22q11.2 Deletion Syndrome Market Summary
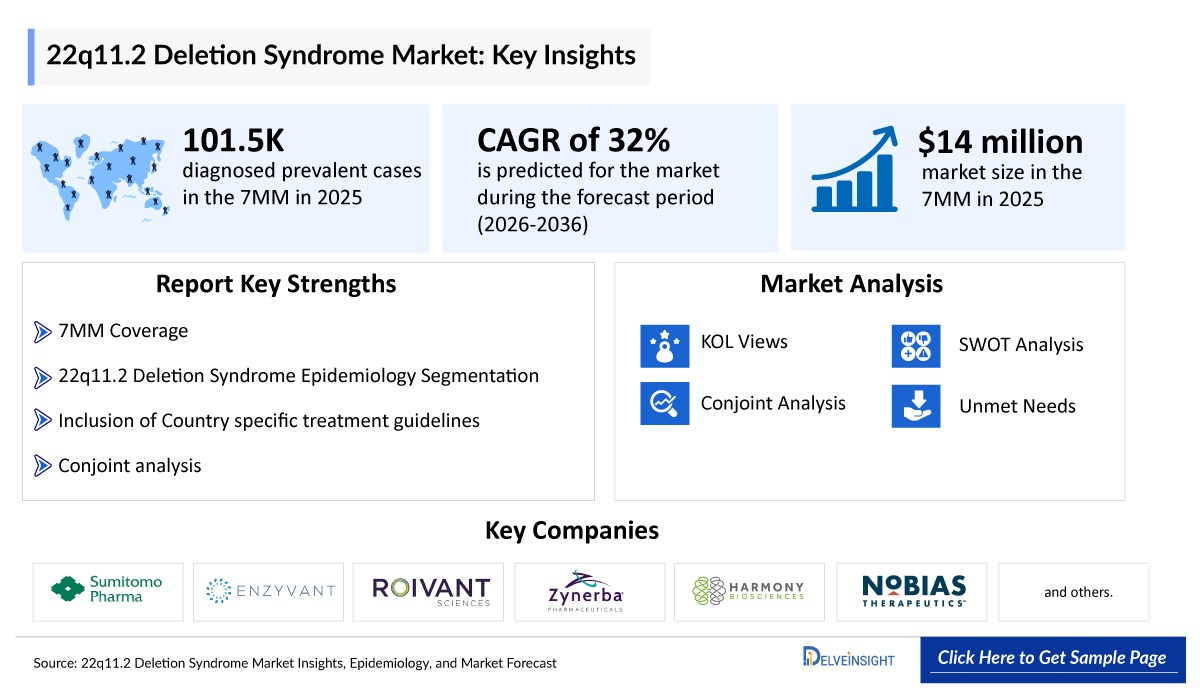
- The 22q11.2 Deletion Syndrome market size was valued at approximately USD 14 million in 2025 and is projected to grow at a CAGR of 32% during the forecast period from 2026 to 2036.
- The leading 22q11.2 Deletion Syndrome companies developing therapies in the treatment market include - Sumitomo Pharma, Enzyvant, Roivant Sciences, Zynerba Pharmaceuticals, Harmony Biosciences, Nobias Therapeutics, and others.
22q11.2 Deletion Syndrome Market & Epidemiology Insights
- 22q11.2 Deletion Syndrome also known as DiGeorge syndrome or velocardiofacial syndrome, is a genetic disorder caused by a microdeletion on chromosome 22q11.2, resulting in multisystem manifestations including congenital heart defects, immune dysfunction, hypocalcemia, and distinct craniofacial features.
- It is a lifelong condition and the most common chromosomal microdeletion syndrome, significantly impacting development, cognition, and behavioral health, with an estimated prevalence of ~1 in 4,000 live births.
- A substantial proportion of patients experience neurocognitive impairment and psychiatric disorders, including a markedly elevated risk of schizophrenia (~25% in adults), making 22q11.2DS one of the strongest known genetic risk factors for severe mental illness.
- The underlying pathophysiology involves gene dosage imbalance (notably TBX1), disrupted embryologic development, immune dysfunction, and altered neurodevelopmental pathways, complicating target identification and therapeutic development.
- Diagnosis is often delayed due to phenotypic heterogeneity and absence of standardized clinical screening pathways, with many cases identified only after multiple specialist referrals, highlighting a persistent diagnostic gap and missed early intervention window.
- The current management remains fragmented and symptom-driven, requiring lifelong multidisciplinary care across cardiology, immunology, endocrinology, and psychiatry, which contributes to high healthcare utilization and caregiver burden.
- There are no FDA-approved disease-modifying therapies, and treatment is limited to supportive interventions such as cardiac surgery, calcium supplementation, immune support, and psychiatric management, underscoring a significant unmet medical need.
- The emerging pipeline for 22q11.2 Deletion Syndrome remains highly limited, with a few investigational therapies in development, including NB-001 (Nobias Therapeutics).
22q11.2 Deletion Syndrome Market Size and Forecast in the 7MM
-
2025 22q11.2 Deletion Syndrome Market Size: ~USD 14 million
-
22q11.2 Deletion Syndrome Growth Rate (2026–2036): ~32% CAGR
Download the Sample PDF to Get More Insight @ 22q11.2 Deletion Syndrome Market Forecast
Key Factors Driving the 22q11.2 Deletion Syndrome Market
- Advancements in Genetic Testing and Diagnosis: Improved molecular diagnostic technologies and prenatal screening are enabling earlier and more accurate detection of 22q11.2 Deletion Syndrome, supporting market growth.
- Increasing Awareness of Rare Genetic Disorders: Rising awareness among healthcare professionals, caregivers, and patient advocacy organizations is driving higher diagnosis and treatment rates.
- Growing Research and Development Activities: Pharmaceutical and biotechnology companies are expanding research efforts to develop targeted therapies and supportive treatment options for associated complications.
- Supportive Government and Regulatory Initiatives: Orphan drug incentives, research funding, and favorable regulatory frameworks are encouraging innovation in rare disease therapeutics.
- Rising Demand for Multidisciplinary Care: The increasing need for integrated management of cardiac, immunological, neurological, and developmental complications is boosting healthcare spending in this market.
DelveInsight's ‘22q11.2 Deletion Syndrome Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the 22q11.2 Deletion Syndrome, historical and forecasted epidemiology, as well as the 22q11.2 Deletion Syndrome market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The 22q11.2 Deletion Syndrome market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates 22q11.2 Deletion Syndrome patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in 22q11.2 Deletion Syndrome and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the 22q11.2 Deletion Syndrome Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
22q11.2 Deletion Syndrome Market CAGR (Forecast period) |
~32% (2026–2036) |
|
22q11.2 Deletion Syndrome Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
22q11.2 Deletion Syndrome Companies |
|
|
22q11.2 Deletion Syndrome Therapies |
|
|
22q11.2 Deletion Syndrome Market |
Segmented by
|
|
Analysis |
• Addressable Patient Population • Market Drivers and Market Barriers • Cost Assumptions and Pricing Analogues • KOL Views • SWOT Analysis • Reimbursement • Conjoint Analysis • Unmet Need |
22q11.2 Deletion Syndrome Disease Understanding
22q11.2 Deletion Syndrome Overview and Diagnosis
22q11.2 Deletion Syndrome (22q11.2DS) is a chromosomal microdeletion disorder caused by a hemizygous deletion at the 22q11.2 locus, leading to haploinsufficiency of critical developmental genes, particularly TBX1. It presents with multisystem involvement, most commonly including conotruncal congenital heart defects, thymic hypoplasia with immune dysfunction, hypoparathyroidism-associated hypocalcemia, and craniofacial anomalies, along with significant neurodevelopmental and psychiatric manifestations such as learning disabilities, autism spectrum disorder, and a markedly increased risk of schizophrenia. With a prevalence of approximately 1 in 3,000–4,000 live births, it is the most common chromosomal microdeletion syndrome; however, variable expressivity and age-dependent clinical evolution often lead to underdiagnosis, particularly in individuals without major congenital anomalies.
Diagnosis of 22q11.2DS is genetically confirmed, with chromosomal microarray (CMA) as the current gold standard, enabling detection of both typical (~3 Mb) and smaller nested deletions, while FISH is now less preferred due to limited sensitivity for atypical variants. Clinical suspicion is typically raised in patients presenting with congenital heart disease, hypocalcemia, immune deficiency, or developmental delay, but the absence of standardized screening criteria contributes to frequent diagnostic delays. Increasing adoption of genomic-first approaches and prenatal screening (NIPT) is improving early detection, although milder phenotypes continue to be diagnosed later in childhood or adulthood, reflecting persistent gaps in recognition and testing practices.
Further details are provided in the report....
Current 22q11.2 Deletion Syndrome Treatment Landscape
Management of 22q11.2 Deletion Syndrome is supportive, symptom-driven, and requires lifelong multidisciplinary care, as no disease-modifying therapies are currently available. Treatment focuses on addressing organ-specific manifestations, including surgical correction of congenital heart defects, calcium and vitamin D supplementation for hypocalcemia, and immune management ranging from infection prophylaxis to immunoglobulin replacement in selected patients, with thymus transplantation reserved for severe T-cell deficiency. Additional interventions include surgical repair of palatal abnormalities, early speech, occupational, and behavioral therapies for developmental delays, and psychiatric management for conditions such as ADHD, anxiety, and schizophrenia. Given the evolving and multisystem nature of the disorder, care coordination across specialties is critical to optimize long-term outcomes and quality of life.
Further details related to country-based variations are provided in the report....
22q11.2 Deletion Syndrome Unmet Needs
The section “unmet needs of 22q11.2 Deletion Syndrome” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of FDA-approved therapies
- The associated risk of neurological and psychological disorders
- Hidden disease burden driven by diagnostic delays
- Lack of standardized lifespan management frameworks, and others…..
Note: Comprehensive unmet needs insights in 22q11.2 Deletion Syndrome and their strategic implications are provided in the full report....
22q11.2 Deletion Syndrome Epidemiology
The 22q11.2 Deletion Syndrome epidemiology section provides insights about the historical and current 22q11.2 Deletion Syndrome patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the 22q11.2 Deletion Syndrome market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings from 22q11.2 Deletion Syndrome Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, in 2025, the total number of diagnosed prevalent cases of 22q11.2 deletion syndrome in the 7MM was ~ 101,500.
- In the United States, among age-specific diagnosed prevalent cases of 22q11.2 deletion syndrome, infant accounted for the majority of cases (~12,550), followed by 1 ̶ 5 years (~10,300), 6 ̶ 12 years (~19,550), 18+ years (~7,000), and 13 ̶ 17 years (~4,800) in 2025.
- Among EU4 and the UK Germany accounted for the highest number of diagnosed prevalent cases of 22q11.2 Deletion Syndrome (~9,300 cases), while Spain reported the lowest burden (~6,100 cases) in 2025, highlighting regional variability in diagnosis and disease recognition.
- In Japan, the diagnosed prevalent cases of 22q11.2 deletion syndrome with behavioral and psychiatric phenotypes were approximately 7,650 in 2025.
22q11.2 Deletion Syndrome Epidemiology Segmentation
- Total Prevalent Cases of 22q11.2 Deletion Syndrome
- Total Diagnosed Prevalent Cases of 22q11.2 Deletion Syndrome
- Age-specific Diagnosed Prevalent Cases of 22q11.2 Deletion Syndrome
- Total Diagnosed Prevalent Cases of 22q11.2 Deletion Syndrome with Behavioral And Psychiatric
- Total Treated Cases of 22q11.2 Deletion Syndrome
Recent Developments in the 22q11.2 Deletion Syndrome Treatment Landscape
- As per Harmony Biosciences’ Q3 2025 presentation, Zygel (ZYN002) development program in 22q11.2 deletion syndrome has been paused pending the full review of the RECONNECT data
- In June 2025, Nobias Therapeutics has secured preliminary alignment with the US FDA on registrational endpoints for NB-001 for the treatment of neuropsychiatric symptoms associated with 22q11.2 deletion syndrome. This critical milestone clarifies the regulatory expectations for potential approval.
22q11.2 Deletion Syndrome Drug Analysis & Competitive Landscape
The 22q11.2 deletion syndrome drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III 22q11.2 Deletion Syndrome clinical trials. It covers the mechanism of action, clinical trial data, patents, collaborations, and strategic partnerships, upcoming key catalysts for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the 22q11.2 deletion syndrome treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the 22q11.2 deletion syndrome therapeutics market.
Approved Therapies for 22q11.2 deletion syndrome
RETHYMIC (formerly RVT-802): Sumitomo Pharma/Enzyvant/Roivant Sciences
RETHYMIC is a thymus tissue–based regenerative therapy indicated for patients with congenital athymia, a rare and life-threatening immune deficiency often associated with 22q11.2 Deletion Syndrome. The therapy involves implantation of allogeneic cultured thymus tissue to restore T-cell development and immune function, addressing the underlying cause of immunodeficiency. Originally developed by Enzyvant, the therapy received Breakthrough Therapy and Regenerative Medicine Advanced Therapy (RMAT) designations from the US FDA, enabling expedited development and regulatory support.
- Approved in 2021, RETHYMIC represents a first-in-class, disease-modifying treatment option with the potential to significantly improve survival and immune reconstitution in affected pediatric patients.
22q11.2 Deletion Syndrome Pipeline Analysis
NB-001: Nobias Therapeutics
NB-001 is an investigational non-stimulant activator of multiple metabotropic glutamate receptors (mGluRs) for 22q11.2 deletion syndrome targeting disrupted neuronal signaling underlying cognitive and psychiatric symptoms. By modulating synaptic plasticity and key neurobiological pathways, it aims to deliver disease-modifying benefits beyond symptomatic management, with potential to improve cognition, behavior, and overall functional outcomes in this underserved patient population.
The drug has completed Phase II clinical trial and has received the FDA’s Rare Pediatric Disease (RPD) designation, recognizing its potential to address serious conditions affecting children.
Note: Detailed emerging therapies assessment will be provided in the final report....
22q11.2 Deletion Syndrome Key Players, Market Leaders and Emerging Companies
- Sumitomo Pharma
- Enzyvant
- Roivant Sciences
- Zynerba Pharmaceuticals
- Harmony Biosciences
- Nobias Therapeutics, and others
22q11.2 Deletion Syndrome Market Outlook
As of now, there is no curative therapy for 22q11.2 deletion syndrome, and management remains largely supportive and multidisciplinary. Treatment is tailored to clinical severity, age, and organ involvement, with early-life focus on congenital heart defects, immune dysfunction, hypocalcemia, feeding issues, and palatal abnormalities. As patients age, care shifts toward neurodevelopmental, cognitive, and psychiatric management. Current interventions are symptomatic, including antibiotics, calcium and vitamin D supplementation, hormone replacement, surgical correction, and developmental therapies. While these approaches have improved survival, they do not address the underlying genetic defect, leaving significant unmet needs.
Notably, RVT-802, developed by Reviva Pharmaceuticals, has received FDA approval in October 2021 for immune reconstitution in pediatric patients with congenital athymia associated with complete DiGeorge syndrome/anomaly, marking a significant advancement in addressing severe immunodeficiency within this spectrum.
Furthermore, the emerging landscape of 22q11.2 deletion syndrome is not very robust. Zygel (transdermal cannabidiol gel) by Zynerba Pharmaceuticals emerged as a novel approach to treat behavioral symptoms in 22q11.2 deletion syndrome, leveraging THC-free cannabidiol via a transdermal route to ensure stable exposure and improved tolerability. In 2020, the Phase II INSPIRE study was initiated, and by 2022, results demonstrated meaningful improvements in anxiety, mood, and behavior with a favorable safety profile, validating early clinical potential. Despite receiving FDA orphan drug designation, the company decided to pause the development of Zygel in 22q11.2 deletion syndrome pending a full review of data from the RECONNECT trial in Fragile X syndrome, reflecting strategic pipeline prioritization and leaving the asset as a promising but currently on-hold opportunity.
In addition, NB-001 (fasoracetam) by Nobias Therapeutics is a mechanism-driven therapy targeting glutamatergic dysfunction in 22q11.2 deletion syndrome. The drug has completed Phase II trial and demonstrated favorable safety and encouraging improvements in anxiety, ADHD, and behavioral symptoms. With orphan and rare pediatric disease designations and regulatory alignment achieved, NB-001 is advancing as a leading late-stage candidate in 22q11.2DS.
Overall, the 22q11.2 deletion syndrome market is poised for steady evolution, driven by rising awareness, and early but promising targeted therapies. Despite cautious pipeline progression and strategic pauses, emerging mechanism-based treatments are expected to gradually shape market growth across the 7MM from 2022–2036.
- Among the 7MM, the US accounted for the largest market size of 22q11.2 Deletion Syndrome. i.e., USD ~8 million in 2025.
- In EU4 and the UK, Germany and the UK captured the largest share of 22q11.2 deletion syndrome market, while Spain accounted for the least share.
- The entry of NB-001 is expected capture a significant share in the 22q11.2 Deletion Syndrome landscape.
- Regulatory alignment on registrational endpoints for NB-001 marks a critical inflection point for the 22q11.2 deletion syndrome market, signaling increased development maturity and reducing approval uncertainty. This milestone is expected to accelerate clinical advancement and reinforce momentum toward the first disease-specific therapies in a high unmet need landscape.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies
The 22q11.2 Deletion Syndrome market comprises regenerative therapies, small molecules, and other emerging modalities, each targeting key mechanisms such as peripheral nerve hyperexcitability, ion channel modulation, and neuroprotection.
- Regenerative therapies: This includes agents such as RETHYMIC (formerly RVT-802), a thymus tissue–based regenerative therapy addressing underlying immune deficiency.
- Small molecules: This segment includes agents such as NB-001 (fasoracetam), which targets glutamatergic pathways via metabotropic glutamate receptor modulation, aiming to address core neuropsychiatric manifestations including anxiety, ADHD, and social dysfunction.
These emerging therapies reflect a shift toward mechanism-based, disease-specific approaches, moving beyond symptomatic management to address the underlying neurobiology of 22q11.2 deletion syndrome.
22q11.2 Deletion Syndrome Drug Uptake
This section focuses on the uptake rate of potential 22q11.2 Deletion Syndrome drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the 22q11.2 Deletion Syndrome drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The uptake of emerging therapies is expected to vary based on clinical positioning, mechanism of action, and strength of evidence, particularly across immune restoration and neuropsychiatric management. As an approved therapy, RETHYMIC (thymus tissue–based therapy) is expected to witness relatively faster uptake in eligible patients due to its disease-modifying potential in addressing the underlying immune deficiency.
While NB-001 (fasoracetam) is expected to gain traction steadily, supported by its mechanism-driven approach targeting glutamatergic dysfunction and growing regulatory clarity. Overall, in the absence of approved therapies specifically for neuropsychiatric symptoms in 22q11.2 deletion syndrome, uptake dynamics are expected to remain measured but increasingly favorable with strong clinical data and real-world evidence.
Detailed insights into emerging therapies' drug uptake are included in the report....
22q11.2 Deletion Syndrome Market Access and Reimbursement of Approved Therapies
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
The United States
|
US Reimbursement of Therapies Approved for 22q11.2 Deletion Syndrome | |
|
Drug/Therapy |
Access Program |
|
RETHYMIC |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
22q11.2 Deletion Syndrome Therapies Price Scenario & Trends
Pricing and analogue assessment of 22q11.2 deletion syndrome therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and the understanding of how pricing influences market access, adherence, and long-term uptake.
Industry Experts and Physician Views for 22q11.2 Deletion Syndrome
To keep up with 22q11.2 deletion syndrome market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on 22q11.2 deletion syndrome emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in 22q11.2 deletion syndrome, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“The comprehensive prenatal ultrasound monitoring plays a pivotal role in the management of fetuses with 22q11.2 deletion syndrome, enabling early detection of growth restriction, polyhydramnios, and associated structural anomalies. Such proactive assessment facilitates timely multidisciplinary planning for neonatal care, ensuring that clinical teams are better equipped to manage immediate postnatal needs while also allowing for early parental counseling and preparedness.” |
|
Germany |
“The clinical management of 22q11.2 deletion syndrome must be dynamic and age-specific, with treatment priorities evolving from structural and immunological concerns in early life to neurodevelopmental, endocrine, and psychiatric management in adolescence and adulthood.” |
DelveInsight’s analysts connected with 10+ KOLs to gather insights at the country level. Centers such as University of Michigan Hospitals & Health Centers, University of Duisburg-Essen, King’s College London, and University of Verona, etc. were contacted. Their opinion helps understand and validate current and emerging 22q11.2 deletion syndrome therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in 22q11.2 deletion syndrome.
22q11.2 Deletion Syndrome Report Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of 22q11.2 Deletion Syndrome, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the 22q11.2 Deletion Syndrome Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of 22q11.2 Deletion Syndrome, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the 22q11.2 Deletion Syndrome market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM 22q11.2 Deletion Syndrome market.
22q11.2 Deletion Syndrome Market Report Insights
- 22q11.2 Deletion Syndrome Patient Population Forecast
- 22q11.2 Deletion Syndrome Therapeutics Market Size
- 22q11.2 Deletion Syndrome Pipeline Analysis
- 22q11.2 Deletion Syndrome Market Size and Trends
- 22q11.2 Deletion Syndrome Market Opportunity (Current and forecasted)
22q11.2 Deletion Syndrome Market Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-Enabled Market Research Report
- 11-Year Forecast
- 22q11.2 Deletion Syndrome Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (By Geography)
- 22q11.2 Deletion Syndrome Treatment Addressable Market (TAM)
- 22q11.2 Deletion Syndrome Competitive Landscape
- 22q11.2 Deletion Syndrome Major Companies Insights
- 22q11.2 Deletion Syndrome Price Trends and Analogue Assessment
- 22q11.2 Deletion Syndrome Therapies Drug Adoption/Uptake
- 22q11.2 Deletion Syndrome Therapies Peak Patient Share Analysis
22q11.2 Deletion Syndrome Market Report Assessment
- 22q11.2 Deletion Syndrome Current Treatment Practices
- 22q11.2 Deletion Syndrome Unmet Needs
- 22q11.2 Deletion Syndrome Clinical Development Analysis
- 22q11.2 Deletion Syndrome Emerging Drugs Product Profiles
- 22q11.2 Deletion Syndrome Market attractiveness
- 22q11.2 Deletion Syndrome Qualitative Analysis (SWOT and Conjoint analysis)
- 22q11.2 Deletion Syndrome Market Drivers
- 22q11.2 Deletion Syndrome Market Barriers
FAQs Related to 22q11.2 Deletion Syndrome Market Report:
22q11.2 Deletion Syndrome Market Insights
- What was the 22q11.2 Deletion Syndrome market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of 22q11.2 Deletion Syndrome?
- What are the disease risks, burdens, and unmet needs of 22q11.2 Deletion Syndrome? What will be the growth opportunities across the 7MM concerning the 22q11.2 Deletion Syndrome patient population?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of 22q11.2 Deletion Syndrome? What are the current guidelines for treating 22q11.2 Deletion Syndrome in the US, Europe, and Japan?