Botaretigene sparoparvovec Market Sales
Key Factors Driving Botaretigene sparoparvovec Growth
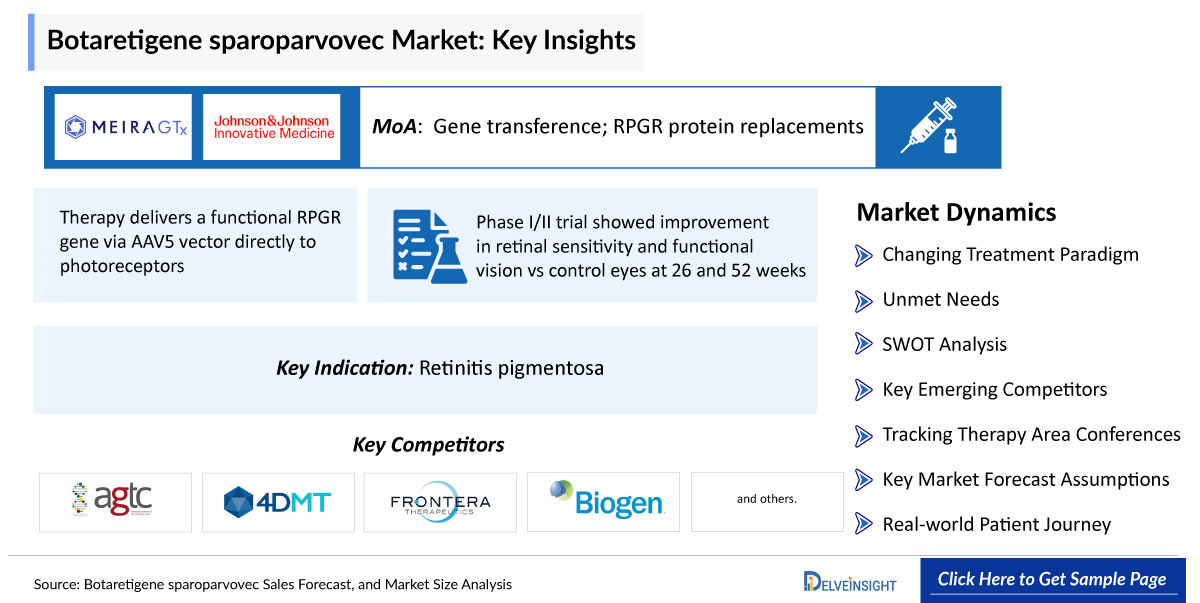
1. Demonstrated clinical efficacy with functional vision improvement
- Phase I/II trial (published in American Journal of Ophthalmology / PubMed) showed:
- Improvement in retinal sensitivity and functional vision vs control eyes at 26 and 52 weeks
- Additional analyses showed:
- Improved static perimetry and mobility performance in treated eyes
2. Favorable and manageable safety profile
- No dose-limiting toxicities reported in Phase I/II
- Most adverse events:
- Transient and procedure-related
- Responsive to corticosteroids
3. Strong biological rationale targeting RPGR mutation
- Therapy delivers a functional RPGR gene via AAV5 vector directly to photoreceptors
- RPGR mutations are a leading cause of X-linked retinitis pigmentosa (XLRP)
- XLRP leads to:
- Progressive vision loss - legal blindness by ~4th decade
- No curative treatment currently available
4. High unmet medical need and orphan indication
- Retinitis pigmentosa affects ~1 in 3,000–4,000 individuals globally
- RPGR-associated XLRP is among the most severe and rapidly progressing forms
5. Regulatory incentives accelerating development
Botaretigene sparoparvovec has received:
- FDA Fast Track designation
- Orphan Drug designation (US & EU)
- PRIME designation (EMA)
These provide:
- Faster review timelines
- Market exclusivity (7–10 years)
- Reduced regulatory burden
Botaretigene sparoparvovec Recent Developments
- In February 2024, MeiraGTx received USD 50 million milestone after initiation of the extension study for the Phase III LUMEOS clinical trial for botaretigene sparoparvovec (bota-vec, formerly AAV-RPGR) for the treatment of X-linked retinitis pigmentosa (XLRP).
“Botaretigene sparoparvovec Sales Forecast, and Market Size Analysis – 2034” report provides comprehensive insights of Botaretigene sparoparvovec for potential indication like Retinitis pigmentosa in the 7MM. A detailed picture of Botaretigene sparoparvovec’s existing usage in anticipated entry and performance in potential indications in the 7MM, i.e., the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan for the study period 2020 –2034 is provided in this report along with a detailed description of the Botaretigene sparoparvovec for potential indications. The Botaretigene sparoparvovec market report provides insights about Botaretigene sparoparvovec’s sales forecast, mechanism of action (MoA), dosage and administration, as well as research and development including regulatory milestones, along with other developmental activities. Further, it also consists of historical and current Botaretigene sparoparvovec performance, future market assessments inclusive of the Botaretigene sparoparvovec market forecast analysis for potential indications in the 7MM, SWOT, analysts’ views, comprehensive overview of market competitors, and brief about other emerging therapies in respective indications. It also provides analysis of Botaretigene sparoparvovec sales forecasts, along with factors driving its market.
Botaretigene sparoparvovec Drug Summary
Botaretigene sparoparvovec is an investigational adeno-associated virus (AAV)-based gene therapy developed by MeiraGTx in collaboration with Johnson & Johnson for the treatment of X-linked retinitis pigmentosa (XLRP) caused by mutations in the RPGR. The therapy is designed to deliver a functional copy of the RPGR gene directly to retinal photoreceptor cells via subretinal injection, thereby restoring normal protein expression and preserving visual function. By addressing the underlying genetic defect, botaretigene sparoparvovec aims to slow or halt disease progression and potentially improve vision in affected individuals. Clinical studies have demonstrated encouraging results, including improvements in retinal sensitivity and functional vision, along with a generally manageable safety profile, supporting its potential as a one-time, disease-modifying treatment for inherited retinal degenerative disorders. The report provides Botaretigene sparoparvovec’s sales, growth barriers and drivers, post usage and approvals in multiple indications.
Scope of the Botaretigene sparoparvovec Market Report
The report provides insights into:
- A comprehensive product overview including the Botaretigene sparoparvovec MoA, description, dosage and administration, research and development activities in potential indication like Retinitis pigmentosa.
- Elaborated details on Botaretigene sparoparvovec regulatory milestones and other development activities have been provided in Botaretigene sparoparvovec market report.
- The report also highlights Botaretigene sparoparvovec‘s cost estimates and regional variations, reported and estimated sales performance, research and development activities in potential indications across the United States, Europe, and Japan.
- The Botaretigene sparoparvovec market report also covers the patents information, generic entry and impact on cost cut.
- The Botaretigene sparoparvovec market report contains current and forecasted Botaretigene sparoparvovec sales for potential indications till 2034.
- Comprehensive coverage of the late-stage emerging therapies for respective indications.
- The Botaretigene sparoparvovec market report also features the SWOT analysis with analyst views for Botaretigene sparoparvovec in potential indications.
Methodology
The Botaretigene sparoparvovec market report is built using data and information sourced primarily from internal databases, primary and secondary research and in-house analysis by DelveInsight’s team of industry experts. Information and data from the secondary sources have been obtained from various printable and nonprintable sources like search engines, news websites, global regulatory authorities websites, trade journals, white papers, magazines, books, trade associations, industry associations, industry portals and access to available databases.
Botaretigene sparoparvovec Analytical Perspective by DelveInsight
- In-depth Botaretigene sparoparvovec Market Assessment
This Botaretigene sparoparvovec sales market forecast report provides a detailed market assessment of Botaretigene sparoparvovec for potential indication like Retinitis pigmentosa in the seven major markets, i.e., the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan. This segment of the report provides current and forecasted Botaretigene sparoparvovec sales data uptil 2034.
- Botaretigene sparoparvovec Clinical Assessment
The Botaretigene sparoparvovec market report provides the clinical trials information of Botaretigene sparoparvovec for potential indications covering trial interventions, trial conditions, trial status, start and completion dates.
Botaretigene sparoparvovec Competitive Landscape
The report provides Insights on competitors and marketed products within the domain, along with a summary of emerging products and their respective launch dates, posing significant competition in the market.
Botaretigene sparoparvovec Market Potential & Revenue Forecast
- Projected market size for the Botaretigene sparoparvovec and its key indications
- Estimated Botaretigene sparoparvovec sales potential (Botaretigene sparoparvovec peak sales forecasts)
- Botaretigene sparoparvovec Pricing strategies and reimbursement landscape
Botaretigene sparoparvovec Competitive Intelligence
- Number of competing drugs in development (pipeline analysis)
- Botaretigene sparoparvovec Market positioning compared to existing treatments
- Botaretigene sparoparvovec Strengths & weaknesses relative to competitors
Botaretigene sparoparvovec Regulatory & Commercial Milestones
- Botaretigene sparoparvovec Key regulatory approvals & expected launch timelines
- Commercial partnerships, licensing deals, and M&A activity
Botaretigene sparoparvovec Clinical Differentiation
- Botaretigene sparoparvovec Efficacy & safety advantages over existing drugs
- Botaretigene sparoparvovec Unique selling points
Botaretigene sparoparvovec Market Report Highlights
- In the coming years, the Botaretigene sparoparvovec market scenario is set to change due to strong adoption, increased prescriptions and broader uptake in multiple immunological indications; which would expand the size of the market.
- The Botaretigene sparoparvovec companies are developing therapies that focus on novel approaches to treat/improve the disease condition, assess challenges, and seek opportunities that could influence Botaretigene sparoparvovec’s dominance.
- Other emerging products for Retinitis pigmentosa are expected to give tough market competition to Botaretigene sparoparvovec and launch of late-stage emerging therapies in the near future will significantly impact the market.
- A detailed description of regulatory milestones, and developmental activities, provide the current development scenario of Botaretigene sparoparvovec in potential indications.
- Analyse Botaretigene sparoparvovec cost, pricing trends and market positioning to support strategic decision-making in the immunology landscape.
- Our in-depth analysis of the forecasted Botaretigene sparoparvovec sales data uptil 2034 will support the clients in decision-making process regarding their therapeutic portfolio by identifying the overall scenario of Botaretigene sparoparvovec in potential indications.
Key Questions Answered In The Botaretigene sparoparvovec Market Report
- What is the class of therapy, route of administration and mechanism of action of Botaretigene sparoparvovec? How strong is Botaretigene sparoparvovec’s clinical and commercial performance?
- What is Botaretigene sparoparvovec’s clinical trial status in each individual indications such as Retinitis pigmentosa and study completion date?
- What are the key collaborations, mergers and acquisitions, licensing and other activities related to the Botaretigene sparoparvovec Manufacturers?
- What are the key designations that have been granted to Botaretigene sparoparvovec for potential indications? How are they going to impact Botaretigene sparoparvovec’s penetration in various geographies?
- What is the current and forecasted Botaretigene sparoparvovec market scenario for potential indications? What are the key assumptions behind the forecast?
- What are the current and forecasted sales of Botaretigene sparoparvovec in the seven major countries, including the United States, Europe (Germany, France, Italy, Spain) and the United Kingdom, and Japan?
- What are the other emerging products available and how are these giving competition to Botaretigene sparoparvovec for potential indications?
- Which are the late-stage emerging therapies under development for the treatment of potential indications?
- How cost-effective is Botaretigene sparoparvovec? What is the duration of therapy and what are the geographical variations in cost per patient