Agitation in Alzheimer’s Disease Market Summary
Agitation in Alzheimer's Disease Insights and Trends
- Increasing recognition of agitation in Alzheimer’s disease across clinical settings is being supported by wider use of standardized behavioral assessment tools and greater reliance on caregiver-reported observations, improving identification and differentiation from other neuropsychiatric symptoms.
- According to DelveInsight’s analysis, the agitation in Alzheimer’s disease market across the 7MM surpassed USD 600 million in 2025, reflecting a growing clinical and commercial focus on addressing this high-burden neuropsychiatric manifestation within the broader Alzheimer’s disease landscape.
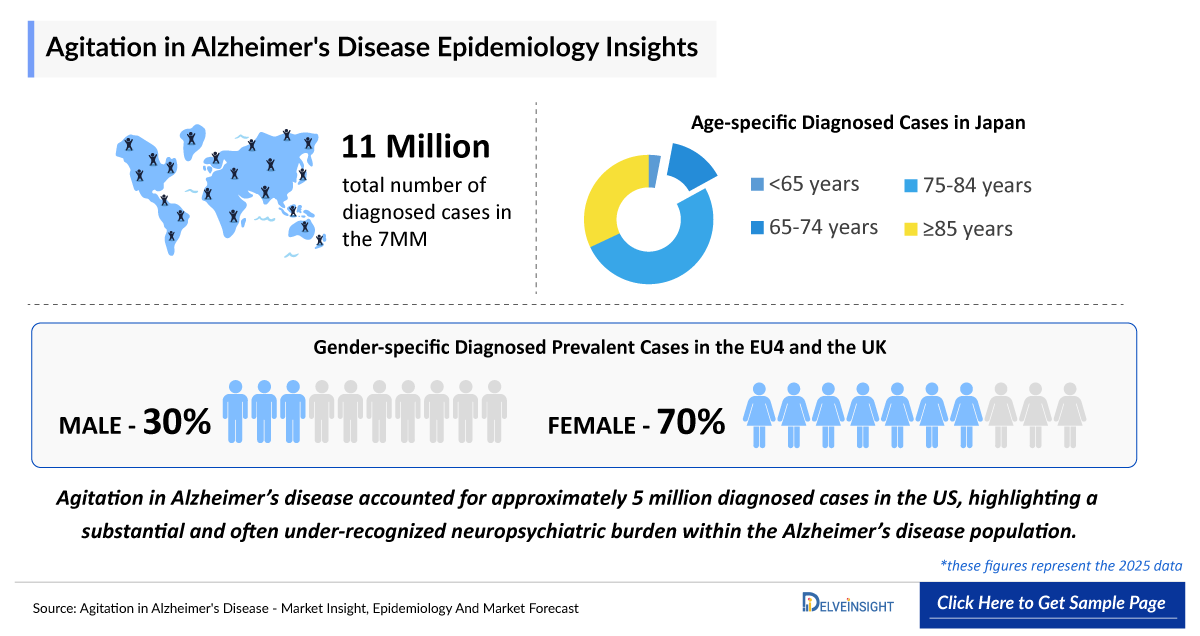
- Agitation in Alzheimer’s disease accounted for approximately 5 million diagnosed cases in the US, highlighting a substantial and often under-recognized neuropsychiatric burden within the Alzheimer’s disease population.
- In 2025, the total number of diagnosed cases of agitation in Alzheimer's disease in the 7MM was around 11 million. Among the 7MM countries, the US accounted for the highest number of diagnosed prevalent cases of Alzheimer’s disease, contributing around 49%, while the UK reported the lowest number of cases in 2025.
- Brexpiprazole (REXULTI) became the first pharmacological treatment approved in the US specifically for agitation associated with Alzheimer’s dementia, addressing a significant unmet need in behavioral symptom management. Clinical data from randomized Phase III studies demonstrated meaningful reductions in agitation severity compared with placebo, supporting its regulatory approval.
- The emergence of pipeline therapies such as masupirdine (SUVN-502), AUVELITY (AXS-05), EXV-801 ± dextromethorphan, melatonin/tetrahydrocannabinol (IGC-AD1), and xanomeline/trospium chloride (COBENFY) is reshaping the agitation in Alzheimer’s disease landscape by targeting diverse mechanisms. These innovations are expected to expand therapeutic options and drive continued market evolution.
- The overall therapeutic strategy focuses on identifying underlying triggers, reducing patient distress, and ensuring safety while balancing potential benefits and risks of different interventions. Given the heterogeneity of symptoms and contributing factors, management typically involves a combination of non-pharmacological and pharmacological approaches, with a strong emphasis on patient-centered care and caregiver involvement. Continued reliance on off-label antipsychotic therapies, which offer modest efficacy and are associated with significant safety concerns, limits optimal long-term outcomes in this vulnerable population.
- Pharmacological management of agitation in Alzheimer’s disease remains constrained by a narrow therapeutic margin, where modest efficacy is frequently offset by clinically meaningful safety risks. The continued reliance on off-label antipsychotics reflects a structural gap.
Agitation in Alzheimer's Disease Market size and forecast
- 2025 Agitation in Alzheimer's Disease Market Size in the 7MM: USD 600 million
- 2036 Projected Agitation in Alzheimer's Disease Market Size in the 7MM: USD 2,800 million
- Agitation in Alzheimer's Disease Growth Rate (2026–2036) in the 7MM: 14.9% CAGR
DelveInsight's ‘Agitation in Alzheimer's disease – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of agitation in Alzheimer’s Disease, historical and forecasted epidemiology, as well as the agitation in Alzheimer’s Disease market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The agitation in Alzheimer's disease market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, agitation in Alzheimer’s Disease patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key unmet medical needs in agitation in Alzheimer’s disease and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Geography Covered
- North America: The US
- Europe: Germany, France, Italy, Spain and the UK
- Asia-Pacific: Japan
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Agitation in Alzheimer's Disease Market CAGR (Forecast period) |
14.9% (2026-2036) |
|
Agitation in Alzheimer's Disease Epidemiology Segmentation Analysis |
Patient Burden Assesment
|
|
Agitation in Alzheimer’s Disease Companies |
|
|
Agitation in Alzheimer’s Disease Therapy |
Brexpiprazole (REXULTI) |
|
Agitation in Alzheimer’s Disease Market |
Segmented by
|
|
Analysis |
|
Agitation in Alzheimer's Disease Understanding and Treatment Algorithm
Agitation in Alzheimer's Disease Overview and Diagnosis
Agitation in Alzheimer’s disease is a common neuropsychiatric manifestation characterized by emotional distress, excessive motor activity, irritability, and, at times, aggressive behaviors, arising from progressive neurodegeneration affecting cognitive and behavioral regulation. The presentation varies across disease stages but often includes restlessness, verbal or physical aggression, mood lability, and resistance to care, significantly impacting patient quality of life and caregiver burden.
Diagnosis is primarily clinical, based on patient history, caregiver reports, and standardized assessment scales, as no definitive biomarkers are currently established. Evaluation focuses on identifying underlying triggers such as environmental stressors, unmet needs, or comorbid conditions. Non-pharmacological approaches, including behavioral and environmental interventions, remain foundational, while pharmacological options are considered in more severe cases despite limitations in efficacy and safety. Ongoing research into targeted therapies and improved assessment tools continues to shape a more structured and patient-centric approach to care.
Further details are provided in the report.
Current Agitation in Alzheimer's Disease Treatment Landscape
Approaches for agitation in alzheimer’s disease focus on reducing symptom severity and improving patient stability through a combination of non-pharmacological and pharmacological strategies. Behavioral and environmental interventions are considered foundational, including structured routines, caregiver education, and minimizing triggers such as noise or unfamiliar settings. In cases of moderate to severe symptoms, pharmacological options such as antipsychotics, antidepressants, and emerging agents targeting serotonergic and cholinergic pathways are utilized, although their use is often limited by variable efficacy and safety concerns. The evolving pipeline, including agents with novel mechanisms, reflects a shift toward more targeted and tolerable options aimed at addressing the complex neurobiology underlying agitation.
Further details related to country-based variations are provided in the report.
Agitation in Alzheimer's Disease Unmet Needs
The section “unmet needs of agitation in Alzheimer’s disease” highlights the key gaps between current clinical practices and the need for safe, effective, and targeted approaches to address this complex neuropsychiatric manifestation. It underscores the challenges faced by patients, caregivers, and clinicians, while pointing toward areas requiring innovation and research focus.
- Lack of standardized assessment tools
- Limited efficacy and safety concerns with existing pharmacological options
- Structural and system-level barriers undermine scalable implementation of nonpharmacological strategies
- Methodological heterogeneity continues to limit reproducibility and interpretability of clinical evidence and others.
Note: Comprehensive unmet needs insights in agitation in Alzheimer's disease and their strategic implications are provided in the full report.
Agitation in Alzheimer's Disease Epidemiology
Key Findings from Agitation in Alzheimer's Disease Epidemiological Analysis and Forecast
- The total number of diagnosed cases of agitation in alzheimer’s in the 7MM is ~11 million in 2025.
- According to DelveInsight’s estimates, in the United States, diagnosed cases of agitation in Alzheimer ’s disease are predominantly made up of females, accounting for ~ 3 million cases in 2025.
- In Japan, the age-specific diagnosed cases of agitation in Alzheimer ’s disease is of 3% for ages <65 years, 14% for 65–74 years, 51% for 75–84 years, and 32% for ≥85 years.
- Diagnosed cases of agitation in Alzheimer’s disease across EU4 and the UK were predominantly female, accounting for approximately 2 million cases in 2025, with a gender distribution of about 30% male and 70% female.
Agitation in Alzheimer's Disease Drug Analysis & Competitive Landscape
The Agitation in Alzheimer's Disease drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I-III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the agitation in alzheimer's disease treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the agitation in alzheimer's disease therapeutics market.
Approved Therapies for Agitation in Alzheimer's Disease
Brexpiprazole (REXULTI); Otsuka Pharmaceutical/Lundbeck: Brexpiprazole, discovered by Otsuka and co-developed with Lundbeck is the first approved drug for managing agitation associated with dementia due to Alzheimer’s disease. It operates through a mechanism of serotonin–dopamine activity modulation and its efficacy is believed to stem from partial agonist activity at serotonin 5-HT1A and dopamine D2 receptors, alongside antagonist effects on serotonin 5-HT2A receptors.
In September 2024, Otsuka reported that brexpiprazole (REXULTI) received regulatory approval in Japan for an additional indication: treating “an excessive motor activity or physically/verbally aggressive behavior” linked to mood changes and irritability in Alzheimer’s disease patients. This indication aligns with the US and other countries’ approval for agitation symptoms associated with dementia due to Alzheimer’s disease.
|
Agitation in Alzheimer's Disease Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Brexpiprazole (REXULTI) |
Otsuka Pharmaceutical/Lundbeck |
Agitation in Alzheimer’s-related dementia |
Small molecule |
Serotonin–dopamine modulation |
Oral |
US: 2023; JP: 2024 |
|
Drug B |
XX |
XX |
XX |
XX |
XX |
XX |
Agitation in Alzheimer's Disease Pipeline Analysis
Melatonin/tetrahydrocannabinol (IGC-AD1): IGC Pharma
IGC-AD1 (melatonin/tetrahydrocannabinol) is an investigational combination therapy developed by IGC Pharma for agitation associated with Alzheimer's disease. It combines THC and melatonin to modulate neuroinflammation, neuronal signaling, and circadian rhythms, aiming to reduce agitation and improve behavioral symptoms in patients with limited treatment options.
In February 2026, IGC Pharma reported that approximately 70% of the planned enrollment in its ongoing randomized, double-blind, placebo-controlled Phase II CALMA trial evaluating IGC-AD1 for agitation associated with Alzheimer's disease has been completed.
|
Competitive Landscape of Pipeline Drugs | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US | |
|
Masupirdine (SUVN-502) |
Suven Life Sciences |
III |
Agitation in dementia of the Alzheimer’s type. |
Oral |
5-HT6 antagonist |
2028 | |
|
AUVELITY (Bupropion/Dextromethorphan; AXS-05) |
Axsome Therapeutics |
III |
Alzheimer's disease agitation. |
Oral |
NMDA receptor antagonist |
Information is available in the full report | |
|
COBENFY (KarXT; xanomeline–trospium chloride) |
Bristol Myers Squibb/Karuna Therapeutics |
III |
Agitation in Alzheimer's disease. |
Oral |
Muscarinic M1/M4 receptor agonists. |
Information is available in the full report | |
|
EXV-801 ± Dextromethorphan |
Exciva GmbH |
II/III |
Agitation in Alzheimer's disease dementia. |
Oral |
Modulates NMDA and sigma-1 receptors |
Information is available in the full report | |
|
Drug E |
XX |
XX |
XX |
XX |
XX |
XX | |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Agitation in Alzheimer's Disease Key Players, Market Leaders and Emerging Companies
- Otsuka Pharmaceutical/Lundbeck
- Suven Life Sciences
- Axsome Therapeutics
- Bristol Myers Squibb/Karuna Therapeutics
- Exciva GmbH and others
Agitation in Alzheimer's Disease Drug Updates
- In November 2025, Suven Life Sciences reported that its global Phase III clinical trial of masupirdine (SUVN-502) achieved 50% patient enrollment for agitation in Alzheimer's disease, with the multicenter, randomized, double-blind, placebo-controlled study ongoing across North America and Europe
- In April 2025, Axsome Therapeutics presented positive pivotal Phase III results from the ACCORD-2 trial evaluating AXS-05 for the treatment of agitation associated with Alzheimer’s disease at the 2025 American Academy of Neurology (AAN) Annual Meeting.
- In November 2024, Exciva GmbH reported that its lead candidate EXV-802 received FTD from the US FDA for the treatment of agitation associated with Alzheimer’s disease.
Agitation in Alzheimer's Disease Market Outlook
The treatment landscape for agitation in alzheimer’s disease remains highly heterogeneous and setting-dependent, with outcomes influenced as much by caregiver environment, comorbidity burden, and clinician preference as by underlying disease severity. Despite evolving evidence, care continues to rely heavily on off-label antipsychotic use, careful dose titration, and close safety monitoring, resulting in substantial variability in treatment patterns and patient outcomes across regions. These limitations are driving a strategic shift toward more targeted and better-tolerated therapies, improved behavioral assessment frameworks, and precision-based approaches aimed at standardizing care, minimizing safety burden, and achieving more consistent and durable symptom control.
Key marketed therapies shaping current management
Brexpiprazole (REXULTI): An atypical antipsychotic approved in multiple regions for the treatment of agitation associated with Alzheimer’s disease, brexpiprazole delivers clinically meaningful symptom improvement through balanced serotonin–dopamine modulation. Its comparatively favorable tolerability profile supports use in a vulnerable elderly population, offering a more refined therapeutic option in an area historically limited by safety and efficacy constraints.
The emergence of the first approved therapy marks an initial shift in the management of agitation in Alzheimer’s disease from broad, off-label symptom control toward a more targeted, indication-specific approach, while ongoing innovation continues to explore mechanism-driven strategies to improve durability and safety of clinical outcomes.
- Overall, improved testing, and rising disease awareness are expected to drive steady growth in the 7MM Agitation in Alzheimer’s Disease market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- Among the 7MM, the US accounted for the largest market size of agitation in alzheimer's disease i.e., USD ~ 500 million in 2025.
- Among EU4 and the UK, Germany generated the highest revenue, exceeding USD 20 million in 2025, followed by France, with this figure expected to increase further by 2036.
- The agitation in Alzheimer’s disease market in Japan was estimated at approximately USD 50 million in 2025, representing around 9% of the 7MM agitation in Alzheimer’s disease market.
- In 2036, among all the therapies for agitation in alzheimer's disease, the highest revenue is estimated to be generated by COBENFY in the 7MM.
- Real-world treatment patterns for agitation in Alzheimer’s disease reflect a highly individualized and pragmatic approach, balancing symptomatic control with tolerability and safety risks. Atypical antipsychotics remain the most frequently prescribed agents (55.5% overall; 56.7% community; 47.7% LTC), followed by SSRIs (29.3%; 29.6%; 27.1%) and benzodiazepines (23.2%; 21.8%; 32.7%), indicating a tiered, symptom-driven strategy.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Agitation in Alzheimer’s Disease (2022–2036 Forecast)
Agents such as quetiapine, risperidone, olanzapine, aripiprazole, lurasidone, and ziprasidone are commonly utilized off-label due to their relatively favorable tolerability profiles compared to older agents. They are often selected based on patient-specific factors, though clinical responses remain variable and not consistently sustained.
Haloperidol continues to be used in certain acute or severe agitation scenarios due to its rapid onset of action. However, its use is generally limited by a higher risk of extrapyramidal symptoms and other adverse effects, particularly in elderly populations.
Clozapine and pimavanserin represent alternative off-label options explored in select cases, particularly where standard agents are ineffective or poorly tolerated. Their use remains limited due to monitoring requirements, safety considerations, and relatively limited evidence in this setting.
Despite widespread use, off-label antipsychotic therapies are associated with increased risks, including sedation, metabolic effects, cardiovascular events, and mortality. These limitations highlight the ongoing need for safer, more targeted, and evidence-based pharmacological alternatives for managing agitation in Alzheimer’s disease.
Agitation in Alzheimer’s Disease Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the agitation in Alzheimer’s disease drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Masupirdine (SUVN-502) : Suven Life Sciences
A selective 5-HT6 receptor antagonist, masupirdine, is designed to modulate serotonergic pathways implicated in neuropsychiatric symptoms of Alzheimer’s disease. It reflects a shift toward non-dopaminergic mechanisms for agitation, aiming to deliver symptomatic stabilization with a potentially improved tolerability profile compared to conventional antipsychotics.
Masupirdine (SUVN-502) is currently in a global Phase III trial for agitation in Alzheimer's disease dementia, with expected completion by the end of 2026 and topline data anticipated in Q1/Q2 2027.
AUVELITY (AXS-05) : Axsome Therapeutics
A multimodal NMDA receptor antagonist and sigma-1 receptor agonist combination in Phase III, AUVELITY targets glutamatergic and neuroplasticity pathways associated with behavioral disturbances. Its development highlights growing interest in rapid-acting, mechanism-driven CNS therapies that may address agitation alongside broader neuropsychiatric symptom domains.
Axsome Therapeutics reported that the US FDA accepted its sNDA for AUVELITY for the treatment of agitation associated with Alzheimer’s disease and granted the application Priority Review designation, with a PDUFA target action date set for April 30, 2026.
EXV-801 ± Dextromethorphan : Exciva GmbH
A combination neuroactive therapeutic platform in Phase II/III, EXV-802, with or without EXV-801, is being explored for its potential to modulate multiple CNS signaling pathways linked to agitation. This approach exemplifies the trend toward combinatorial or multi-target strategies aimed at enhancing efficacy while maintaining functional tolerability in complex behavioral syndromes.
In November 2024, Exciva GmbH reported that its lead candidate EXV-802 received FTD from the US FDA for the treatment of agitation associated with Alzheimer’s disease.
Detailed insights of emerging therapies' drug uptake is included in the report…
Market Access and Reimbursement of Approved therapies in Agitation in Alzheimer's Disease
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
The United States
|
The US Reimbursement of Therapies Approved for Agitation in Alzheimer's Disease | |
|
Drug/Therapy |
Access Program |
|
Brexpiprazole (REXULTI) |
State Health Insurance Assistance Program (SHIP) |
Agitation in Alzheimer's Disease therapies Price Scenario & Trends
Pricing and analogue assessment of agitation in alzheimer's disease therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of agitation in alzheimer's disease approved drugs
A structured secondary analysis was undertaken to contextualize the pricing of brexpiprazole (REXULTI) by integrating dosing assumptions and treatment duration into a real-world cost framework. With a US WAC of approximately USD 1,515 and a typical treatment duration of up to 12 weeks, the total per-patient treatment cost is estimated at ~USD 4,500.
Industry Experts and Physician Views for Agitation in Alzheimer's Disease
To keep up with agitation in alzheimer's disease market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the agitation in alzheimer's disease emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in agitation in alzheimer's disease, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights at country level. Centers such as the The University of Texas Health Science Center, Gerontological Society of America, German Center for Neurodegenerative Diseases, University Hospital of Toulouse etc. were contacted.
Their opinion helps understand and validate current and emerging agitation in alzheimer's disease, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in agitation in alzheimer's disease.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Agitation in Alzheimer’s disease and related dementias is increasingly recognized as a significant neurobehavioral burden with substantial caregiver impact. Growing evidence suggests that agitation is closely linked to disease severity and reflects underlying frontal lobe dysfunction, which may amplify behavioral responses to coexisting psychopathology and environmental triggers, positioning it as a distinct frontal lobe-driven syndrome within the broader Alzheimer’s disease spectrum.” |
|
Germany |
“Emerging evidence highlights a neurobiological basis for agitation in Alzheimer’s disease, implicating early tau-related changes in the locus coeruleus and dysregulation of affective executive networks. These findings suggest compensatory noradrenergic overactivity and emotion regulation deficits may differentially influence agitation risk and severity, warranting further investigation.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of agitation in alzheimer's disease, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.Attribute analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of agitation in Alzheimer’s Disease, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the agitation in Alzheimer’s Disease market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM agitation in Alzheimer’s Disease market.
Report Insights
- Agitation in Alzheimer’s Disease Patient Population Forecast
- Agitation in Alzheimer’s Disease Therapeutics Market Size
- Agitation in Alzheimer’s Disease Pipeline Analysis
- Agitation in Alzheimer’s Disease Market Size and Trends
- Agitation in Alzheimer’s Disease Market Opportunity (Current and forecasted)
Report Key Strengths
- Epidemiology‑Based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- Agitation in Alzheimer’s Disease Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Agitation in Alzheimer’s Disease Treatment Addressable Market (TAM)
- Agitation in Alzheimer’s Disease Competitive Landscape
- Agitation in Alzheimer’s Disease Major Companies Insights
- Agitation in Alzheimer’s Disease Price trends and Analogue Assessment
- Agitation in Alzheimer’s Disease Therapies Drug Adoption/Uptake
- Agitation in Alzheimer’s Disease Therapies Peak Patient Share analysis
Report Assessment
- Agitation in Alzheimer’s Disease Current Treatment Practices
- Agitation in Alzheimer’s Disease Unmet Needs
- Agitation in Alzheimer’s Disease Clinical Development Analysis
- Agitation in Alzheimer’s Disease Emerging Drugs Product Profiles
- Agitation in Alzheimer’s Disease Market Attractiveness
- Agitation in Alzheimer’s Disease Qualitative Analysis (SWOT and conjoint analysis)
FAQs
Market Insights
- What was the agitation in Alzheimer’s disease market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of agitation in Alzheimer ’s disease?
- What are the disease risks, burdens, and unmet needs of agitation in Alzheimer ’s disease? What will be the growth opportunities across the 7MM concerning the patient population with agitation in Alzheimer ’s disease?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of agitation in Alzheimer ’s disease? What are the current guidelines for treating Agitation in Alzheimer ’s disease in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the agitation in Alzheimer ’s disease market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.