Download Case Study to Unlock Valuable Insights
Fill out the form to gain access to exclusive content and data-driven strategies

ASCO 2024 Conference Intelligence around HER2 Negative Breast Cancer assets
- Home
- case study
- conference intelligence product assessment
Objective
The objective of DelveInsight's engagement with this large US-headquartered pharmaceutical company was to provide comprehensive, strategic conference coverage of HER2-negative breast cancer drug development activities at ASCO 2024 (American Society of Clinical Oncology Annual Meeting). The initiative aimed to capture critical competitive intelligence in real time, including detailed product descriptions, research and development activities, pipeline milestones, future development plans, and key opinion leader (KOL) perspectives. By systematically monitoring, analyzing, and synthesizing data presentations across multiple concurrent sessions, the client could maintain competitive awareness, inform strategic decision-making, and enable senior leadership to respond proactively to emerging competitive dynamics and scientific developments within the rapidly evolving HER2-negative breast cancer therapeutic landscape.
The complexity of the challenge was multifaceted. ASCO presents thousands of abstracts, oral presentations, and poster sessions simultaneously across numerous therapeutic areas and drug classes. For a pharmaceutical company tracking competitive activity in HER2-negative breast cancer, a high-priority indication with intense competitive dynamics, manually covering all relevant sessions while filtering signal from noise proved operationally impossible without dedicated, expert resources.
Moreover, the company required more than passive data collection. Strategic conference coverage demanded:
- Pre-congress planning: Identifying which sessions, presentations, and posters contained relevant HER2-negative breast cancer data
- Real-time capture: Attending high-priority sessions and documenting presentations, clinical data, development milestones, and strategic positioning
- Primary research integration: Conducting KOL interviews and stakeholder consultations during the congress to capture expert perspectives on data significance and competitive implications
- Strategic synthesis: Translating raw data and presentations into actionable business intelligence aligned with the client's key intelligence topics (KITs) and key intelligence questions (KIQs)
- Rapid dissemination: Delivering timely updates to senior leadership during the congress to enable a proactive competitive response
Our Methodology: Focused Conference Coverage Plan and Real-Time Intelligence Delivery
DelveInsight deployed a comprehensive, structured approach to conference coverage, combining pre-congress planning, prioritized real-time session monitoring, primary KOL research, and strategic synthesis aligned with client intelligence needs.
Congress Coverage Support: Strategic Planning and Session Prioritization
DelveInsight supported the client by creating a focused conference coverage plan designed to maximize intelligence value and ensure a comprehensive assessment of targeted HER2-negative breast cancer drug candidates. This pre-congress planning phase included:
- Session extraction and classification: The DelveInsight team systematically reviewed the ASCO 2024 scientific program, extracting all sessions, oral presentations, poster discussions, and abstract presentations relevant to HER2-negative breast cancer therapeutics
- Priority-based segmentation: Sessions were classified by strategic priority based on competitive relevance, data novelty, clinical development stage, and alignment with client intelligence requirements
- Resource allocation strategy: High-priority sessions were designated for dedicated live coverage with real-time capture, while lower-priority sessions were monitored through post-congress abstract review and selective follow-up
Primary Research: KOL and Stakeholder Insights
In addition to capturing presentation data, DelveInsight conducted targeted primary research during ASCO 2024 to supplement quantitative data with qualitative expert perspectives. This included:
- Engaging key opinion leaders (KOLs) attending the congress to gather insights on clinical data interpretation, competitive positioning, and therapeutic significance
- Conducting stakeholder interviews with researchers, clinicians, and industry experts to contextualize development milestones within broader treatment paradigm shifts
- Capturing real-time expert perspectives on data quality, regulatory implications, and commercial potential
Real-Time Capture and Analysis
During ASCO 2024, the DelveInsight team executed the conference coverage plan:
- Detailed presentation capture: Attended prioritized sessions and documented comprehensive details including clinical trial design, efficacy and safety data, patient population characteristics, development milestones, regulatory strategy, and future development plans
- Business insight extraction: Translated clinical presentations into strategic business intelligence, identifying competitive threats, partnership opportunities, market access implications, and portfolio positioning insights
- Key Intelligence Topics (KITs) and Key Intelligence Questions (KIQs) alignment: All captured data and insights were structured around the client's pre-defined strategic intelligence priorities, ensuring relevance and actionability
Rapid Synthesis and Communication
Throughout the congress, DelveInsight delivered timely intelligence updates to the client's senior leadership, enabling near-real-time awareness of competitive developments and informed strategic response during the congress itself.
Results: Proactive Competitive Response and Strategic Decision Support
The comprehensive conference coverage engagement delivered critical strategic outcomes:
Proactive Response to Competitor Activities at ASCO 2024
The deliverables enabled the pharmaceutical company to respond proactively to competitor activities showcased at ASCO 2024. By receiving real-time intelligence on competitive HER2-negative breast cancer drug candidates, including clinical data results, development milestones, regulatory strategies, and positioning statements, the company could assess competitive threats, identify partnership opportunities, and adjust strategic priorities during the congress rather than discovering critical developments weeks later through secondary channels.
Timely Leadership Communication
Senior internal stakeholders, including executive leadership, clinical development teams, and commercial strategy groups, were kept informed of the latest business-critical updates throughout ASCO 2024. This real-time intelligence dissemination enabled leadership to:
- Make informed strategic decisions regarding pipeline prioritization and resource allocation
- Identify emerging competitive dynamics requiring immediate strategic response
- Assess partnership or licensing opportunities revealed through congress presentations
- Communicate externally with investors, partners, and stakeholders from an informed competitive position
Comprehensive Intelligence Portfolio
Beyond real-time coverage, the engagement delivered a comprehensive post-congress intelligence portfolio, including:
- Detailed documentation of all relevant HER2-negative breast cancer presentations and clinical data
- Product descriptions, research and development timelines, and key milestones for competitive assets
- Primary research-derived KOL insights contextualizing data within therapeutic and competitive landscapes
- Strategic synthesis aligned with the client's key intelligence topics and questions
- Actionable recommendations for competitive response and strategic positioning
Strategic Competitive Advantage
By outsourcing specialized conference coverage to DelveInsight's expert team, the pharmaceutical company achieved the strategic competitive advantage without diverting critical internal clinical and commercial resources from core responsibilities. The client received expert-level intelligence capture, strategic synthesis, and KOL insights, the capabilities that would have required significant internal coordination, travel logistics, and opportunity costs.
Our Related Services
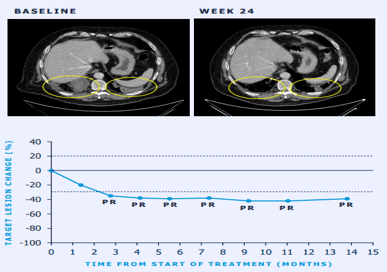
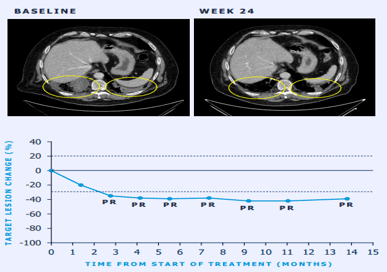
Sample Visuals
Get Visuals