8 Billion
- PAH Growth Rate (2022-2036): 6.7% CAGR (Compound Annual Growth Rate)
Request for Unlocking the Sample Page of the "Pulmonary Arterial Hypertension Treatment Market"
Factors Contributing to the Growth of the Pulmonary Arterial Hypertension Market by DelveInsight
-
Rising Prevalence of PAH
Increasing incidence of pulmonary arterial hypertension due to genetic factors, connective tissue diseases, and lifestyle-related conditions is driving demand for effective therapies.
-
Improved Diagnostic Techniques
Advancements in screening tools, imaging technologies, and early diagnosis are enabling timely treatment initiation, boosting market growth.
-
Growing Awareness and Disease Education
Increased awareness among healthcare professionals and patients is leading to higher diagnosis rates and improved disease management.
-
Advancements in Targeted Therapies
Development of novel drug classes such as endothelin receptor antagonists (ERAs), phosphodiesterase-5 inhibitors (PDE-5), and prostacyclin analogs is enhancing treatment outcomes.
-
Robust Pipeline and Ongoing Clinical Trials
Strong presence of emerging therapies and continuous R&D activities by pharmaceutical companies is expanding treatment options.
-
Favorable Regulatory Support
Orphan drug designations, fast-track approvals, and supportive regulatory frameworks are accelerating drug development and commercialization.
-
Increasing Healthcare Expenditure
Rising healthcare investments globally are improving access to advanced therapies and specialized care.
-
Expansion of Specialty Care Centers
The growth of specialized pulmonary and cardiology centers is improving patient access to advanced PAH treatments.
-
Improved Survival Rates and Long-term Management
Better treatment regimens are increasing patient survival, thereby contributing to sustained market demand.
DelveInsight’s “Pulmonary Arterial Hypertension Market Insights, Epidemiology, and Market Forecast – 2036” report delivers an in-depth understanding of PAH, historical and forecasted epidemiology, as well as the Pulmonary Arterial Hypertension Market Trends in the United States, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan.
The Pulmonary Arterial Hypertension Treatment Market Report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates Pulmonary Arterial Hypertension Patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth Pulmonary Arterial Hypertension Market Size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key Pulmonary Arterial Hypertension Unmet Medical Needs and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Pulmonary Arterial Hypertension Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Pulmonary Arterial Hypertension Market CAGR |
6.7% (2022-2036) |
|
Pulmonary Arterial Hypertension Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Pulmonary Arterial Hypertension Companies |
|
|
Pulmonary Arterial Hypertension Therapies |
|
|
Pulmonary Arterial Hypertension Market |
Segmented By
|
|
Analysis |
|
Pulmonary Arterial Hypertension Understanding
PAH Overview and Diagnosis
Pulmonary Arterial Hypertension, characterized by increased pulmonary vascular resistance and arterial pressure, affects an estimated 15–60 per million of the population and is more commonly diagnosed in women. PAH causes a range of nonspecific symptoms (including breathlessness, fatigue, chest pain, and weakness) and is associated with significant morbidity and mortality triggered by the debilitating progressive nature of the disease, which eventually leads to right heart failure and death. The effect of disease symptoms on the patient’s physical mobility and emotional state adversely affects health-related quality of life (HRQoL).
PAH is characterized by similar symptoms that appear during heart and lung conditions. Several techniques and blood tests, such as chest X-rays, blood tests, cardiac catheterization, electrocardiograms, and echocardiograms, are used for diagnosing PAH.
Further details related to country-based variations are provided in the report…
Current Pulmonary Arterial Hypertension Treatment Landscape
The four drug classes commonly used to treat PAH—PDE5 inhibitors, sGC stimulators, Endothelin Receptor Antagonists (ERAs), and prostacyclin analogs and agonists—target three major signaling pathways: prostacyclin, endothelin, and nitric oxide.
Macitentan is an endothelin receptor antagonist used to treat PAH, improving exercise capacity and reducing symptoms by lowering pulmonary vascular resistance. While the optimal initiation and adjustment of macitentan therapy remain unclear, it is considered a key option for managing PAH, especially for advanced cases. Prostacyclin medications are the oldest group used to treat PAH; these increase cAMP concentrations to produce pulmonary vasodilation. Epoprostenol was the first drug introduced specifically for treating PAH and was approved by the US FDA in 1995. However, it was limited in its versatility by its restriction to IV use in the outpatient setting, requiring central venous access. There are several different prostacyclin derivatives with various modes of delivery that are improved; these include inhaled iloprost and treprostinil, SC or IV treprostinil, oral treprostinil diolamine, and an oral prostacyclin agonist selexipag.
Pulmonary Arterial Hypertension Unmet Needs
The section “unmet needs of PAH” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Scarce epidemiology data
- Biomarker based diagnosis
- Curative targeted therapies
- Early diagnosis and awareness
- Comprehensive treatment algorithm and others…..
Note: Comprehensive unmet needs insights in PAH and their strategic implications are provided in the full report.
Pulmonary Arterial Hypertension Epidemiology
Key Findings from PAH Epidemiological Analysis and Forecast
- In 2025, the US accounted for the highest diagnosed prevalent cases of PAH with approximately 40,000 cases, which are expected to increase by 2036.
- Idiopathic/heritable PAH had the highest number of subtype-specific cases in the US in 2025 with nearly (19,000 cases), which is predicted to rise by 2036.
- In 2025, the Germany reported the highest number of diagnosed prevalent cases of PAH among EU4 and the UK, with approximately 7,000 cases.
- In 2025, Japan reported nearly 19,000 diagnosed prevalent cases of PAH in females and nearly 5,000 cases in males, with numbers projected to rise by 2036.
Pulmonary Arterial Hypertension Epidemiology Segmentation in the 7MM
- Total Pulmonary Arterial Hypertension Prevalent Cases
- Total Pulmonary Arterial Hypertension Diagnosed Prevalent Cases
- Pulmonary Arterial Hypertension Age-specific Cases
- Pulmonary Arterial Hypertension Class-specific Cases
- Pulmonary Arterial Hypertension Gender-specific Cases
- Pulmonary Arterial Hypertension Subtype-specific Cases
- Total Pulmonary Arterial Hypertension Treated Cases
Pulmonary Arterial Hypertension Drug Analysis & Competitive Landscape
The Pulmonary Arterial Hypertension Drug Chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I-III Pulmonary Arterial Hypertension Clinical Trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Pulmonary Arterial Hypertension Treatment Landscape, supporting market assessment, competitive analysis, and growth forecasting for the Pulmonary Arterial Hypertension Therapeutics Market.
Approved Therapies for Pulmonary Arterial Hypertension
-
Inhaled treprostinil (YUTREPIA): Liquidia Technologies
Treprostinil (YUTREPIA), developed by Liquidia Technologies, is an inhaled dry powder formulation of treprostinil delivered through a convenient, palm-sized device designed using Liquidia’s PRINT technology to enhance deep-lung delivery to treat PAH.
-
- In May 2025, FDA authorized YUTREPIA as a new therapeutic option for adults with PAH (WHO Group 1) and PH-ILD (WHO Group 3).
PAH Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Treprostinil (YUTREPIA) |
Liquidia Technologies |
PAH FC II─IV |
Small molecule |
Prostacyclin receptor agonist |
Inhalation |
US: 2025 |
|
TYVASO/ TYVASO DPI/ TREPROST (inhalation) |
United Therapeutics/ Mochida Pharmaceutical |
PAH (WHO Group I) with NYHA FC III |
Small molecule |
Prostacyclin analog (vasodilator and platelet aggregation inhibitors) |
Inhalation |
US:2009(inhalation solution) US:2022(inhalation powder) JP:2022 (inhalation solution) |
|
Macitentan and tadalafil (OPSYNVI/YUVACI) |
Johnson & Johnson |
PAH (WHO Group I) with FC II-III |
Small molecule |
Endothelin receptor antagonist (ERA), phosphodiesterase 5 (PDE5) inhibitor |
Oral |
US:2024 EU:2024 JP:2024 |
Note: Detailed marketed therapies assessment will be provided in the final report.
Pulmonary Arterial Hypertension Pipeline Analysis
-
Ralinepag: United Therapeutics
Ralinepag is a novel, oral, selective, and potent prostacyclin receptor agonist being developed by United Therapeutics for the treatment of PAH. In vitro studies indicate that ralinepag has high binding affinity and selectivity at the human prostacyclin (IP) receptor. In phase II studies, ralinepag demonstrated a potential for a once-a-day dosing profile and potentially enhanced affinity compared to selexipag.
- In March 2026, United Therapeutics announced that ralinepag had achieved a 55% reduction in the risk of clinical worsening in a pivotal PAH study, delivering exceptional and highly statistically significant efficacy.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Ralinepag |
United Therapeutics |
III |
PAH FC II and III |
Oral |
Selective prostacyclin receptor agonist |
Information is available in the full report |
|
Seralutinib (GB002) |
Gossamer Bio |
III |
PAH FC II or III |
Inhalation |
PDGFR, CSF1R, and c-KIT kinase inhibitor |
Information is available in the full report |
|
L606 (liposomal treprostinil) |
Pharmosa Biopharm/ Liquidia |
III |
PAH FC II, III, or IV |
Inhalation |
DP1, EP2, and IP receptor agonists |
2027 |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Note: Detailed emerging therapies assessment will be provided in the final report.
Pulmonary Arterial Hypertension Companies
- United Therapeutics
- Mochida Pharmaceutical
- Johnson & Johnson
- Merck
- Gossamer Bio
- Pharmosa Biopharm/ Liquidia
- Respira Therapeutics and others.
Pulmonary Arterial Hypertension Drug Updates
- United Therapeutics intends to submit a New Drug Application (NDA) for ralinepag to the US Food and Drug Administration (FDA) by the second half of 2026.
- In February 2026, Gossamer Bio announced topline Phase III results for seralutinib, an investigational inhaled therapy for adults with PAH.
- In May 2025, the FDA officially approved Liquidia Corporation’s treprostinil inhalation powder (YUTREPIA) for the treatment of PAH and pulmonary hypertension associated with interstitial lung disease (PH-ILD) in adults.
Pulmonary Arterial Hypertension Market Outlook
Current therapies primarily work by dilating pulmonary blood vessels to reduce resistance and improve heart function, with treatment strategies guided by risk assessment based on clinical, functional, and hemodynamic parameters to achieve a low-risk status and improve outcomes such as survival, quality of life, and symptom control. The main drug classes used include PDE5 inhibitors, sGC stimulators, endothelin receptor antagonists, and prostacyclin analogs/agonists, which target the nitric oxide, endothelin, and prostacyclin pathways.
There are several Pulmonary Arterial Hypertension Marketed Drugs, each with distinct mechanisms. TYVASO/TYVASO DPI/TREPROSTINIL, ORENITRAM, and OPSYNVI/YUVANCI target prostacyclin pathways. Sotatercept (WINREVAIR) modulates signaling to improve vascular function.
The emerging therapies developed to treat Pulmonary Arterial Hypertension Patients include late-stage and mid-stage candidates. As per DelveInsight’s estimates, the potential Pulmonary Arterial Hypertension drugs that can mark a significant change in the forecast period include seralutinib (GB-002), treprostinil liposomal (L-606), Ralinepag and others.
- The Pulmonary Arterial Hypertension Market Size of EU4 and the UK was estimated to be approximately USD 400 million in 2025.
- Among EU4 and the UK, Germany accounted for the highest Pulmonary Arterial Hypertension Market Share with approximately USD 100 million in 2025, followed by France and the UK with approximately USD 80 million in the respective year in 2025.
- In 2025, the Pulmonary Arterial Hypertension Market Size was approximately USD 300 million in Japan which is anticipated to increase during the forecast period (2026–2036).
Pulmonary Arterial Hypertension Drug Class/Insights into Leading Emerging Therapies (2022–2036 Forecast)
Small Molecule: Small-molecule therapies are playing an increasingly pivotal role in the management of Pulmonary Arterial Hypertension, driven by their ability to precisely target key molecular pathways involved in disease progression. Their low molecular weight enables efficient delivery, particularly via inhalation, ensuring direct drug deposition in the pulmonary vasculature while minimizing systemic exposure. This targeted approach allows modulation of critical mechanisms such as vasoconstriction, vascular proliferation, and remodeling. Agents like seralutinib (GB002) exemplify this strategy by inhibiting PDGF receptor signaling to address vascular remodeling. Similarly, inhaled formulations of treprostinil, delivered through portable dry powder inhalers, enhance convenience and localized efficacy.
Recombinant fusion proteins: Recombinant fusion proteins represent an advanced class of biologic therapies in Pulmonary Arterial Hypertension, engineered by combining functional domains of different proteins to enhance therapeutic activity. These agents target key signaling pathways involved in vascular remodeling and abnormal cell proliferation, offering a disease-modifying approach beyond conventional vasodilators. A leading example is sotatercept (WINREVAIR), an activin signaling inhibitor that restores the balance between pro- and anti-proliferative pathways in the pulmonary vasculature.
Pulmonary Arterial Hypertension Drug Uptake
This section focuses on the uptake rate of potential Pulmonary Arterial Hypertension drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the PAH drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The current mainstay therapies to treat PAH act to dilate the pulmonary vasculature, decreasing pulmonary vascular resistance and secondarily improving right ventricular function, thereby improving functional capacity. The overall treatment goal is to improve survival, quality of life, exercise capacity, symptom burden, and clinical worsening, with risk stratification tools increasingly used to guide therapy and improve each of these elements.
Vardenafil (RT234), developed by Respira Therapeutics, is a first-in-class inhaled, as-needed (PRN) therapy for PAH that delivers vardenafil via a novel Axial Oscillating Sphere (AOS) dry powder inhaler, enabling rapid pulmonary vasodilation and on-demand relief of exertional breathlessness while improving exercise tolerance. Positioned as a complementary option to chronic background therapies, RT234 is expected to see moderate adoption, with an estimated ~7 years to peak uptake.
Detailed insights of emerging therapies' drug uptake is included in the report
Pulmonary Arterial Hypertension Market Access and Reimbursement of Therapies
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Pulmonary Arterial Hypertension therapies Price Scenario & Trends
Pricing and analogue assessment of PAH therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
- Pricing of PAH Approved Drugs
Selexipag (UPTRAVI) is an oral, selective prostacyclin IP receptor agonist approved for the treatment of PAH, more than three-quarters (76%) of PAH patients treated with UPTRAVI (selexipag) either maintained (56%) or reduced (20%) their 1-year mortality risk score. It is associated with a relatively high annual treatment cost in the United States, estimated at approximately USD 260,000.
Pulmonary Arterial Hypertension Industry Experts and Physician Views
To keep up with PAH market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the PAH emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in PAH, including MD, PhD, Instructor, Pos tdoctoral Researcher, Professor, Researcher, and others.
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Nearly half of the PAH cases are idiopathic, hereditary, or anorexigen-induced PAH. Women between the ages of 30 and 60 are typically affected by PAH. However, it may occur in men and is frequently linked to worse clinical consequences.” |
|
Germany |
“In PAH, risk assessment is critical; patients with a low or intermediate risk of 1-year mortality should normally start with a double oral combination medication, which is more effective than starting with monotherapy, but high-risk patients should start with combination therapy including an IV prostacyclin analog. To enhance prognosis, patients who are not low-risk at follow-up should have their therapy escalated.” |
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as the University of Pennsylvania Hospital, University of Chicago, University of Pittsburgh, Medizinische Hochschule Hannover etc. were contacted. Their opinion helps understand and validate current and emerging PAH therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in PAH.
Pulmonary Arterial Hypertension Qualitative Analysis: SWOT and Attribute Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and attribute analysis.
In the SWOT analysis of PAH, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provide attribute analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Pulmonary Arterial Hypertension Market Report
- The Pulmonary Arterial Hypertension Therapeutics Market Report covers a segment of key events, an executive summary, and a descriptive overview, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the Pulmonary Arterial Hypertension epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current Pulmonary Arterial Hypertension Treatment landscape.
- A detailed review of the PAH market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM PAH market.
Pulmonary Arterial Hypertension Market Report Insights
- PAH Patient Population Forecast
- PAH Therapeutics Market Size
- PAH Pipeline Analysis
- PAH Market Size and Trends
- PAH Market Opportunity (Current and Forecasted)
Pulmonary Arterial Hypertension Market Report Key Strengths
- Epidemiology‑based (Epi‑based) bottom‑up forecasting
- Artificial Intelligence (AI) - enabled market research report
- 11-year forecast
- PAH Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- PAH Treatment Addressable Market (TAM)
- PAH Competitive Landscape
- PAH Major Companies Insights
- PAH Price Trends and Analogue Assessment
- PAH Therapies Drug Adoption/Uptake
- PAH Therapies Peak Patient Share analysis
Pulmonary Arterial Hypertension Market Report Assessment
- PAH Current Treatment Practices
- PAH Unmet Needs
- PAH Clinical Development Analysis
- PAH Emerging Drugs Product Profiles
- PAH Market Attractiveness
- PAH Qualitative Analysis (SWOT and attribute analysis)
Key Questions Answered in the Pulmonary Arterial Hypertension Market Report
Pulmonary Arterial Hypertension Market Insights
- What was the PAH market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of PAH?
- What are the disease risks, burdens, and unmet needs of PAH? What will be the growth opportunities across the 7MM concerning the patient population with PAH?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of PAH? What are the current guidelines for treating PAH in the US, Europe, and Japan?
Reasons to Buy the Pulmonary Arterial Hypertension Market Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the PAH market.
- Bottom-up forecasting builds from the affected population to product forecasts, delivering a robust, data-driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) - enabled report summarize and simplify complex datasets with in the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data-driven decisions.
Stay updated with us for Recent Articles
- WINREVAIR Approval for Pulmonary Arterial Hypertension Treatment: Is It A Game-Changer for Merck?
- New Clinical Developments in the Pulmonary Arterial Hypertension Treatment Domain
- Pulmonary Arterial Hypertension Therapies
- Outbreak of Companies Challenging New Pathways for Treatment of Pulmonary Arterial Hypertension
- Pulmonary Arterial Hypertension Market Infographics
- Pulmonary Arterial Hypertension Newsletter
- New DelveInsight Blogs