prostate cancer market
DelveInsight's "Prostate cancer Market Insights, Epidemiology, and Market Forecast-2030" report delivers an in-depth understanding of the Prostate cancer, historical and forecasted epidemiology as well as the Prostate cancer market trends in the United States, EU5 (Germany, Spain, Italy, France, and United Kingdom) and Japan.
The Prostate cancer market report provides current treatment practices, emerging drugs, Prostate cancer market share of the individual therapies, current and forecasted Prostate cancer market Size from 2017 to 2030 segmented by seven major markets. The Report also covers current Prostate cancer treatment practice/algorithm, market drivers, market barriers and unmet medical needs to curate best of the opportunities and assesses the underlying potential of the market.
Geography Covered
- The United States
- EU5 (Germany, France, Italy, Spain, and the United Kingdom)
- Japan
Study Period: 2017-2030
Prostate cancer Disease Understanding and Treatment Algorithm
Prostate cancer a type of malignancy that occurs in the prostate gland. The prostate is a small walnut-shaped gland in men that produces the seminal fluid that nourishes and transports sperm. It is located next to the bladder and can be examined by getting a digital rectal exam.
Prostate cancer is one of the most common types of cancer found in men. This cancer usually grows slowly and is confined to the prostate gland initially, where it may not cause serious harm. According to the Cancer Treatment Centers of America (CTCA), more than 99% of prostate cancers are adenocarcinomas, which develop in the gland cells. It is common in men of 50–64 years and over age 65; however, it can occur in men younger than 50 years. Symptoms of adenocarcinoma of the prostate include blood in the semen, frequent urge to urinate, and painful urination and ejaculation. The symptoms of prostate cancer do not usually appear until the prostate is large enough to affect the tube that carries urine from the bladder out of the penis. Clinical Stage and treatment pattern are key factors in Prostate Cancer.
“In clinical-stage-wise cases of Prostate Cancer, most of the cases were of the localized stage, which accounted for approximately 60% of the total diagnosed prevalent cases in 2017. On the other hand, metastatic cases accounted for only 7% of the total diagnosed prevalent cases.”
Diagnosis
Many patients undergo regular prostate cancer screening before symptoms appear. Screening may involve one or more of the following tests: prostate-specific antigen (PSA), digital rectal exam (DRE), prostate ultrasound, prostate MRI, prostate Mp-MRI. Screening is a type of testing to find cancer in people before they have symptoms. It is not clear, however, if the benefits of prostate cancer screening outweigh the risks for most men. Still, after discussing the pros and cons of screening with their doctors, some men might reasonably choose to be screened. If the result of one of these tests is abnormal, patients will probably need a prostate biopsy to know for sure the occurrence of cancer.
Treatment
The prostate cancer experts develop a comprehensive treatment plan for each patient specifically. Treatment strategies of prostate cancers depend on the stage and progression of cancer. However, prostate cancer often grows slowly, and active surveillance may be the preferred treatment option for some men, with the oncologist closely monitoring the disease with tests and holding off on treatment until a later date. Besides, other treatment options for prostate cancers include chemotherapy, hormone therapy, immunotherapy, radiation therapy, and surgery.
Patients who have never received, i.e., are sensitive to ADT known as hormone-sensitive prostate cancer (HSPC) or castrate-sensitive prostate cancer (CSPC). Further down the line, many prostate cancers patients ultimately fail to respond to ADT and are termed castrate-resistant prostate cancer (CRPC).
“It was accessed that around 78% of biochemical recurrence/ progressive cases are non-metastatic castrate sensitive prostate cancer (nmCSPC/ nmHSPC). Contrary to this near to 22% of biochemical recurrence/ progressive cases progresses to be non-metastatic castrate resistant prostate cance (nmCRPC). Of the metastatic prostate cancer cases, approximately 55% have mCSPC/ mHSPC, and round to 45% progresses to be mCRPC.”
Prostate cancer Recent Developments
- In March 2025, Quibim announced that its QP-Prostate® CAD solution for prostate cancer lesion detection received FDA 510(k) clearance. This milestone advances Quibim’s mission to improve prostate cancer detection and treatment.
- In September 2024, Ipsen announced that the Phase III CONTACT-02 trial for Cabometyx® and atezolizumab in mCRPC showed a non-significant improvement in overall survival but met the progression-free survival (PFS) endpoint.
- In September 2024, Foundation Medicine received FDA approval for FoundationOne®CDx and Liquid CDx as companion diagnostics for Lynparza® in BRCA-mutated mCRPC.
- In July 2024, the ARANOTE trial showed that NUBEQA® plus ADT significantly improved radiological PFS in metastatic hormone-sensitive castrate-resistant prostate cancer (mHSPC).
- In July 2024, the FDA granted fast-track designation to SYNC-T SV-102 for metastatic castrate-resistant prostate cancer (mCRPC).
- In June 2024, Kangpu Biopharmaceuticals received FDA approval for a Phase II/III trial of KPG-121 with Abiraterone for mCRPC.
- In May 2024, Fusion Pharmaceuticals began the Phase 2 AlphaBreak trial of FPI-2265 in mCRPC patients.
- In April 2024, Astellas Pharma received European approval for XTANDI™ in high-risk biochemical recurrent non-metastatic hormone-sensitive prostate cancer (nmHSPC).
- In April 2024, FibroGen announced positive Phase 1 trial data for FG-3246 in mCRPC.
- On November 16, 2023, the FDA approved enzalutamide (Xtandi) for non-metastatic castration-sensitive prostate cancer (nmCSPC) with high-risk biochemical recurrence.
Prostate cancer Epidemiology
The Prostate cancer epidemiology division provide insights about historical and current Prostate cancerpatient pool and forecasted trend for every seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the DelveInsight report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings
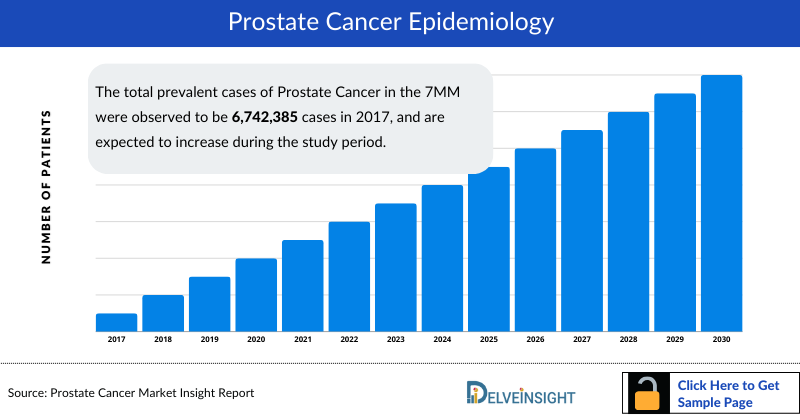
- The total prevalent cases of Prostate Cancer in the 7MM were observed to be 6,742,385 cases in 2017, and are expected to increase during the study period.
The disease epidemiology covered in the report provides historical as well as forecasted Prostate Cancer epidemiology [segmented as Total Prevalent Cases of Prostate Cancer, Total Diagnosed Cases of Prostate Cancer, Age-specific Cases of Prostate Cancer, Total Diagnosed cases of Prostate Cancer patients by Clinical Stages, Total Non-metastatic and Metastatic Cases of CSPC/HSPC and CRPC, Total Treated cases of Non-metastatic CSPC/HSPC and CRPC, Total Treated Cases of metastatic HSPC/CSPC, and CRPC] scenario of Prostate Cancer in the 7MM covering United States, EU5 countries (Germany, France, Italy , Spain, and United Kingdom), and Japan from 2017 to 2030.
Country Wise- Prostate cancer Epidemiology
- Estimates show the highest diagnosed population of Prostate Cancer is in the United States. in EU-5 countries, the highest number of diagnosed prevalent cases of prostate cancer was found in Germany with 360,916 cases in 2017, followed by France and Italy.
- Our estimation suggested that the total treated patient pool of mCSPC/HSPC was 75,600 in 2017, will likely increase throughout the study period, besides nearly 73,452 cases of mCRPC treated in first-line.
- According to the DelveInsight, in 2017, total treated pool nmCRPC progressed from first-line to the second-line was found to be 160,287 in the 7MM. On the other hand, the total treated patient pool of nmCSPC/HSPC was 668,577 cases in the 7MM (2017).
Prostate cancer Drug Chapters
Drug chapter segment of the Prostate cancer report encloses the detailed analysis of Prostate cancer marketed drugs and late stage (Phase-III and Phase-II) pipeline drugs. It also helps to understand the Prostate cancerclinical trial details, expressive pharmacological action, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug and the latest news and press releases.
Marketed Drugs
Xtandi (Enzalutamide): Astellas Pharma/Pfizer
It is an orally bioavailable, organic, non-steroidal small molecule targeting the androgen receptor (AR) with potential antineoplastic activity. It is indicated for the treatment of patients with castration-resistant prostate cancer (CRPC), and metastatic castration-sensitive prostate cancer (mCSPC). Xtandi approved initially in August 2012. It was approved by the US FDA for the treatment of patients with mCRPC who have previously received docetaxel. In December 2019, Pfizer and Astellas Pharma announced that the US FDA has approved a supplemental New Drug Application (sNDA) for Xtandi for the treatment of patients with mCSPC.
Erleada (apalutamide): Janssen Pharmaceutical
Janssen Pharmaceutical’s product Erleada (apalutamide) is a next-generation oral androgen receptor (AR) inhibitor that blocks the androgen signaling pathway in prostate cancer cells. It is indicated for the treatment of patients with non-metastatic castration-resistant prostate cancer (nmCRPC) and for the treatment of patients with metastatic castration-sensitive prostate cancer (mCSPC). It is taken orally, once daily, with or without food. Erleada inhibits the growth of cancer cells in three ways: by preventing the binding of androgen to the AR; by stopping the AR from entering the cancer cells; and by preventing the AR from binding to the DNA of the cancer cell. In January 2020, the European Commission has approved apalutamide (Erleada) for use in combination with androgen deprivation therapy (ADT) for the treatment of adult men with metastatic hormone-sensitive prostate cancer. The EC approval is based on data from the phase III TITAN study, which assessed the addition of apalutamide to ADT in a broad range of patients with mHSPC, regardless of disease volume, prior treatment with docetaxel or staging at initial diagnosis.
Nubeqa (Darolutamide/ODM-201): Bayer HealthCare/Orion Corporation
Bayer HealthCare/Orion Corporation’s product Nubeqa (darolutamide) is an androgen receptor inhibitor (ARi) with a distinct chemical structure that competitively inhibits androgen binding, AR nuclear translocation, and AR-mediated transcription. Nubeqa is approved for the treatment of patients with nmCRPC. In January 2020, the Japanese Ministry of Health, Labor and Welfare (MHLW) granted marketing authorization to Nubeqa for the treatment of men with nmCRPC. Nubeqa (darolutamide) is also being investigated for mHSPC/mCSPC.In ARASENS trial, approximately 1,300 subjects enrolled with mHSPC. The patients will be randomized (1:1 ratio) to receive 600 mg (2 × 300 mg tablets) of Darolutamide (ODM-201)/placebo twice daily with food, equivalent to a total daily dose of 1,200 mg, in addition to standard ADT and docetaxel. Subjects will be stratified at randomization for the extent of disease and for Alkaline Phosphatase levels.
Products detail in the report…
Prostate cancer Emerging Drugs
Lynparza (olaparib): AstraZeneca/Merck Sharp & Dohme
Lynparza (olaparib) is first and best-in-class oral poly ADP-ribose polymerase (PARP) inhibitor, and the ¬first targeted treatment to block DDR in tumours harbouring a deficiency in homologous recombination repair (HRR), such as mutations in BRCA1 and/or BRCA2. AstraZeneca have global strategic oncology collaboration with Merck to co-develop and co-commercialize Lynparza. Recently in January 2020, the US FDA accepted the supplemental New Drug Application for Lynparza and granted Priority Review in the US for patients with metastatic castration-resistant prostate cancer and deleterious or suspected deleterious germline or somatic homologous recombination repair (HRR) gene mutations, which have progressed following prior treatment with a new hormonal agent. A Prescription Drug User Fee Act (PDUFA) date is set for the second quarter of 2020. The priority review is based on the results from the Phase III PROfound trial, which were presented during the Presidential Symposium at the 2019 European Society of Medical Oncology congress.
Talzoparib: Pfizer/Astellas Pharma
Talzoparib (Pfizer/Astellas Pharma) is an orally PARP inhibitor with potential antineoplastic activity, currently under evaluation in Phase III clinical stage. It selectively binds to PARP and prevents PARP-mediated DNA repair of single strand DNA breaks via the base-excision repair pathway. A phase III study (NCT03395197; TALAPRO-2) was initiated to compare rPFS in men with mCRPC treated with Talazoparib plus enzalutamide vs. enzalutamide after confirmation of the starting dose of Talazoparib in combination with enzalutamide (Xtandi).
Rubraca (Rucaparib): Clovis Oncology
Clovis Oncology is investigating one of their lead candidates named Rubraca (Rucaparib) for various cancer indications. It is an oral, small-molecule inhibitor of PARP1, PARP2 and PARP3 being developed in Prostate Cancer, and Ovarian Cancer as well as several additional solid tumor indications. It is worth mentioning that, the US Food and Drug Administration has accepted the company’s supplemental New Drug Application (sNDA) for Rubraca and granted priority review status to the application with a Prescription Drug User Fee Act (PDUFA) date of May 15, 2020.
Keytruda: Merck Sharp & Dohme
Merck Sharp & Dohme’s immunotherapy Keytruda, also known as pembrolizumab, is a PD-1 blocking antibody. It is mainly used for cancers that are advanced, have spread to other parts of the body or are not responding to other treatments. This is currently being investigated in Phase III clinical trials in combination with Enzalutamide for the treatment of patients affected by mHSPC. Moreover, several other trials are also ongoing for the treatment of patients affected with mCRPC.
Ipatasertib (RG7440): Hoffmann-La Roche
Hoffmann-La Roche investigating Ipatasertib (RG7440) for Prostate Cancer. It is a small molecule Pan-Akt inhibitor which is under investigation by Roche in Phase III clinical trial. The PI3K/Akt/mTOR pathway regulates cell growth and survival.
ProstAtak: Advantagene
Advantagene is developing ProstAtak for the treatment of patients suffering from prostate cancer. ProstAtak jump-starts the body’s own immune system to detect and destroy recurring cancer cells. The treatment is done together with standard radiation therapy. Last year, Candel Therapeutics (Advantagene) announced that it has completed the enrollment of the "ULYSSES" trial, a Phase II study of ProstAtak for prostate cancer patients choosing Active Surveillance (PrTK04- NCT02768363). However, enrollment is still open for prostate cancer patients choosing radiation therapy as their primary treatment (PrTK03- NCT01436968).
177Lu-PSMA-617: Novartis
177Lu-PSMA-617 is an investigational radioligand therapy (RLT) in development for mCRPC by Novartis. Currently this drug is in a Phase III trial (VISION) for the treatment of patients with progressive PSMA-positive mCRPC.
Products detail in the report…
Prostate cancer Market Outlook
The Prostate cancer market outlook of the report helps to build the detailed comprehension of the historic, current, and forecasted Prostate cancer market trends by analyzing the impact of current therapies on the market, unmet needs, drivers and barriers and demand of better technology.
This segment gives a thorough detail of Prostate cancer market trend of each marketed drug and late-stage pipeline therapy by evaluating their impact based on annual cost of therapy, inclusion and exclusion criteria's, mechanism of action, compliance rate, growing need of the market, increasing patient pool, covered patient segment, expected launch year, competition with other therapies, brand value, their impact on the market and view of the key opinion leaders. The calculated market data are presented with relevant tables and graphs to give a clear view of the market at first sight.
According to DelveInsight, Prostate cancer market in 7MM is expected to change in the study period 2017-2030.
Key Findings
According to DelveInsight, Prostate Cancer market in the 7MM is expected to change in the study period 2017–2030.
Country Wise Market Outlook
- According to the estimates, the highest market size of Prostate Cancer is found in the United States followed by the Germany and Japan in 2017 in the seven major markets.
Prostate cancer Drugs Uptake
- In mCSPC, currently market is dominated by hormonal therapies, mainly the first-generation hormonal therapies however label expansion of Xtandi and abiraterone acetate, approval of Erleada in mCSPC expected to help in driving the overall market of CSPC.
- Likewise, in nmCSPC also market is also mainly dominated by first-generation hormonal therapies. However, in future nmCSPC market size will be mainly driven by expected launch of ProstAtak and RV001.
- In metastatic CRPC, the current market is mainly dominated by Zytiga and Xtandi.
- In non-metastatic CRPC, Erleada with better clinical profile and early mover advantage will expect to generate maximum revenue in this setting
Prostate cancer Pipeline Development Activities
The drugs which are in pipeline includes:
1. Lynparza: Pre-registration
2. Rucaparib: Pre-registration
3. Opdivo (nivolumab): Phase III
4. 177Lu-PSMA-617: Phase III
5. Niraparib ± Abiraterone Acetate: Phase III
6. HC-1119 (Deuterated form of enzalutamide) : Phase III
7. Ipatasertib (RG7440) + Abiraterone Acetate: Phase III
8. Talazoparib ± Enzalutamide: Phase III
9. ProstAtak (AdV-tk) + valacyclovir: Phase III
10. Relugolix: Phase III
11. Capivasertib: Phase II
12. Seviteronel: Phase II
13. PRX302 (topsalysin) Phase II
14. RV001: Phase II
Product details and complete list of emerging products will be provided in the full report...
Pipeline Development Activities
Key Points
- In January 2020, the US FDA accepted the sNDA for Lynparza and granted Priority Review in the US for patients with mCRPC and deleterious or suspected deleterious germline or somatic HRR gene mutations, which have progressed following prior treatment with a new hormonal agent.
- It is worth mentioning that, the US FDA has accepted the company’s sNDA for Rubraca and granted priority review status to the application with a Prescription Drug User Fee Act (PDUFA) date of May 15, 2020. Clovis submitted the NDA submission for Rucaparib as monotherapy treatment of adult patients with BRCA1/2-mutant recurrent, metastatic castrate-resistant prostate cancer in November 2019.
Reimbursement Scenario in Prostate cancer
- In May 2019, Xtandi (enzalutamide) was not recommended, within its marketing authorization, treating high-risk hormone-relapsed non-metastatic prostate cancer in adults by National Institute for Health and Care Excellence (NICE).
- In March 2017, Zytiga (abiraterone acetate) received was favorable decision from the Haute Autorité de santé (HAS), within its marketing authorization for asymptomatic or low symptom castration-resistant metastatic prostate cancer in patients who failed androgen deprivation therapy.
KOL- Views
To keep up with current market trends, we take KOLs and SME's opinion working in Prostate cancer domain through primary research to fill the data gaps and validate our secondary research. Their opinion helps to understand and validate current and emerging therapies treatment patterns or Prostate cancer market trend. This will support the clients in potential upcoming novel treatment by identifying the overall scenario of the market and the unmet needs.
Competitive Intelligence Analysis
We perform Competitive and Market Intelligence analysis of the Prostate cancer Market by using various Competitive Intelligence tools that include - SWOT analysis, PESTLE analysis, Porter's five forces, BCG Matrix, Market entry strategies etc. The inclusion of the analysis entirely depends upon the data availability.
Scope of the Report
- The report covers the descriptive overview of Prostate cancer, explaining its causes, signs and symptoms, pathophysiology, diagnosis and currently available therapies
- Comprehensive insight has been provided into the Prostate cancer epidemiology and treatment in the 7MM
- Additionally, an all-inclusive account of both the current and emerging therapies for Prostate cancer are provided, along with the assessment of new therapies, which will have an impact on the current treatment landscape
- A detailed review of Prostate cancer market; historical and forecasted is included in the report, covering drug outreach in the 7MM
- The report provides an edge while developing business strategies, by understanding trends shaping and driving the global Prostate cancer market
Report Highlights
- In the coming years, Prostate cancer market is set to change due to the rising awareness of the disease, and incremental healthcare spending across the world; which would expand the size of the market to enable the drug manufacturers to penetrate more into the market
- The companies and academics are working to assess challenges and seek opportunities that could influence Prostate cancer R&D. The therapies under development are focused on novel approaches to treat/improve the disease condition
- Major players are involved in developing therapies for Prostate cancer. Launch of emerging therapies will significantly impact the Prostate cancer market
- A better understanding of disease pathogenesis will also contribute to the development of novel therapeutics for Prostate cancer
- Our in-depth analysis of the pipeline assets across different stages of development (Phase III and Phase II), different emerging trends and comparative analysis of pipeline products with detailed clinical profiles, key cross-competition, launch date along with product development activities will support the clients in the decision-making process regarding their therapeutic portfolio by identifying the overall scenario of the research and development activities
Prostate cancer Report Insights
- Prostate cancer Patient Population
- Prostate cancer Therapeutic Approaches
- Prostate cancer Pipeline Analysis
- Prostate cancer Market Size and Prostate cancer Market Trends
- Prostate cancer Market Opportunities
- Impact of upcoming Therapies
Prostate cancer Report Key Strengths
- 11 Years Forecast
- Prostate cancer 7MM Coverage
- Prostate cancer Epidemiology Segmentation
- Key Cross Competition
- Highly Analyzed Prostate cancer Market
- Prostate cancer Drugs Uptake
Prostate cancer Report Assessment
- Current Prostate cancer Treatment Practices
- Prostate cancer Unmet Needs
- Prostate cancer Pipeline Product Profiles
- Prostate cancer Market Attractiveness
- Prostate cancer Market Drivers and Prostate cancer Market Barriers
Key Questions
Market Insights:
- What was the Prostate cancer market share (%) distribution in 2017 and how it would look like in 2030?
- What would be the Prostate cancer total market size as well as market size by therapies across the 7MM during the forecast period (2017-2030)?
- What are the key findings pertaining to the market across 7MM and which country will have the largest Prostate cancer market size during the forecast period (2017-2030)?
- At what CAGR, the Prostate cancer market is expected to grow in 7MM during the forecast period (2017-2030)?
- What would be the Prostate cancer market outlook across the 7MM during the forecast period (2017-2030)?
- What would be the Prostate cancer market growth till 2030, and what will be the resultant market Size in the year 2030?
- How would the market drivers, barriers and future opportunities affect the market dynamics and subsequent analysis of the associated trends?
Epidemiology Insights:
- What is the disease risk, burden and unmet needs of the Prostate cancer?
- What is the historical Prostate cancer patient pool in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What would be the forecasted patient pool of Prostate cancer in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What will be the growth opportunities in the 7MM with respect to the patient population pertaining to Prostate cancer?
- Out of all 7MM countries, which country would have the highest prevalent population of Prostate cancer during the forecast period (2017-2030)?
- At what CAGR the population is expected to grow in 7MM during the forecast period (2017-2030)?
Current Treatment Scenario, Marketed Drugs and Emerging Therapies:
- What are the current options for the Prostate cancer treatment, along with the approved therapy?
- What are the current treatment guidelines for the treatment of Prostate cancer in the USA, Europe, and Japan?
- What are the Prostate cancer marketed drugs and their MOA, regulatory milestones, product development activities, advantages, disadvantages, safety and efficacy, etc.?
- How many companies are developing therapies for the treatment of Prostate cancer?
- How many therapies are developed by each company for Prostate cancer treatment?
- How many are emerging therapies in mid-stage, and late stage of development for Prostate cancer treatment?
- What are the key collaborations (Industry - Industry, Industry - Academia), Mergers and acquisitions, licensing activities related to the Prostate cancer therapies?
- What are the recent novel therapies, targets, mechanisms of action and technologies developed to overcome the limitation of existing therapies?
- What are the clinical studies going on for Prostate cancer and their status?
- What are the key designations that have been granted for the emerging therapies for Prostate cancer?
- What are the global historical and forecasted market of Prostate cancer?
Reasons to buy
- The Prostate cancer Market report will help in developing business strategies by understanding trends shaping and driving the Prostate cancer market
- To understand the future market competition in the Prostate cancer market and Insightful review of the key market drivers and barriers
- Organize sales and marketing efforts by identifying the best opportunities for Prostate cancer in the US, Europe (Germany, Spain, Italy, France, and the United Kingdom) and Japan
- Identification of strong upcoming players in the market will help in devising strategies that will help in getting ahead of competitors
- Organize sales and marketing efforts by identifying the best opportunities for Prostate cancer market
- To understand the future market competition in the Prostate cancer market
What is Prostate Cancer?
Prostate Cancer is a cancer of prostate gland, which is a part of male reproductive system. It is the fourth most common worldwide and third most prevalent type of cancer in the US.
At what CAGR is Prostate Cancer Market size expected to grow for the study period of 2017-30?
Prostate Cancer Market size is expected to increase at a CAGR of 8.1% for the study period 2017-30.
Which country is expected to dominate the Prostate Cancer Market in the 7MM?
Among 7MM countries, the US is anticipated to occupy the most substantial chunk of the Prostate Cancer Market size by 2030.
What is the Prostate Cancer prevalence expected by the end of 2020 in the 7MM?
Prostate Cancer prevalence in the 7MM is expected to reach 7,494,882 by 2020.
Which country is expected to account for the most significant prevalent cases for Prostate Cancer in the 7MM?
The US is expected to account for the highest Prostate Cancer prevalent cases.
How is the epidemiology of Prostate Cancer segmented?
Prostate Cancer epidemiology is segmented into Age-Specific Cases of Prostate Cancer, Total Treated Cases of mHSPC/CSPC and CRPC, Total Non-Metastatic and Metastatic Cases of CSPC/HSPC and CRPC, Total Diagnosed Cases of Prostate Cancer, Total Diagnosed Cases of Prostate Cancer patients by Clinical Stages and Total Treated cases of Non-Metastatic CSPC/HSPC and CRPC for the study period of 2017-30 for the 7MM.
What are the therapies available in the Prostate Cancer market?
The marketed therapies in the Prostate Cancer market are Xofigo (radium Ra 223 dichloride) of Bayer Health Care, Xtandi (Enzalutamide) of Astellas Pharma, Amgen’s Prolia (denosumab), Janssen’s Erleada (apalutamide), Sanofi’s Jevtana (Cabazitaxel) and Nubeqa (Darolutamide/ODM-201) of Bayer HealthCare/Orion Corporation.
What are the key companies working to advance the Prostate Cancer market?
AstraZeneca, Bristol-Myers Squibb, Novartis, Myovant Sciences, Janssen Research & Development, Hinova Pharmaceuticals USA, Hoffmann-La Roche, Pfizer/Astellas Pharma, Clovis Oncology, Advantagene or Candel Therapeutics, and Merck Sharp & Dohme along with others are the major pharma companies working to fuel the Prostate Cancer market.
What are the emerging therapies in the Prostate Cancer Pipeline for the study period of 2017-30?
Prostate Cancer Pipeline appears robust comprising Lynparza, Rucaparib, Opdivo (nivolumab), 177Lu-PSMA-617, Niraparib ± Abiraterone Acetate, HC-1119 (Deuterated form of enzalutamide), Ipatasertib (RG7440) + Abiraterone Acetate, Talazoparib ± Enzalutamide, ProstAtak (AdV-tk) + valacyclovir, Relugolix, Capivasertib, Seviteronel, PRX302 (topsalysin), and RV001.
.png&w=256&q=75)