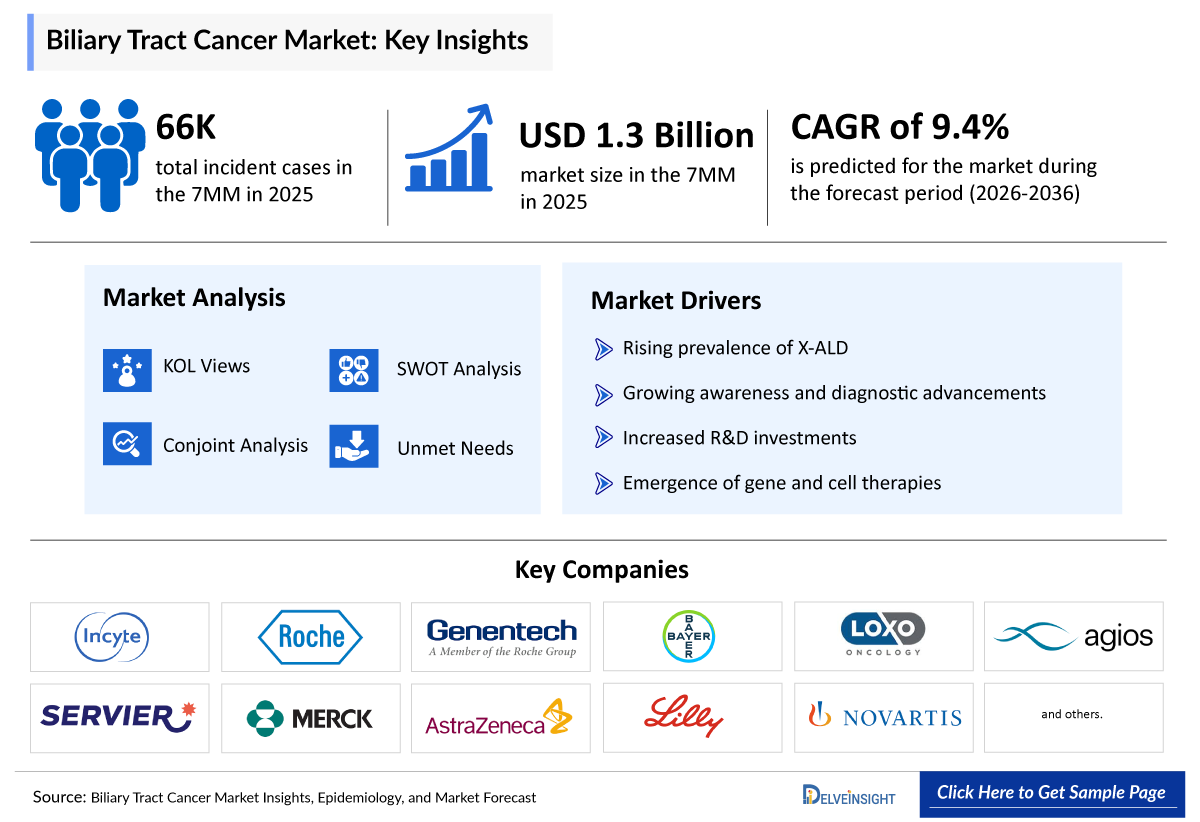
Biliary Tract Cancers Market Summary
- The Biliary Tract Cancer Market Size is anticipated to grow with a significant CAGR during the study period (2020-2034).
- The Biliary Tract Cancer Market companies developing therapies include - AstraZeneca, Merck & Co., Compass Therapeutics, NuCana, Senhwa Biosciences, TransThera Sciences, J-Pharma, Eisai, Seagen, Bold Therapeutics, Virogin Biotech, Replimune, Bayer, RedHill Biopharma, Relay Therapeutics, Elevar Therapeutics, Eli Lilly, Cogent Biosciences,and others.
Biliary Tract Cancer Market and Epidemiology Analysis
- In Western countries, intrahepatic cholangiocarcinoma (iCCA) has shown a rising incidence trend, while extrahepatic cholangiocarcinoma (eCCA) appears relatively stable or slight increasing, suggesting differences in etiologic drivers and diagnostic coding over time.
- Gall Bladder Cancer (GBC) incidence in the United States has generally been declining over the past two decades.
- In East Asian countries like Japan, the proportion of eCCA and GBC cases is higher.
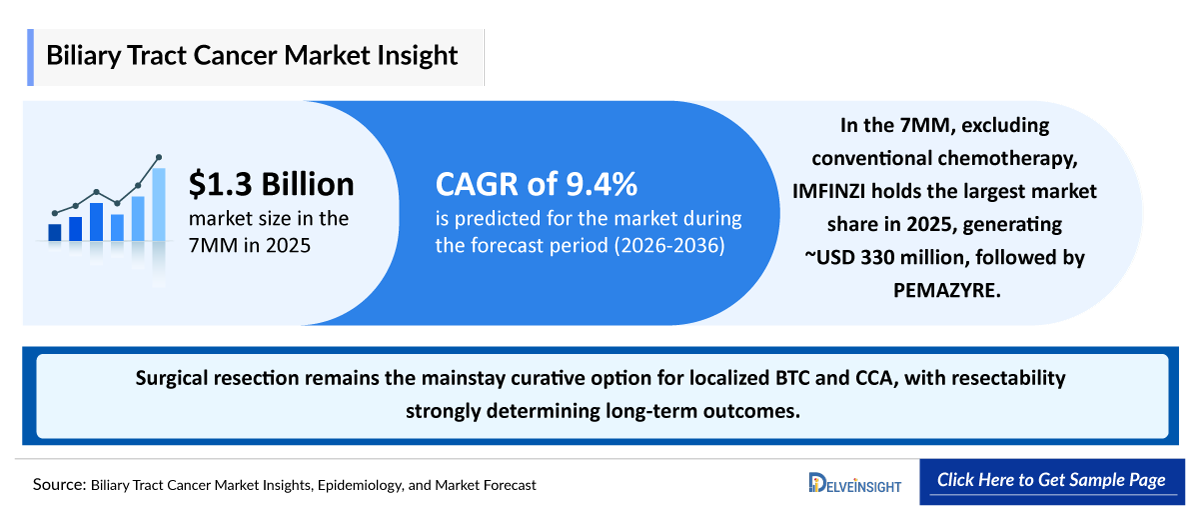
- Surgical resection remains the mainstay curative option for localized BTC and cholangiocarcinoma, with resectability strongly determining long-term outcomes. Patients with early-stage disease who are surgical candidates achieve significantly better prognoses than those with advanced disease.
- Platinum-based combination chemotherapy has historically served as the backbone of systemic therapy in advanced or unresectable disease across the 7MM.
- Recent regulatory approvals have expanded targeted treatment options for BTCs. FGFR inhibitors, such as pemigatinib (PEMAZYRE) and futibatinib (LYTGOBI) are approved for FGFR2-fusion-positive cholangiocarcinoma, while IDH1 inhibitors, ivosidenib (TIBSOVO), is approved for IDH1-mutated disease following prior therapy.
- Additionally, emerging assets include tinengotinib (TT-00420), rilvegostomig, nanvuranlat (JPH203), tovecimig, LENVIMA, TUKYSA, BOLD-100, and others.
- A critical unmet need in BTC is the absence of reliable, non-invasive biomarkers for early detection. Owing to its silent or non-specific presentation, BTC is often diagnosed at advanced stages. Existing markers, including CA19-9 and CEA, lack adequate sensitivity and specificity, and CA19-9 is not expressed in a subset of patients due to genetic variation, resulting in both false negatives and false positives, particularly in benign biliary conditions.
Biliary Tract Cancer Market and Epidemiology Analysis
Rising Incidence of Biliary Tract Cancer
The increasing global prevalence of cholangiocarcinoma and gallbladder cancer is a major growth driver. Factors such as chronic liver diseases, infections, obesity, and aging populations are contributing to a higher patient pool, thereby boosting demand for treatments.
Advancements in Targeted Therapy & Immunotherapy
The introduction of targeted therapies and immuno-oncology drugs is transforming treatment outcomes. These therapies address limitations of traditional chemotherapy and are expanding treatment options, significantly driving market expansion.
Growth in Precision Medicine & Molecular Diagnostics
Technologies such as next-generation sequencing (NGS), biomarker testing, and liquid biopsies are enabling personalized treatment approaches. This shift toward precision medicine is increasing adoption of advanced therapies and improving clinical outcomes.
Increasing Healthcare Infrastructure & Investment
Improved healthcare facilities, especially in emerging markets, along with rising government funding and R&D investments, are supporting better diagnosis and treatment access, thereby fueling market growth.
Growing Awareness and Early Diagnosis Initiatives
Enhanced awareness among healthcare professionals and patients is leading to earlier detection and timely treatment. Screening advancements and improved imaging technologies are also contributing to market growth.
Request a sample to unlock the CAGR for "Biliary Tract Cancer Market Forecast"
DelveInsight's ‘Biliary Tract Cancer (BTC) – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the BTC, historical and forecasted epidemiology, as well as the BTC market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The BTC market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, BTC patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in BTC and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2020 to 2034 |
|
Forecast Period |
2024-2034 |
|
Geographies Covered |
|
|
Biliary Tract Cancer Market |
|
|
Biliary Tract Cancer Market Size |
~USD XX Million in 2022 |
|
Biliary Tract Cancer Companies |
AstraZeneca, Merck & Co., Compass Therapeutics, NuCana, Senhwa Biosciences, TransThera Sciences, J-Pharma, Eisai, Seagen, Bold Therapeutics, Virogin Biotech, Replimune, Bayer, RedHill Biopharma, Relay Therapeutics, Elevar Therapeutics, Eli Lilly, Cogent Biosciences,and others. |
|
Biliary Tract Cancer Market Unmet Need |
The Biliary Tract Cancer market faces unmet needs including late-stage diagnosis, limited targeted therapies, poor prognosis, high recurrence rates, and lack of effective biomarkers for early detection and treatment selection. |
Biliary Tract Cancer Disease Understanding
Biliary Tract Cancer Overview
Biliary Tract Cancer refers to a group of rare but aggressive malignancies that arise in the bile ducts (cholangiocarcinoma) and gallbladder. These cancers are often diagnosed at advanced stages due to vague or absent early symptoms. Risk factors include chronic liver disease, bile duct inflammation, infections, and genetic predisposition. The prognosis remains poor, highlighting the need for improved detection and therapies.
Biliary Tract Cancer Diagnosis
Diagnosis of Biliary Tract Cancer involves a combination of imaging, laboratory tests, and biopsy confirmation. Techniques such as CT scans, MRI, and ultrasound are commonly used to detect abnormalities in the biliary system. Blood tests may include tumor markers like CA 19-9. Advanced diagnostic approaches, including endoscopic procedures and molecular testing, help in accurate staging and treatment planning.
Biliary Tract Cancer Treatment
Treatment of Biliary Tract Cancer depends on the stage and location of the tumor. Surgical resection offers the best chance for cure in early-stage disease. For advanced cases, chemotherapy (e.g., gemcitabine-based regimens), targeted therapies, and immunotherapy are commonly used. Recent advancements in precision medicine and novel drug development are improving patient outcomes and expanding therapeutic options.
Note: Comprehensive unmet needs insights in BTC and their strategic implications are provided in the full report.
Biliary Tract Cancer Market Epidemiology
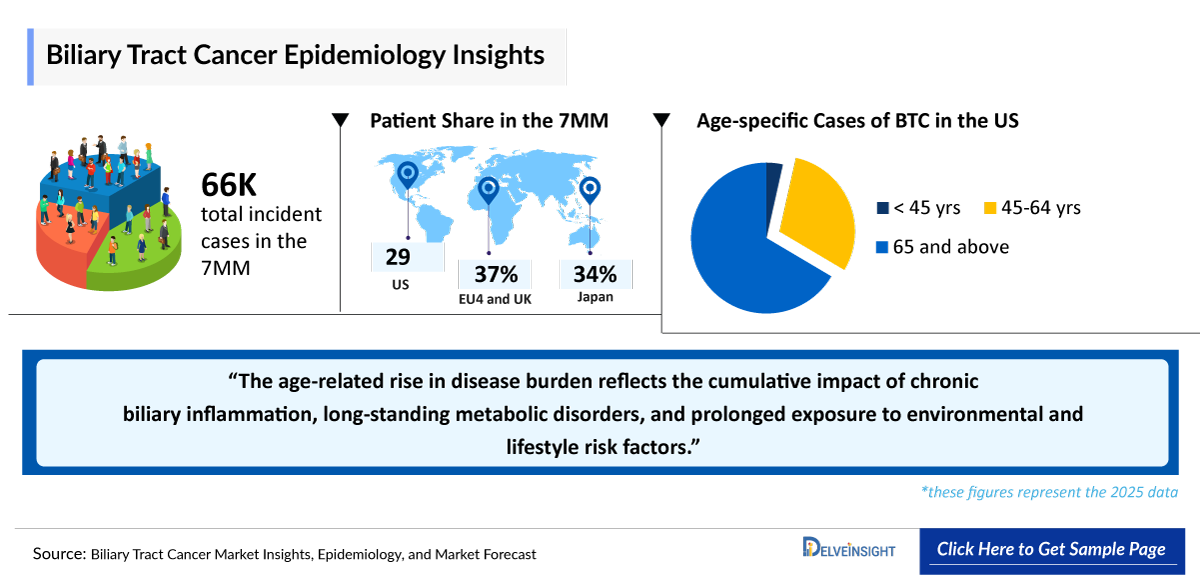
The epidemiology of Biliary Tract Cancer across the 7MM highlights its rarity but high mortality, with notable regional variations. Cholangiocarcinoma constitutes a major share of cases. Japan reports the highest incidence, while Western markets show lower burden. Incidence is higher in older populations, particularly those aged 65+, reflecting demographic and risk factor differences.
Key Findings from Biliary Tract Cancer Epidemiological Analyses and Forecast
- In the 7MM, Japan recorded the highest number of Incident cases of BTC accounting for ~22,000 cases in 2025, which are expected to continue increasing at a CAGR of 1.2%
- Among the EU4 and the UK, Italy accounted for the highest number of BTC cases, followed by the UK and Germany, whereas Spain accounted for the lowest number of cases in 2025.
- In the US, the 65+ age group accounted for ~65% of BTC cases in 2025, while patients 45–64 years represented ~30%, and <44 years accounted for only ~5%.
- BTC represent a rare but highly lethal group of malignancies in the 7MM, with CCA accounting for a substantial proportion of cases and disease burden. The epidemiology of CCA is heterogeneous across the 7MM, reflecting regional differences in underlying risk factors, healthcare access, diagnostic practices, and disease classification.
- Japan demonstrates a consistently higher disease burden compared with Western markets, likely driven by a greater prevalence of biliary tract disorders and long-standing clinical recognition of BTC.
Biliary Tract Cancer Market Recent Developments and Breakthroughs
- In January 2026, TransThera Sciences announced presentation of tinengotinib monotherapy in patients with advanced CCA at American Society of Clinical Oncology Gastrointestinal Cancers Symposium (ASCO GI) 2026.
- In December 2025, J-Pharma announced the successful completion of site activation at the first clinical trial site in the Beacon-BTC global Phase III clinical trial of nanvuranlat for the treatment of advanced BTC.
- In November 2025, AstraZeneca announced that the data from the Phase III (DESTINY-BTC01) trial, evaluating trastuzumab deruxtecan in treatment-naïve patients with advanced HER2-expressing BTC, is anticipated to read out in 2026 or later.
Biliary Tract Cancer Drug Analysis
The BTC drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships, upcoming key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the BTC treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the BTC market.
Biliary Tract Cancer Marketed Drugs
Pemigatinib (PEMAZYRE): Incyte
Pemigatinib is a kinase inhibitor indicated for the treatment of adults with previously treated, unresectable locally advanced or metastatic cholangiocarcinoma with a fibroblast growth factor receptor 2 (FGFR2) fusion or other rearrangement as detected by an FDA-approved test. In August 2025, Incyte discontinued the Phase III FIGHT-302 trial evaluating a first-line therapy for unresectable or metastatic FGFR2-rearranged cholangiocarcinoma due to poor enrollment following a shift in the standard of care. Patent expiry is expected between 2035 and 2040.
Entrectinib (ROZLYTREK): Roche/Genentech
Entrectinib is an oral tyrosine kinase inhibitor used in adults and children (>1 month) with metastatic or unresectable solid tumors harboring NTRK gene fusions or ROS1 fusions. It blocks TRK A/B/C and ROS1 kinase activity to inhibit cancer cell growth and induce tumor cell death. Marketed as ROZLYTREK, its patent expiry is expected to begin around 2028.
Biliary Tract Cancer Marketed Drugs | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Pemigatinib (PEMAZYRE) |
Incyte |
Cholangiocarcinoma with a FGFR2 fusion |
Small molecule |
FGFR 1, 2, and 3 antagonists |
Oral |
US: 2020 EU: 2021 JP: 2021 |
|
Entrectinib (ROZLYTREK) |
Roche/ Genentech |
NTRK fusion-positive solid tumors |
Small molecule |
TRKA/TRKB/TRKC/ROS1/ALK inhibitor |
Oral |
US: 2019 EU: 2020 JP: 2019 |
|
Larotrectinib (VITRAKVI) |
Bayer/ Loxo Oncology |
NTRK fusion-positive solid tumors |
Small molecule |
TRK, TRKA, TRKB, and TRKC inhibitor |
Oral |
US: 2018 EU: 2019 JP: 2021 |
|
Ivosidenib (TIBSOVO) |
Agios Pharmaceuticals/ Servier Pharmaceuticals |
Cholangiocarcinoma with an IDH1 mutation |
Small molecule |
Isocitrate dehydrogenase 1 (IDH1) inhibitor |
Oral |
US: 2021 EU: 2023 |
Biliary Tract Cancer Emerging Drugs
Tinengotinib : TransThera Sciences
Tinengotinib, is an investigational oral multi-kinase inhibitor for cholangiocarcinoma, especially in patients with FGFR-altered tumors who progressed after prior therapy. It targets FGFR1–3, VEGFR, Aurora, and JAK kinases, helping suppress tumor growth, angiogenesis, and resistance. Data were presented at the ASCO Gastrointestinal Cancers Symposium 2026, with a US launch anticipated in 2027.
Rilvegostomig: AstraZeneca
Rilvegostomig is a first-in-class dual-checkpoint bispecific antibody providing PD-1 and TIGIT blockade on the same immune cell to restore antitumor immunity and enable durable responses. Its TIGIT component is derived from COM902, developed by Compugen. In December 2025, Compugen reached an agreement with AstraZeneca to monetize part of its future royalties and amended their 2018 exclusive licensing deal to support its immuno-oncology pipeline.
Biliary Tract Cancer Emerging Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Tinengotinib |
TransThera Sciences |
III |
Cholangiocarcinoma |
Oral |
Multi-kinase inhibitor ( FGFR1–3 alongside VEGFRs, Aurora kinases, and JAK kinases) |
Information is available in the full report |
|
Rilvegostomig |
AstraZeneca |
III |
BTC |
IV infusion |
PD-1/TIGIT inhibitor |
2030 (1L) |
|
Nanvuranlat (JPH203) |
J-Pharma/ OHARA Pharmaceutical |
III |
Advanced BTC |
IV infusion |
L-type amino acid transporter (LAT) 1 inhibitor |
Information is available in the full report |
|
Tovecimig (CTX-009) |
Compass Therapeutics |
II/III |
Metastatic or locally advanced BTC |
IV infusion |
DLL4 and VEGF-A inhibitors |
Information is available in the full report |
|
Lenvatinib mesylate (LENVIMA) |
Merck/Eisai |
II |
Cholangiocarcinoma |
Oral |
Multi- RTK inhibitor |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Biliary Tract Cancer (BTC) Market Outlook
BTC is a heterogeneous group of invasive adenocarcinomas including iCCA, eCCA, GBC, and ampullary carcinoma. Prognosis remains poor, with an overall 5-year survival rate of ~15%, dropping to ~8.5% for iCCA, particularly in patients with metastatic disease. Curative treatment relies on surgical resection followed by adjuvant capecitabine, though recurrence rates are high. For advanced or unresectable BTC, first-line therapy typically includes Cisplatin + Gemcitabine, while second-line options include 5-Fluorouracil-based regimens such as FOLFOX regimen or irinotecan combinations. Recent advances in precision oncology have introduced targeted therapies for molecular alterations including FGFR2 fusions, IDH1 mutations, BRAF V600E, NTRK fusions, HER2 amplification, and MSI-H, shifting treatment toward biomarker-driven approaches, particularly in iCCA.Larotrectinib became the first therapy approved that included BTC under a tumor-agnostic indication for NTRK gene fusion–positive solid tumors. This marked the first regulatory breakthrough beyond chemotherapy in BTC.In 2019, entrectinib, developed by Roche/Genentech, was approved for NTRK fusion–positive solid tumors, including BTC. Entrectinib demonstrated a stronger efficacy signal (ORR ~46% across solid tumors), reinforcing the validity of molecularly driven treatment strategies.
Ivosidenib became the first IDH1 inhibitor approved for BTC, targeting the ~15–20% of cholangiocarcinomas harboring IDH1 mutations. This represented the first therapy addressing IDH1-driven oncogenesis in BTC.In September 2022, durvalumab was approved in combination with gemcitabine and cisplatin for first-line treatment of locally advanced or metastatic BTC. This marked the first immunotherapy-based regimen approved in the frontline setting. While survival improved, the ORR remained modest (~27%), suggesting BTC is relatively immune-resistant.Despite recent therapeutic advances in BTC, many unmet needs remain. These include the elucidation of reliable biomarkers to predict those patients with a prolonged response to immunotherapy, as well as prospective studies comparing systemic and locoregional therapies in patients with advanced disease.
Key Findings from Spinal Cord Injury Market Forecast Report
- In the 7MM, excluding conventional chemotherapy, durvalumab (IMFINZI) holds the largest market share in 2025, generating approximately USD 330 million, followed by Pemigatinib (PEMAZYRE).
- Among EU4 and the UK, the UK captured the maximum market share, followed by Italy and Germany in 2025.
- Platinum-based combination chemotherapy has historically served as the backbone of systemic therapy in advanced or unresectable disease across the 7MM, forming a foundational standard of care that many subsequent regimens build upon.
- The most significant paradigm shift in first-line therapy for BTC has come from the integration of Immunotherapy with standard chemotherapy.
Biliary Tract Cancer Market Competitive Landscape
The Biliary Tract Cancer market competitive landscape is characterized by ongoing innovation driven by the need to address limited treatment options and poor prognosis. The market is witnessing increasing focus on targeted therapies, immunotherapies, and combination approaches. Advancements in biomarker-driven treatments and precision medicine are intensifying competition, while a robust clinical pipeline continues to shape future therapeutic strategies and market dynamics.
Key Biliary Tract Cancer Market Companies
- Incyte
- Roche/Genentech
- Bayer/Loxo Oncology
- Agios Pharmaceuticals/Servier Pharmaceuticals
- Taiho
- Merck
- AstraZeneca
- Eli Lilly, and others
Further details will be provided in the report….
Biliary Tract Cancer Drugs Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the BTC drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Among the emerging first-line therapies, ZIIHERA, ENHERTU + rilvegostomig, Rilvegostomig, Tovecimig, and Silmitasertib (CX-4945)–based combinations, ZIIHERA is expected to capture the largest market share due to its anticipated earlier launch, with early competitive pressure likely from ENHERTU-based regimens.
Biliary Tract Cancer Clinical Trials Activities
The BTC market comprises monoclonal antibodies, small molecules, bispecific antibodies, and others, each targeting different aspects of tumor growth and progression.
Bispecific antibody: Rilvegostomig (AZD2936) and Tovecimig (CTX-009) represent emerging bispecific antibody strategies in advanced Biliary Tract Cancer. Rilvegostomig targets PD-1 and TIGIT, enabling dual immune checkpoint blockade in a tumor type traditionally considered immune-cold, while Tovecimig targets DLL4 and VEGF-A to inhibit complementary angiogenic pathways.
Small molecule: Tasurgratinib received approval in Japan for unresectable BTC with FGFR2 fusions or rearrangements after chemotherapy progression, reflecting regional regulatory momentum in BTC innovation. Ivosidenib became the first IDH1 inhibitor approved for BTC, targeting the ~15–20% of cholangiocarcinomas harboring IDH1 mutations. Dabrafenib + trametinib demonstrated encouraging efficacy in BRAF V600E–mutant BTC and is recommended by NCCN as a Category 2A option for unresectable or metastatic disease with this mutation.
Detailed insights of emerging therapies' drug uptake is included in the report
Biliary Tract Cancer Market Access and Reimbursement
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
NOTE: Further Details are provided in the final report….
Biliary Tract Cancer Price Scenario & Trends
Pricing and analogue assessment of BTC therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Latest KOL Views on Biliary Tract Cancer
To keep up with BTC market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the BTC emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in BTC, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the University of California, Imperial College London, Hokkaido University, and Cholangiocarcinoma Foundation, etc. were contacted.Their opinion helps understand and validate current and emerging BTC therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in BTC.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Despite recent advances, including the approval of targeted agents and immune-based combinations, there remains a clear unmet need for improved treatment options for patients with BTC because only around 50% of tumors harbor actionable molecular aberrations and many effective targeted therapies are still lacking.” |
|
Italy |
“BTC market on the cusp of significant change, moving from a "onesize-fits-all" chemotherapy approach to a highly segmented, biomarker-driven landscape. The successful commercialization of these future therapies will depend heavily on closing the testing gap in community oncology and effectively managing the unique toxicity profiles of these potent new biologic agents.” |
Biliary Tract Cancer Qualitative Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of BTC, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Biliary Tract Cancer Market Report
- The Biliary Tract Cancer Market report covers a segment of key events, an executive summary, a descriptive overview of BTC, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the BTC market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Biliary Tract Cancer Market report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM BTC market.
Biliary Tract Cancer Market Report Insights
- Biliary Tract Cancer (BTC) Patient Population Forecast
- Biliary Tract Cancer (BTC) Therapeutics Market Size
- Biliary Tract Cancer (BTC) Pipeline Analysis
- Biliary Tract Cancer (BTC) Market Size and Trends
- Biliary Tract Cancer (BTC) Market Opportunity (Current and Forecasted)
Biliary Tract Cancer Market Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year Forecast
- Biliary Tract Cancer (BTC) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by Geography)
- Biliary Tract Cancer (BTC) Treatment Addressable Market (TAM)
- Biliary Tract Cancer (BTC) Competitve Landscape
- Biliary Tract Cancer (BTC) Major Companies Insights
- Biliary Tract Cancer (BTC) Price Trends and Analogue Assessment
- Biliary Tract Cancer (BTC) Therapies and Drug Adoption/Uptake
- Biliary Tract Cancer (BTC) Therapies Peak Patient Share Analysis
Biliary Tract Cancer Market Report Assessment
- Biliary Tract Cancer (BTC) Current Treatment Practices
- Biliary Tract Cancer (BTC) Unmet Needs
- Biliary Tract Cancer (BTC) Clinical Development Analysis
- Biliary Tract Cancer (BTC) Emerging Drugs Product Profiles
- Biliary Tract Cancer (BTC) Market Attractiveness
- Biliary Tract Cancer (BTC) Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Biliary Tract Cancer Market Insights
- What was the BTC market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of BTC?
- What are the disease risks, burdens, and unmet needs of BTC? What will be the growth opportunities across the 7MM concerning the patient population with BTC?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of BTC? What are the current guidelines for treating BTC in the US, Europe, and Japan?
Reasons to Buy the Biliary Tract Cancer Market Forecast Report
- The Biliary Tract Cancer Market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the BTC market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets withing the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.


-pipeline-report.png&w=256&q=75)


