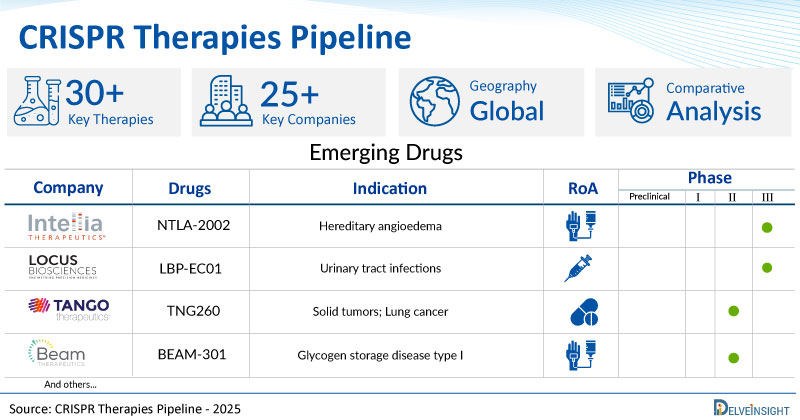
CRISPR Therapies Pipeline
DelveInsight’s, “CRISPR Therapies – Pipeline Insight, 2026,” report provides comprehensive insights about 25+ companies and 30+ pipeline drugs in CRISPR Therapies pipeline landscape. It covers the pipeline drug profiles, including clinical and nonclinical stage products. It also covers the therapeutics assessment by product type, stage, route of administration, and molecule type. It further highlights the inactive pipeline products in this space.
Geography Covered
- Global coverage
CRISPR Therapies Understanding
CRISPR Therapies: Overview
CRISPRs are specialized stretches of DNA. ""CRISPR"" stands for ""clusters of regularly interspaced short palindromic repeats."" It is a specialized region of DNA with two distinct characteristics: the presence of nucleotide repeats and spacers. Repeated sequences of nucleotides — the building blocks of DNA — are distributed throughout a CRISPR region. Spacers are bits of DNA that are interspersed among these repeated sequences. CRISPRs play a role in regulating bacterial immunity.
CRISPR-Cas9 offers a high degree of fidelity and relatively simple construction. It depends on two factors for its specificity: the target sequence and the protospacer adjacent motif (PAM) sequence. The target sequence is 20 bases long as part of each CRISPR locus in the crRNA array. The PAM sequence on the host genome is recognized by Cas9. Cas9 cannot be easily modified to recognize a different PAM sequence. Once these sequences have been assembled into a plasmid and transfected into cells the Cas9 protein with the help of the crRNA finds the correct sequence in the host cell's DNA.
Casgevy is the first FDA-approved therapy utilizing CRISPR/Cas9 in which patients’ hematopoietic (blood) stem cells are modified by genome editing using CRISPR/Cas9 technology. Casgevy, a cell-based gene therapy, which is approved for the treatment of sickle cell disease in patients 12 years of age and older with recurrent vaso-occlusive crises.
The genomes of various organisms encode a series of messages and instructions within their DNA sequences. Genome editing involves changing those sequences, thereby changing the messages. This can be done by inserting a cut or break in the DNA and tricking a cell's natural DNA repair mechanisms into introducing the changes and CRISPR-Cas9 provides a means to do so. CRISPR/Cas9 is a simple two-component system used for effective targeted gene editing. The first component is the single-effector Cas9 protein, which contains the endonuclease domains RuvC and HNH. RuvC cleaves the DNA strand non-complementary to the spacer sequence and HNH cleaves the complementary strand. Together, these domains generate double-stranded breaks (DSBs) in the target DNA. The second component of effective targeted gene editing is a single guide RNA (sgRNA) carrying a scaffold sequence which enables its anchoring to Cas9 and a 20 base pair spacer sequence complementary to the target gene and adjacent to the PAM sequence. This sgRNA guides the CRISPR/Cas9 complex to its intended genomic location. The editing system then relies on either of two endogenous DNA repair pathways: non-homologous end-joining (NHEJ) or homology-directed repair. Now, as a gene-editing tool, CRISPR/Cas9 has revolutionized biomedical research and may soon enable medical breakthroughs in a way few biological innovations. The recent advent of CRISPR technology in clinical trials has paved way for the new era of CRISPR gene therapy to emerge.
Several concerns exist for the implementation of CRISPR for gene editing. First, there are concerns of off-target effects; although some studies have shown that there are insignificant or even undetectable levels of off-target effects, others have clear documentation of large insertions or deletions that may result in unintended consequences. Second, immunogenicity against CRISPR proteins would need to be monitored given observations of pre-existing serum antibodies to Cas9 in some donors or prevalence of Cas9-reactive T cells in some patients Finally, most CRISPR-mediated gene therapy has focused on ex vivo treatment, namely editing of stem cells outside the body and subsequent reintroduction of corrected cells back into patients.
""CRISPR Therapies- Pipeline Insight, 2026"" report by DelveInsight outlays comprehensive insights of present scenario and growth prospects across the indication. A detailed picture of the CRISPR Therapies pipeline landscape is provided which includes the disease overview and CRISPR Therapies treatment guidelines. The assessment part of the report embraces, in depth CRISPR Therapies commercial assessment and clinical assessment of the pipeline products under development. In the report, detailed description of the drug is given which includes mechanism of action of the drug, clinical studies, NDA approvals (if any), and product development activities comprising the technology, CRISPR Therapies collaborations, licensing, mergers and acquisition, funding, designations and other product related details.
Report Highlights
The companies and academics are working to assess challenges and seek opportunities that could influence CRISPR Therapies R&D. The therapies under development are focused on novel approaches to treat/improve CRISPR Therapies.
CRISPR Therapies Emerging Drugs Chapters
This segment of the CRISPR Therapies report encloses its detailed analysis of various drugs in different stages of clinical development, including phase III, II, II/III I, preclinical and Discovery. It also helps to understand clinical trial details, expressive pharmacological action, agreements and collaborations, and the latest news and press releases.
CRISPR Therapies Emerging Drugs
LBP-EC01: Locus Biosciences
LBP-EC01 is a Locus crPhage® therapy in development for the treatment of urinary tract infections and other infections caused by the pathogen Escherichia coli (E. coli). It is a bacteriophage cocktail engineered with a CRISPR-Cas3 construct targeting the E. coli genome. The precision medicine product works through a unique dual mechanism of action utilizing both the natural lytic activity of the bacteriophage and the DNA-targeting activity of CRISPR-Cas3. Laboratory tests and small animal models of urinary tract infection have demonstrated LBP-EC01 is significantly more effective at killing E. coli than corresponding natural bacteriophages, and LBP-EC01 met all primary and secondary endpoints and demonstrated safety and tolerability in a Phase 1b trial. LBP-EC01 is currently being evaluated in a registrational Phase II/III trial for the treatment of UTIs caused by E. coli.
NTLA-2002: Intellia Therapeutics, Inc
NTLA-2002 is a wholly owned first single-dose investigational CRISPR therapeutic candidate designed to inactivate the kallikrein B1 (KLKB1) gene, which encodes for prekallikrein, the kallikrein precursor protein. NTLA-2002 is Intellia’s second investigational CRISPR therapeutic candidate to be administered systemically, by intravenous infusion, to edit disease-causing genes inside the human body with a single dose of treatment. Intellia’s proprietary non-viral platform deploys lipid nanoparticles to deliver to the liver a two-part genome editing system: guide RNA specific to the disease-causing gene and messenger RNA that encodes the Cas9 enzyme, which together carry out the precision editing. Currently, the drug is in the Phase I/II stage of its development for the treatment of CRISPR Therapies
CB-010: Caribou Biosciences, Inc
CB-010 is the lead clinical-stage product candidate from Caribou’s allogeneic CAR-T cell therapy platform, and it is being evaluated in patients with relapsed or refractory B cell non-Hodgkin lymphoma (r/r B-NHL) in the ongoing ANTLER Phase I clinical trial and will be evaluated in patients with lupus nephritis (LN) and extrarenal lupus (ERL) in the GALLOP Phase 1 clinical trial. In ANTLER, Caribou is enrolling second-line patients with large B cell lymphoma (LBCL) comprised of different subtypes of aggressive r/r B-NHL (DLBCL NOS, PMBCL, HGBL, tFL, and tMZL). To Caribou’s knowledge, CB-010 is the first allogeneic CAR-T cell therapy in the clinic with a PD-1 knockout, a genome-editing strategy designed to improve activity against diseases by limiting premature CAR-T cell exhaustion. CB-010 is also, to Caribou’s knowledge, the first anti-CD19 allogeneic CAR-T cell therapy to be evaluated in the second-line LBCL setting and, for r/r B-NHL, CB-010 has been granted Regenerative Medicine Advanced Therapy (RMAT), Fast Track, and Orphan Drug designations by the FDA. Currently, the drug is in the Phase I stage of its development for the treatment of CRISPR Therapies
Further product details are provided in the report……..
CRISPR Therapies: Therapeutic Assessment
This segment of the report provides insights about the different CRISPR Therapies drugs segregated based on following parameters that define the scope of the report, such as:
Major Players in CRISPR Therapies
There are approx. 25+ key companies which are developing the therapies for CRISPR Therapies. The companies which have their CRISPR Therapies drug candidates in the most advanced stage, i.e. Phase III include, Locus Biosciences.
Phases
DelveInsight’s report covers around 30+ products under different phases of clinical development like
- Late stage products (Phase III)
- Mid-stage products (Phase II)
- Early-stage product (Phase I) along with the details of
- Pre-clinical and Discovery stage candidates
- Discontinued & Inactive candidates
Route of Administration
CRISPR Therapies pipeline report provides the therapeutic assessment of the pipeline drugs by the Route of Administration. Products have been categorized under various ROAs such as
- Intravenous
- Subcutaneous
- Oral
- Intramuscular
Molecule Type
Products have been categorized under various Molecule types such as
- Monoclonal antibody
- Small molecule
- Peptide
Product Type
Drugs have been categorized under various product types like Mono, Combination and Mono/Combination.
CRISPR Therapies: Pipeline Development Activities
The report provides insights into different therapeutic candidates in phase II, I, preclinical and discovery stage. It also analyses CRISPR Therapies therapeutic drugs key players involved in developing key drugs.
Pipeline Development Activities
The report covers the detailed information of collaborations, acquisition and merger, licensing along with a thorough therapeutic assessment of emerging CRISPR Therapies drugs.
CRISPR Therapies Report Insights
- CRISPR Therapies Pipeline Analysis
- Therapeutic Assessment
- Unmet Needs
- Impact of Drugs
CRISPR Therapies Report Assessment
- Pipeline Product Profiles
- Therapeutic Assessment
- Pipeline Assessment
- Inactive drugs assessment
- Unmet Needs
Key Questions
Current Treatment Scenario and Emerging Therapies:
- How many companies are developing CRISPR Therapies drugs?
- How many CRISPR Therapies drugs are developed by each company?
- How many emerging drugs are in mid-stage, and late-stage of development for the treatment of CRISPR Therapies?
- What are the key collaborations (Industry–Industry, Industry–Academia), Mergers and acquisitions, licensing activities related to the CRISPR Therapies therapeutics?
- What are the recent trends, drug types and novel technologies developed to overcome the limitation of existing therapies?
- What are the clinical studies going on for CRISPR Therapies and their status?
- What are the key designations that have been granted to the emerging drugs?