Alcohol-associated Liver Disease (ALD) Market
Alcohol-associated Liver Disease (ALD) Insights and Trends
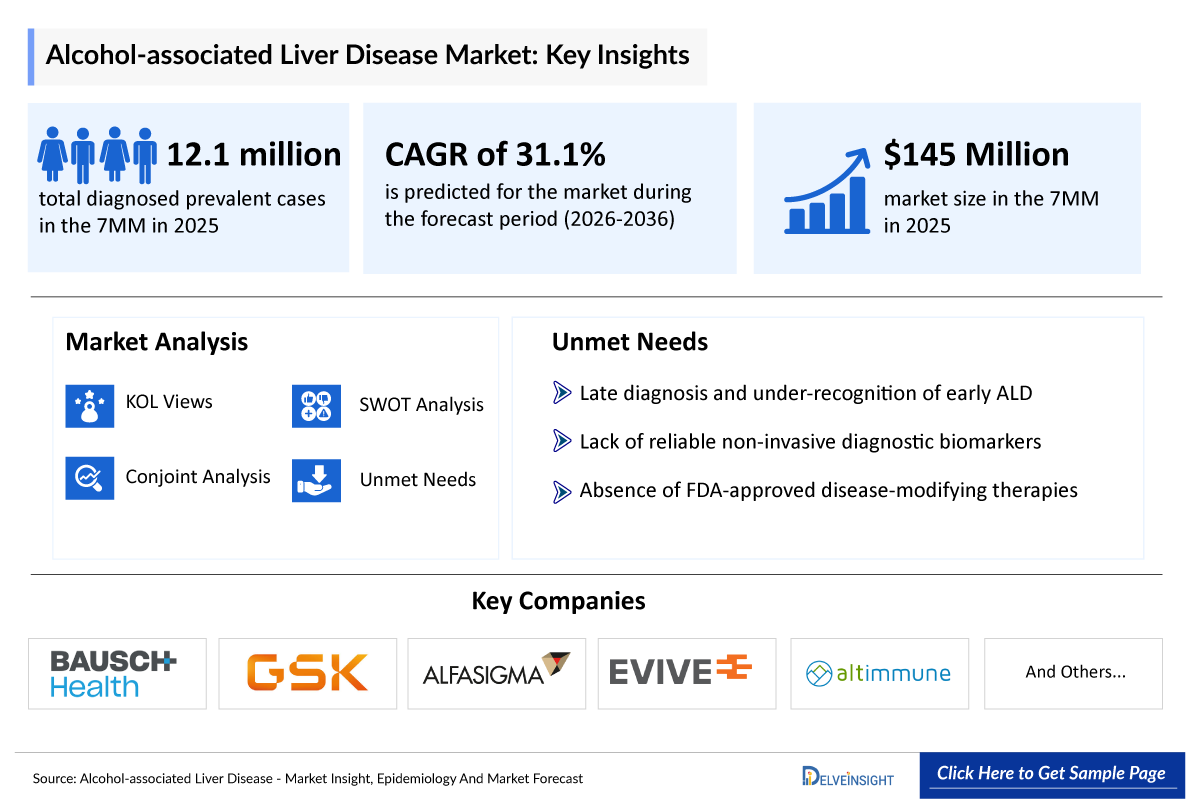
- According to DelveInsight’s analysis, ALD market size was found to be aound USD 145 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- ALD is a chronic, progressive spectrum of liver disorders caused by sustained excessive alcohol consumption, resulting in metabolic dysregulation, inflammation, fibrosis, and irreversible structural liver damage. It is one of the leading causes of liver-related morbidity and mortality globally and accounts for approximately half of cirrhosis cases in the United States. ALD significantly impairs survival, quality of life, and healthcare systems due to frequent hospitalizations, complications, and limited therapeutic options.
- According to the World Health Organization, alcohol consumption accounted for an estimated 2.6 million deaths globally in 2019, with ALD representing a significant contributor to this burden.
- Higher prevalence of ALD among Asian populations, as well as in individuals with higher education levels and daily smoking habits, indicates that risk is shaped by a complex interplay of genetic, cultural, and behavioral factors beyond alcohol exposure alone.
- Management of ALD remains largely supportive and stage-dependent, with sustained alcohol abstinence representing the most effective intervention across all disease stages. Corticosteroids are commonly used in selected patients with severe alcohol-associated hepatitis, though benefits are inconsistent and limited to short-term survival. Nutritional rehabilitation, complication management, and multidisciplinary care are central to improving outcomes, while liver transplantation remains the only definitive option for eligible patients with end-stage disease.
- Despite its substantial clinical burden, ALD lacks FDA-approved disease-modifying therapies. Current care strategies focus on symptom control and complication management rather than targeting underlying disease biology.
- For patients with advanced ALD who are unresponsive to medical therapy, liver transplantation remains the only intervention capable of significantly improving prognosis. More recently, acute alcoholic hepatitis has emerged as a potential early indication for transplantation in carefully selected non-responders, with growing evidence demonstrating favorable outcomes in this high-risk population.
- Several investigational therapies are advancing through early- to mid-stage development and are expected to reshape the ALD treatment landscape (2026–2036), including larsucosterol (Bausch Health), GSK4532990 (GSK), INT-787 (Alphasigma [Intercept Pharmaceuticals]), pemvidutide (Altimmune), and NTR-101 (Nterica), with a focus on targeting metabolic dysfunction, inflammation, fibrosis, and improving long-term outcomes beyond supportive care and alcohol abstinence.
Alcohol-associated Liver Disease (ALD) Market Size and Forecast in the 7MM
- 2025 Alcohol-associated Liver Disease (ALD) Market Size: ~USD 145 million
- 2036 Projected Alcohol-associated Liver Disease (ALD) Market Size: ~USD 2,200 million
- Alcohol-associated Liver Disease (ALD) Growth Rate (2026–2036): 31.1 % CAGR
DelveInsight's ‘Alcohol-associated Liver Disease (ALD) – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Alcohol-associated Liver Disease (ALD), historical and forecasted epidemiology, as well as the Alcohol-associated Liver Disease (ALD) market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Alcohol-associated Liver Disease (ALD) market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Alcohol-associated Liver Disease (ALD) patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Alcohol-associated Liver Disease (ALD) and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
ALD Market CAGR (Forecast period) |
31.1% (2026–2036) |
|
ALD Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
ALD Companies |
|
|
ALD Therapies |
|
|
ALD Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Alcohol-associated Liver Disease (ALD) Market
Rising global alcohol consumption and shifting drinking patterns
Increasing alcohol intake, particularly among men, continues to drive the overall burden of ALD; however, a notable rise in alcohol consumption among women, who may have greater biological susceptibility to liver injury, is contributing to a disproportionately faster increase in ALD risk, thereby amplifying the need for targeted and scalable therapeutic interventions across genders.
Significant overlap between ALD, obesity, and metabolic dysfunction
The intersection of ALD with metabolic disorders such as obesity and insulin resistance is reshaping disease understanding, creating opportunities for metabolic-targeted therapies (e.g., GLP-1–based agents) and combination approaches.
Emergence of microbiome-based and precision medicine approaches
The pipeline is gradually evolving toward targeted, biology-driven interventions addressing inflammation, fibrosis, and metabolic dysregulation (e.g., larsucosterol, INT-787, pemvidutide), with growing interest in precision medicine and novel modalities to improve outcomes in this high-unmet-need population.
Alcohol-associated Liver Disease (ALD) Understanding and Treatment Algorithm
Alcohol-associated Liver Disease (ALD) Overview and Diagnosis
ALD is a progressive spectrum of chronic liver injury driven by prolonged excessive alcohol intake and remains a leading cause of liver-related morbidity and mortality globally. It ranges from early-stage steatosis, characterized by fat accumulation in hepatocytes, to more severe conditions such as alcohol-associated steatohepatitis (ASH), fibrosis, cirrhosis, and hepatocellular carcinoma (HCC), with some patients developing acute alcohol-associated hepatitis marked by high short-term mortality. Pathophysiologically, alcohol metabolism in the liver disrupts metabolic homeostasis, inducing oxidative stress, lipid dysregulation, inflammation, and hepatocellular damage. While steatosis occurs in most heavy drinkers, only a subset progress to advanced disease, reflecting the complex interplay of metabolic, inflammatory, and fibrotic pathways underlying ALD progression.
Diagnosis of alcohol-associated hepatitis is largely clinical and supported by laboratory findings such as elevated bilirubin levels, increased Aspartate Aminotransferase (AST), and a characteristic AST/ALT ratio >1.5. Severe forms of alcoholic hepatitis are associated with high short-term mortality and limited therapeutic options.
Further details are provided in the report.
Current Alcohol-associated Liver Disease (ALD) Treatment Landscape
Management of ALD remains largely supportive and multidisciplinary, as no FDA-approved disease-modifying therapies currently exist. The cornerstone of treatment is complete alcohol abstinence, which can reverse early-stage disease, slow or halt progression, and significantly improve survival; structured behavioral support and supervised care are critical to sustaining long-term abstinence. In patients with severe alcohol-associated hepatitis, corticosteroids such as prednisolone may be used to suppress inflammation, although benefits are variable and require early response assessment. Nutritional optimization is another key pillar, as malnutrition is common and contributes to poor outcomes, patients often require high-calorie, protein-rich diets along with targeted vitamin and micronutrient supplementation. For individuals with advanced or end-stage disease who do not respond to medical management, liver transplantation remains the only definitive treatment, offering substantial survival and quality-of-life benefits in carefully selected patients.
Further details related to country-based variations are provided in the report.
Alcohol-associated Liver Disease (ALD) Unmet Needs
The section “unmet needs of ALD” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Late diagnosis and under-recognition of early ALD
- Lack of reliable non-invasive diagnostic biomarkers
- Absence of FDA-approved disease-modifying therapies
- Variability and ethical challenges in liver transplant selection, and others…..
Note: Comprehensive unmet needs insights in ALD and their strategic implications are provided in the full report.
Alcohol-associated Liver Disease (ALD) Epidemiology
Key Findings from Alcohol-associated Liver Disease (ALD) Epidemiological Analysis and Forecast
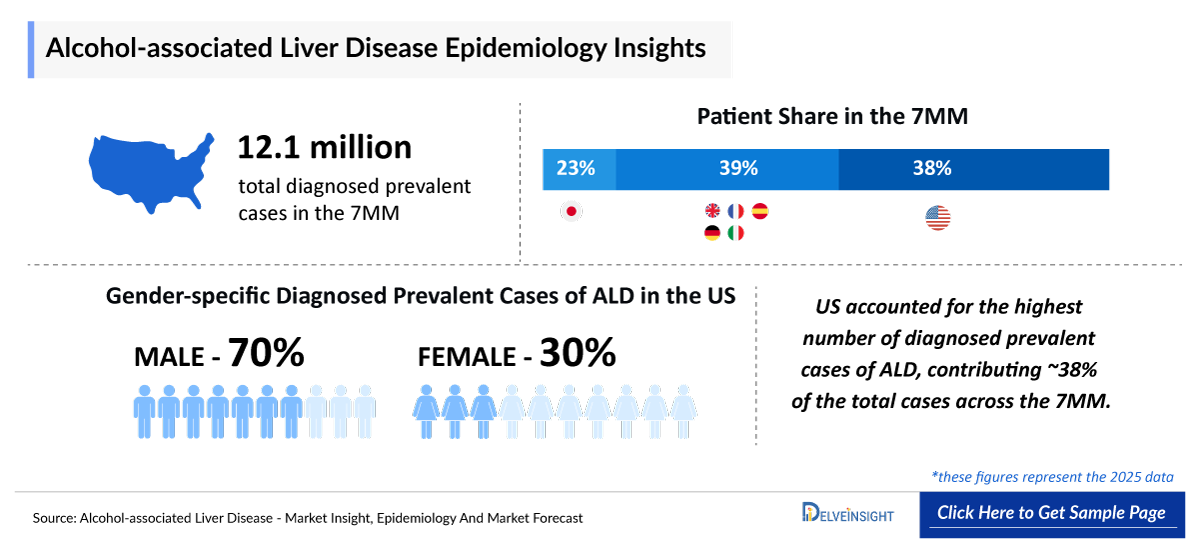
- According to DelveInsight’s estimates, in 2025, the total number of diagnosed prevalent cases of ALD in the 7MM were ~12.1 million.
- In the US, among stage-specific diagnosed prevalent cases of ALD, alcohol-associated steatosis accounted for the majority of cases (~3,214,800) in 2025, followed by alcohol-associated steatohepatitis (918,500 cases), and alcohol-associated cirrhosis (459,300 cases), in 2025.
- In Japan, males accounted the higher number of ALD cases, with approximately 2.26 million cases as compared to females.
- Stage-specific analysis of ALD reveals a predominance of patients in early disease stages, particularly alcohol-associated steatosis, with progressively fewer individuals advancing to steatohepatitis and cirrhosis. This distribution reflects the natural, gradual progression of ALD, in which only a subset of patients develop severe, irreversible liver damage. The substantial early-stage population underscores a critical window for intervention, whereas the smaller cohort with advanced disease drives a disproportionate share of morbidity, mortality, and healthcare resource utilization.
Alcohol-associated Liver Disease (ALD) Drug Analysis & Competitive Landscape
The Alcohol-associated Liver Disease (ALD) drug chapter provides a detailed, market-focused review of the emerging pipeline across Phase I–III clinical trials. It covers mechanism of action, clinical trial data, patents, collaborations, and strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the ALD treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the ALD therapeutics market.
Alcohol-associated Liver Disease (ALD) Pipeline Analysis
Larsuscosterol (DUR-928): Bausch Health
Larsucosterol is an endogenous sulfated oxysterol and epigenetic modulator that inhibits DNA methyltransferases (DNMT1, DNMT3a, DNMT3b), reducing pathological hypermethylation in ALD. By modulating pathways involved in inflammation, lipid metabolism, and cellular stress, it aims to limit hepatocyte injury and support liver recovery in alcoholic hepatitis.
The US FDA has granted larsucosterol Fast Track and Breakthrough Therapy designations, and a Phase III trial in severe alcoholic hepatitis was initiated in early 2026, with results expected in 2028; based on a projected primary completion in June 2028, it is assumed that the therapy could receive regulatory approval by early 2029 and be commercially launched in the later in 2029, positioning it as a potential first approved therapy for this condition.
|
Emerging Competitive Landscape of ALD | ||||||
|
Product |
Company |
Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Larsucosterol |
Bausch Health |
III |
Alcoholic hepatitis |
IV infusion |
DNA methyltransferase inhibitor |
2029 |
|
GSK4532990 |
GSK |
II |
ALD |
SC |
HSD17B13 inhibitors |
Information is available in the full report |
|
INT-787 |
Alphasigma (Intercept Pharmaceuticals) |
II |
Severe alcohol associated hepatitis |
Oral |
Farnesoid X receptor (FXR) agonist |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Alcohol-associated Liver Disease (ALD) Key Players, Market Leaders and Emerging Companies
- Bausch Health
- GSK
- Alphasigma (Intercept Pharmaceuticals)
- Evive Biotech
- Altimmune
- Nterica, and others
Alcohol-associated Liver Disease (ALD) Drug Updates
- In March 2026, Altimmune announced that the use of pemvidutide for treating ALD, including reducing liver fibrosis, is protected under a pending US provisional patent application, independent of the Mederis IP License Agreement. The company plans to file a non-provisional US application along with a corresponding international (PCT) filing, and if granted, the resulting patents are expected to extend protection until at least March 2047, subject to standard extensions and fee payments.
- In November 2025, GSK presented results from its advancing liver pipeline, including GSK4532990 for treatment of ALD, at the American Association for the Study of Liver Diseases (AASLD) The Liver Meeting 2025.
- In September 2025, Bausch Health Companies completed its tender offer to acquire DURECT Corporation, purchasing all outstanding shares for USD 1.75 per share in cash, totaling about USD 63 million upfront. The deal also includes up to USD 350 million in additional milestone payments tied to future net sales, payable upon achieving specified targets within the agreed timeframe.
Alcohol-associated Liver Disease (ALD) Market Outlook
ALD represents a progressive spectrum of liver injury driven by chronic alcohol exposure, advancing from alcohol-associated steatosis to alcoholic hepatitis and ultimately cirrhosis, with each stage linked to increasing morbidity and mortality. While early-stage disease is potentially reversible with sustained abstinence, progression to advanced stages is associated with poor clinical outcomes and high short-term mortality. Notably, there are no FDA-approved therapies specifically indicated for ALD, highlighting a critical gap in disease-modifying treatment options.
Current management remains largely supportive and stage-dependent, centered on sustained alcohol abstinence, nutritional therapy, and monitoring for complications such as hepatocellular carcinoma and varices. Corticosteroids are used selectively in severe alcoholic hepatitis, while liver transplantation remains the only definitive treatment for eligible patients with advanced disease.
The treatment landscape is gradually shifting toward mechanism-based approaches, with emerging candidates such as larsucosterol, GSK4532990, INT-787, pemvidutide, and NTR-101 targeting inflammation, fibrosis, and metabolic dysfunction. However, most therapies remain in early to mid-stage development, underscoring the significant unmet need in ALD.
Overall, the ALD market is expected to evolve steadily, driven by incremental innovation, improving disease awareness, and a growing focus on targeted, biology-driven therapies. The potential introduction of first disease-modifying treatments could reshape the treatment paradigm across 7MM ALD market from 2022–2036 and unlock meaningful clinical and commercial opportunities.
- Among the 7MM, the US accounted for the largest market size of ALD. i.e., USD ~85 million in 2025.
- In 2036, among all the therapies for ALD, the highest revenue is estimated to be generated by larsucosterol, in the US.
- The entry of mid-stage candidates such as GSK4532990, INT-787, and Pemvidutide are expected to intensify competition in the ALD treatment landscape during the latter half of the forecast period.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Alcohol-associated Liver Disease (ALD) (2022–2036 Forecast)
The ALD market comprises small molecules, small interfering RNA (siRNA), peptide-based therapies, and other emerging modalities, each targeting key pathways involved in disease progression such as inflammation, fibrosis, and metabolic dysfunction.
Small interfering RNA (siRNA): This emerging class aims to modulate gene expression linked to liver injury and fibrosis. While still in early stages for ALD, siRNA-based approaches such as GSK4532990 hold potential to target underlying disease drivers with high specificity and durability.
Small molecules: This class represents the core of the ALD pipeline, including agents such as larsucosterol and INT-787, which target pathways related to inflammation, bile acid signaling, fibrosis, and metabolic regulation. These therapies are designed to reduce hepatocellular injury and promote liver repair, addressing key unmet needs in disease modification.
Peptide-based therapies: therapies such as pemvidutide (a GLP-1/glucagon receptor agonist) reflect a growing focus on metabolic modulation and weight-related pathways, which may indirectly improve liver outcomes. Additional novel modalities are being explored to expand the therapeutic landscape.
Overall, small molecules currently dominate the innovation landscape, with most pipeline activity concentrated in this segment, while next-generation approaches such as siRNA and peptide therapies are expected to contribute to future diversification of ALD treatment strategies.
Alcohol-associated Liver Disease (ALD) Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the ALD drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The uptake of therapies in ALD is likely to vary depending on clinical positioning, mechanism of action, and maturity of clinical evidence. Emerging candidates such as larsucosterol may achieve relatively faster uptake, particularly if supported by strong efficacy data and potential first-in-class positioning, while GSK4532990 is expected to see moderate adoption as evidence and clinician confidence build over time.
In contrast, therapies like INT-787 may experience a more gradual uptake trajectory, with adoption contingent on demonstrated long-term efficacy, safety, and clear differentiation within the evolving treatment landscape. Overall, given the current lack of approved disease-modifying options, uptake dynamics are expected to remain measured but responsive to robust clinical outcomes.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Alcohol-associated Liver Disease (ALD)
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Alcohol-associated Liver Disease (ALD) Therapies Price Scenario & Trends
Pricing and analogue assessment of ALD therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Industry Experts and Physician Views for Alcohol-associated Liver Disease (ALD)
To keep up with ALD market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on ALD emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in ALD, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as University of Michigan Hospitals & Health Centers, University of Nebraska Medical Centre, Imperial College, and University of Würzburg, etc. were contacted. Their opinion helps understand and validate current and emerging ALD therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in ALD.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Alcoholic hepatitis demonstrates a broad and heterogeneous clinical presentation, with patients ranging from asymptomatic or mildly symptomatic disease to acute and severe hepatic decompensation, including liver failure.” |
|
Germany |
“Despite decades of research, the absence of FDA-approved therapies underscores a major translational gap in ALD, largely driven by the inability of current preclinical models to fully recapitulate severe human disease. Existing mouse models fail to mimic key pathological and immunological features (e.g., neutrophil biology, chemokine signaling), suggesting that next-generation models incorporating genetic modifications, dietary factors, and advanced tools like CRISPR-based liver editing are critical to better understand disease mechanisms and enable effective drug development.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Alcohol-associated Liver Disease (ALD), strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Alcohol-associated Liver Disease (ALD), explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Alcohol-associated Liver Disease (ALD) market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Alcohol-associated Liver Disease (ALD) market.
Report Insights
- Alcohol-associated Liver Disease (ALD) Patient Population Forecast
- Alcohol-associated Liver Disease (ALD) Therapeutics Market Size
- Alcohol-associated Liver Disease (ALD) Pipeline Analysis
- Alcohol-associated Liver Disease (ALD) Market Size and Trends
- Alcohol-associated Liver Disease (ALD) Market Opportunity (Current and forecasted)
Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-Enabled Market Research Report
- 11-Year Forecast
- Alcohol-associated Liver Disease (ALD) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (By Geography)
- Alcohol-associated Liver Disease (ALD) Treatment Addressable Market (TAM)
- Alcohol-associated Liver Disease (ALD) Competitve Landscape
- Alcohol-associated Liver Disease (ALD) Major Companies Insights
- Alcohol-associated Liver Disease (ALD) Price Trends and Analogue Assessment
- Alcohol-associated Liver Disease (ALD) Therapies Drug Adoption/Uptake
- Alcohol-associated Liver Disease (ALD) Therapies Peak Patient Share Analysis
Report Assessment
- Alcohol-associated Liver Disease (ALD) Current Treatment Practices
- Alcohol-associated Liver Disease (ALD) Unmet Needs
- Alcohol-associated Liver Disease (ALD) Clinical Development Analysis
- Alcohol-associated Liver Disease (ALD) Emerging Drugs Product Profiles
- Alcohol-associated Liver Disease (ALD) Market attractiveness
- Alcohol-associated Liver Disease (ALD) Qualitative Analysis (SWOT and Conjoint analysis)
FAQs
Market Insights
- What was the Alcohol-associated Liver Disease (ALD) market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Alcohol-associated Liver Disease (ALD)?
- What are the disease risks, burdens, and unmet needs of Alcohol-associated Liver Disease (ALD)? What will be the growth opportunities across the 7MM concerning the patient population with Alcohol-associated Liver Disease (ALD)?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Alcohol-associated Liver Disease (ALD)? What are the current guidelines for treating Alcohol-associated Liver Disease (ALD) in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Alcohol-associated Liver Disease (ALD) market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets withing the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.