Axial Spondyloarthritis market size is projected to grow rapidly with a significant CAGR by 2034
Get a Sneak Peek at the Latest axial spondyloarthritis market size and forecast Report
The Axial Spondyloarthritis market size is anticipated to grow with a significant CAGR during the study period (2020-2034). DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Axial Spondyloarthritis landscape.
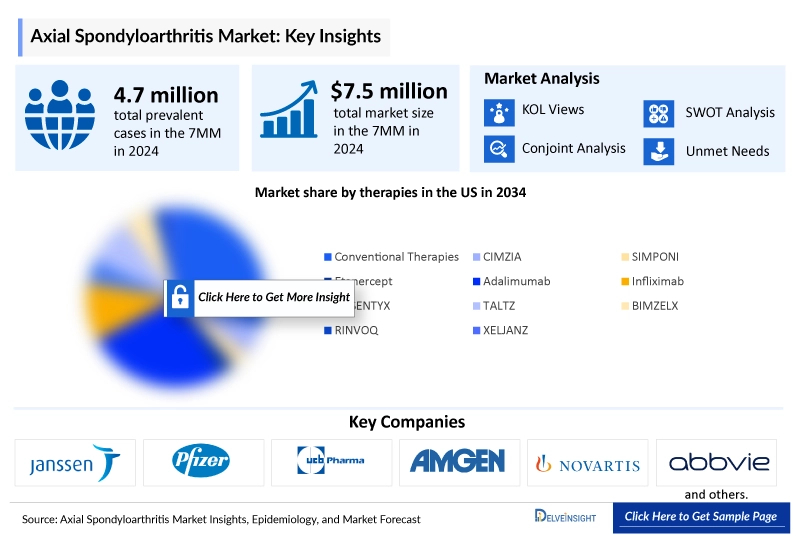
According to DelveInsight, the total prevalent population of axial spondyloarthritis (axSpA) in the 7 major markets (7MM) was approximately 4,575,000 in 2023, and this figure is projected to increase significantly over the study period from 2020 to 2034. Of these, about 3,981,346 individuals were identified as overall prevalent cases in the 7MM, with numbers expected to continue rising between 2024 and 2034.
DelveInsight estimates that the total prevalent population of axial spondyloarthritis (axSpA) in the 7 major markets (7MM) was approximately 4,575,000 in 2023, with a notable increase expected over the period from 2020 to 2034. In the US, the diagnosed population of non-radiographic axSpA was around 569,000 cases in 2023. Among the EU4 and UK, Germany reported the highest number of prevalent axSpA cases, followed closely by Spain.
DelveInsight’s report “Axial Spondyloarthritis Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Axial Spondyloarthritis landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Axial Spondyloarthritis market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Axial Spondyloarthritis market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Axial Spondyloarthritis Market Forecast
Some of the key facts of the Axial Spondyloarthritis Market Report:
- The US FDA has approved UCB’s CIMZIA, Eli Lilly’s TALTZ, Novartis’s COSENTYX, and AbbVie’s RINVOQ for the treatment of both radiographic (r-axSpA) and non-radiographic axial spondyloarthritis (nr-axSpA). In comparison, Immunex/Amgen’s ENBREL, Pfizer’s XELJANZ, and Johnson & Johnson’s SIMPONI are approved solely for ankylosing spondylitis. Additionally, LUMICEF is approved in Japan for both r-axSpA and nr-axSpA
- Key Axial Spondyloarthritis Companies: Janssen Pharmaceuticals, Pfizer, UCB Biopharma, Celgene, Inmagene Biopharmaceuticals, Galapagos NV, Kyowa Kirin Co., Ltd., Eli Lilly and Company, Merck Sharp & Dohme LLC, Pfizer, Novartis, Jiangsu HengRui Medicine, UCB Biopharma, and others
- Key Axial Spondyloarthritis Therapies: SIMPONI (golimumab), XELJANZ (tofacitinib), Bimekizumab, CC-99677, ABY- 035, Filgotinib, KHK4827, Ixekizumab, Golimumab, etanercept, Secukinumab, SHR0302, Certolizumab Pegol, and others
- The Axial Spondyloarthritis epidemiology based on gender analyzed that the prevalent population of Axial Spondyloarthritis showed a male predominance, whereas, nr-axSpA showed female predominance.
- The Axial Spondyloarthritis market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Axial Spondyloarthritis pipeline products will significantly revolutionize the Axial Spondyloarthritis market dynamics.
Axial Spondyloarthritis Overview
Axial Spondyloarthritis is a type of chronic inflammatory arthritis primarily affecting the spine and the sacroiliac joints, which connect the lower spine to the pelvis. This condition can lead to pain, stiffness, and decreased mobility in the affected areas. AxSpA encompasses two subtypes: non-radiographic axial spondyloarthritis (nr-axSpA), where there is no visible damage on X-rays, and ankylosing spondylitis (AS), where changes are visible on X-rays, including possible fusion of the vertebrae.
Get a Free sample for the Axial Spondyloarthritis Market Report:
https://www.delveinsight.com/report-store/axial-spondyloarthritis-market
Key Factors Driving The Axial Spondyloarthritis Market:
- Rising Disease Prevalence and Improved Recognition: Increasing awareness and better clinical differentiation between radiographic (AS) and non-radiographic axSpA are expanding the diagnosed patient population.
- Strong Adoption of Biologics and Targeted Therapies: Growing use of TNF inhibitors, IL-17 inhibitors, and JAK inhibitors significantly boosts market demand due to their proven efficacy in symptom control and disease progression.
Advancements in Imaging and Diagnostic Tools: Wider use of MRI and early diagnostic protocols helps identify axSpA sooner, leading to earlier treatment initiation and higher therapy uptake. - Expansion of Treatment Guidelines and Reimbursement Support: Updated international clinical guidelines and broader insurance coverage improve access to advanced therapies.
- Active Pipeline and Continuous R&D Investment: Ongoing research into novel biologics, small-molecule inhibitors, and personalized therapeutic approaches strengthens future market growth.
Axial Spondyloarthritis Epidemiology
The Axial Spondyloarthritis epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Axial Spondyloarthritis Epidemiology Segmentation:
The Axial Spondyloarthritis market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalent Cases of Axial Spondyloarthritis in the 7MM
- Total Diagnosed Cases of Axial Spondyloarthritis in the 7MM
- Gender-specific Cases of Axial Spondyloarthritis in the 7MM
- Gene-specific Cases of Axial Spondyloarthritis in the 7MM
- Age-specific Cases of Axial Spondyloarthritis in the 7MM
- Total Treated Cases of Axial Spondyloarthritis in the 7MM
Download the report to understand which factors are driving Axial Spondyloarthritis epidemiology trends @ Axial Spondyloarthritis Epidemiology Forecast
Recent Developments In The Axial Spondyloarthritis Treatment Landscape:
- In January 2025, MoonLake Immunotherapeutics (Nasdaq: MLTX), a clinical-stage biotechnology company developing advanced therapies for inflammatory diseases, announced that patient screening has begun for three new trials evaluating sonelokimab, an investigational Nanobody® therapy. These trials expand the clinical program to include adolescent hidradenitis suppurativa (HS), palmoplantar pustulosis (PPP), and axial spondyloarthritis (axSpA), alongside existing studies in adult patients with HS and active psoriatic arthritis (PsA).
- In November 2024, UCB, a global biopharmaceutical company, announced new two-year data demonstrating a sustained clinical response for BIMZELX® (bimekizumab-bkzx), an IL-17A and IL-17F inhibitor, in adults with active psoriatic arthritis (PsA), active non-radiographic axial spondyloarthritis (nr-axSpA) with objective signs of inflammation, and active ankylosing spondylitis (AS). These findings come from the Phase 3 studies BE OPTIMAL (PsA), BE COMPLETE (PsA), and their open-label extension BE VITAL, as well as BE MOBILE 1 (nr-axSpA), BE MOBILE 2 (AS), and their open-label extension BE MOVING.
- In September 2024, UCB, a global biopharmaceutical company, announced that the U.S. Food and Drug Administration (FDA) has approved BIMZELX® (bimekizumab-bkzx) for three indications: the treatment of adults with active psoriatic arthritis (PsA), active non-radiographic axial spondyloarthritis (nr-axSpA) with objective signs of inflammation, and active ankylosing spondylitis (AS). BIMZELX is the first treatment approved for all three conditions, designed to selectively inhibit two key cytokines involved in inflammation—interleukin 17A (IL-17A) and interleukin 17F (IL-17F). These approvals follow its initial U.S. approval in October 2023 for moderate-to-severe plaque psoriasis in adults eligible for systemic therapy or phototherapy.
Axial Spondyloarthritis Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Axial Spondyloarthritis market or expected to get launched during the study period. The analysis covers Axial Spondyloarthritis market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Axial Spondyloarthritis Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Axial Spondyloarthritis Therapies and Key Companies
- SIMPONI (golimumab): Janssen Pharmaceuticals
- XELJANZ (tofacitinib): Pfizer
- Bimekizumab: UCB Biopharma
- CC-99677: Celgene
- ABY- 035: Inmagene Biopharmaceuticals
- Filgotinib: Galapagos NV
- KHK4827: Kyowa Kirin Co., Ltd.
- Ixekizumab: Eli Lilly and Company
- Golimumab: Merck Sharp & Dohme LLC
- etanercept: Pfizer
- Bimekizumab: UCB Biopharma
- Secukinumab: Novartis
- SHR0302: Jiangsu HengRui Medicine
- Certolizumab Pegol: UCB Biopharma
Discover more about therapies set to grab major Axial Spondyloarthritis market share @ Axial Spondyloarthritis Treatment Market
Axial Spondyloarthritis Market Drivers
- Rising prevalence and improved recognition of axSpA, including both radiographic (AS) and non-radiographic forms.
- Strong adoption of biologics and targeted therapies, such as TNF inhibitors, IL-17 inhibitors, and JAK inhibitors.
- Advancements in imaging technologies (MRI) and early diagnostic protocols facilitating timely treatment.
- Updated clinical guidelines and growing reimbursement support improving patient access to advanced therapies.
- Active R&D and robust pipeline of novel therapies, enhancing future treatment options.
Axial Spondyloarthritis Market Barriers
- High cost of biologic therapies limiting accessibility for many patients.
- Delay in diagnosis due to overlapping symptoms with other musculoskeletal disorders.
- Side effects and long-term safety concerns associated with current treatment options.
- Limited awareness about non-radiographic axSpA, leading to underdiagnosis in some populations.
- Stringent regulatory requirements for novel therapies, potentially slowing market entry.
Scope of the Axial Spondyloarthritis Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Axial Spondyloarthritis Companies: Janssen Pharmaceuticals, Pfizer, UCB Biopharma, Celgene, Inmagene Biopharmaceuticals, Galapagos NV, Kyowa Kirin Co., Ltd., Eli Lilly and Company, Merck Sharp & Dohme LLC, Pfizer, Novartis, Jiangsu HengRui Medicine, UCB Biopharma, and others
- Key Axial Spondyloarthritis Therapies: SIMPONI (golimumab), XELJANZ (tofacitinib), Bimekizumab, CC-99677, ABY- 035, Filgotinib, KHK4827, Ixekizumab, Golimumab, etanercept, Secukinumab, SHR0302, Certolizumab Pegol, and others
- Axial Spondyloarthritis Therapeutic Assessment: Axial Spondyloarthritis current marketed and Axial Spondyloarthritis emerging therapies
- Axial Spondyloarthritis Market Dynamics: Axial Spondyloarthritis market drivers and Axial Spondyloarthritis market barriers
- Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Axial Spondyloarthritis Unmet Needs, KOL’s views, Analyst’s views, Axial Spondyloarthritis Market Access and Reimbursement
To know more about Axial Spondyloarthritis companies working in the treatment market, visit @ Axial Spondyloarthritis Clinical Trials and Therapeutic Assessment
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Axial Spondyloarthritis (axSpA) - Epidemiology Insight - 2034
DelveInsight's Axial Spondyloarthritis (axSpA) - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of Axial Spondy..
Axial Spondyloarthritis - Pipeline Insight, 2025
Axial Spondyloarthritis Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across the Axial Spondyloarthritis market.