Down Syndrome Market
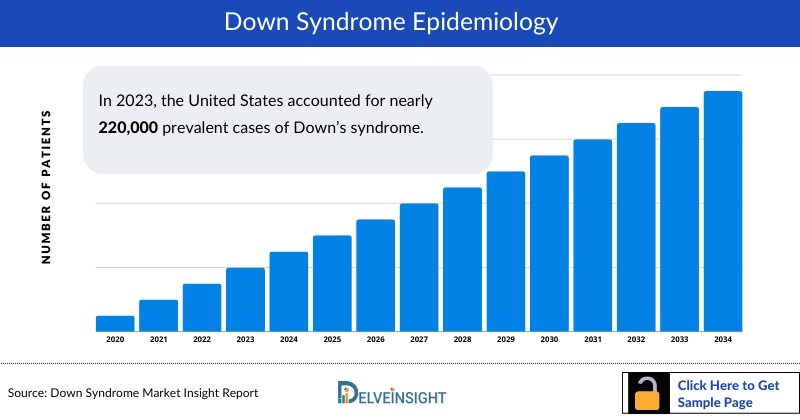
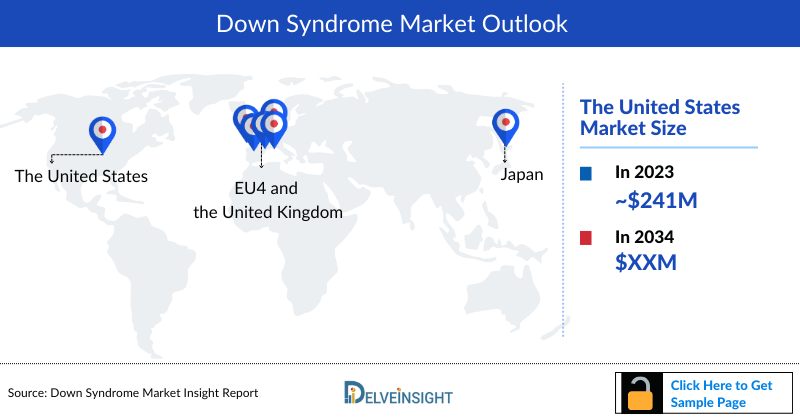
- The Down Syndrome market size in 2023 was approximately USD 241 million in the United States.
- Down Syndrome is a genetic condition caused by an error in cell division called "nondisjunction," resulting in an individual having a full or partial extra copy of chromosome 21. This additional genetic material alters development, leading to the physical and intellectual characteristics associated with Down Syndrome.
- In 2025, the US reported the highest diagnosed prevalence of Down syndrome among the 7MM, with approximately 315 thousand cases. DelveInsight forecasts this number to rise throughout the 2026–2036 period.
- Down Syndrome is more common in females than males.
- The types of Down Syndrome include Trisomy 21, Robertsonian translocation, and Mosaicism. In 2023, among all types, Trisomy 21 accounted for the highest number of prevalent cases in the 7MM.
- There is no single, standard treatment for Down Syndrome. Treatments are based on each individual’s physical and intellectual needs, as well as their strengths and limitations. People with Down Syndrome can receive proper care while living at home and in the community.
Download the Sample PDF to Get More Insight @ Down Syndrome Market
DelveInsight’s "Down Syndrome – Market Insight, Epidemiology, and Market Forecast – 2036" report delivers an in-depth understanding of Down Syndrome, historical and forecasted epidemiology as well as the Down Syndrome market trends in the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan.
The Down Syndrome market report provides current treatment practices, emerging down syndrome therapies, Down Syndrome market share of individual therapies, and current and forecasted Down Syndrome market size from 2022 to 2036, segmented by seven major markets. The report also covers current Down Syndrome treatment practices/algorithms and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the down syndrome treatment market.
Geography Covered
- The United States
- EU4 (Germany, France, Italy, and Spain) and the United Kingdom
- Japan
Key Factors Driving the Down Syndrome Market
Rising Down Syndrome Prevalence
In 2025, the US reported the highest diagnosed prevalence of Down syndrome among the 7MM, with approximately 315 thousand cases. DelveInsight forecasts this number to rise throughout the 2026–2036 period.
Technological Innovations in Diagnostic Tools
The development of advanced diagnostic tools, such as high-resolution imaging and genetic testing platforms, has improved the accuracy and efficiency of Down syndrome detection. These innovations facilitate early diagnosis, enabling prompt therapeutic interventions and better management of the condition.
Growing Focus on Personalized Medicine
There is an increasing emphasis on personalized medicine approaches tailored to the unique genetic profiles of individuals with Down syndrome. This shift allows for more effective and individualized treatment plans, addressing the specific needs and challenges faced by each patient.
Expected Launch of Emerging Down Syndrome Drugs
Some of the drugs in the pipeline for Down Syndrome include AEF0117 (Aelis Frama), Valiltramiprosate (Alzheon), Lecanemab (BioArctic/Eisai), among others.
Down Syndrome Understanding and Treatment Algorithm
Down Syndrome is a genetic condition caused by an error in cell division called "nondisjunction," resulting in an individual having a full or partial extra copy of chromosome 21. This additional genetic material alters development, leading to the physical and intellectual characteristics associated with Down Syndrome. Down Syndrome manifests in three primary forms: Trisomy 21, Translocation Down Syndrome, and Mosaic Down Syndrome. Trisomy 21, the most prevalent type, results from an extra copy of chromosome 21 in every cell, constituting about more than 90% of cases. Each type carries its own set of characteristics and variations in severity, shaping the spectrum of the condition's expression.
Down Syndrome Diagnosis
A screening test can tell a woman and her healthcare provider whether her pregnancy has a lower or higher chance of having Down Syndrome. Screening tests do not provide an absolute diagnosis, but they are safer for the mother and the developing baby. Diagnostic tests can typically detect whether or not a baby will have Down Syndrome, but they can be more risky for the mother and developing baby. Neither screening nor diagnostic tests can predict the full impact of Down Syndrome on a baby; no one can predict this. Screening tests often include a combination of a blood test, which measures the amount of various substances in the mother’s blood (e.g., Noninvasive prenatal testing, MS-AFP, Triple Screen, Quad-screen), and an ultrasound, which creates a picture of the baby. During an ultrasound, one of the things the technician looks at is the fluid behind the baby’s neck. Extra fluid in this region could indicate a genetic problem. These screening tests can help determine the baby’s risk of Down Syndrome.
Further details related to diagnosis will be provided in the report…
Down Syndrome Treatment
There is no single, standard treatment for Down Syndrome. Treatments are based on each individual’s physical and intellectual needs, as well as their strengths and limitations. People with Down Syndrome can receive proper care while living at home and in the community. A child with Down Syndrome likely will receive care from a team of health professionals, including, but not limited to, physicians, special educators, speech therapists, occupational therapists, physical therapists, and social workers. All professionals who interact with children with Down Syndrome should provide stimulation and encouragement.
Further details related to treatment will be provided in the report…..
Down Syndrome Epidemiology
The Down Syndrome epidemiology chapter in the report provides historical as well as forecasted epidemiology segmented by the Total Down Syndrome prevalence, Type-specific Cases of Down Syndrome, Gender-specific Cases of Down Syndrome, Down Syndrome prevalence by clinical manifestations, Prevalent Cases of Down Syndrome by Age in the 7MM covering the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan from 2022 to 2036.
- In 2025, the US reported the highest diagnosed prevalence of Down syndrome among the 7MM, with approximately 315 thousand cases. DelveInsight forecasts this number to rise throughout the 2026–2036 period.
- DelveInsight analysis indicates that in 2025, Germany had approximately 31 thousand diagnosed prevalent cases of Down syndrome in males and 26 thousand in females.
- Down syndrome, categorized into Trisomy 21, Translocation, and Mosaicism, showed ~147 thousand; ~3 thousand; and ~5 thousand cases respectively in Japan in 2025. According to DelveInsight, Trisomy 21 had the highest prevalence and Translocation the lowest.
- Based on classification of diagnosed cases of Down syndrome by clinical manifestations, in 2025, the UK had the highest prevalence of Down syndrome cases involving hearing loss, with more than 32 thousand cases.
- Age-specific cases of Down syndrome are classified into groups: 0–9 years, 10–19 years, 20–29 years, 30–39 years, 40–49 years, 50–59 years, and 60 and above. DelveInsight estimates indicate that Down syndrome is most prevalent in the 0–9 age group, with ~70 thousand cases reported in the US in 2025.
Get detailed insights into the historical as well as forecasted epidemiology trends in the 7MM, at: Down Syndrome Epidemiology Forecast
Down Syndrome Drugs
The down syndrome drugs chapter segment of the Down Syndrome report encloses a detailed analysis of the late-stage (Phase III) and mid-stage pipeline drugs. The current down syndrome companies include Aelis Farma and AC Immune. The drug chapter also helps understand the Down Syndrome clinical trials details, pharmacological action, agreements and collaborations, approval, and patent details, and the latest news and press releases.
Down Syndrome Emerging Drugs
AEF0217: Aelis Farma
AEF0217, developed by Aelis Farma as a CB1 receptor-specific inhibitor, targets the brain's endocannabinoid system. Currently in Phase IIb trials for Down syndrome, AEF0217 exhibits notable features, including high potency in reversing cognitive deficits across validated models of Down syndrome, Fragile X syndrome, and age-related cognitive impairments, without detectable behavioral side effects in fragile trisomic and aged mice.
Clinical development of AEF0217, for which its first therapeutic indication is the cognitive deficits in Down syndrome, is supported by a grant from the EU (Horizon Programme H2020; Improving Cognition in Down syndrome, Grant N° 899986).
The favorable PDCO opinion strengthens the credibility of the AEF0217 program, de-risks later-stage regulatory requirements, and improves visibility on the European pathway toward potential marketing authorization for this pediatric-focused therapy.
ACI-24.060: AC Immune/Takeda
ACI-24.060, developed by AC Immune in partnership with Takeda Pharmaceutical Company, is an anti-amyloid beta (Aβ) active immunotherapy currently in Phase Ib/II for Alzheimer's disease associated with Down syndrome. The vaccine is designed to generate antibodies targeting misfolded Aβ species, to reduce amyloid plaque accumulation and support biomarker-driven disease modification.
In a Phase Ib randomized trial published in May 2022 involving adults with Down syndrome, ACI-24 demonstrated favorable safety and tolerability, elicited immunogenic responses in several participants, and showed measurable effects on Alzheimer’s disease biomarkers, supporting further clinical development.
CTH120: Connecta Therapeutics
CTH120 is a first-in-class small-molecule neuroplasticity modulator being investigated for neurodevelopmental disorders, including Down syndrome. Developed after evaluating more than 30 neuroplasticity-related targets, preclinical studies show the compound enhances neuroplasticity and demonstrates potential pro-cognitive and social behavior–modulating effects. Currently, CTH120 remains in the preclinical stage for Down syndrome. In March 2025, Connecta Therapeutics reported positive Phase I results for CTH120, a neuroplasticity modulator being explored for disorders including Down syndrome and Fragile X syndrome.
A detailed assessment will be provided in the final report.
Down Syndrome Market Outlook
Down Syndrome (DS) is the most common genomic disorder of intellectual disability and is caused by the trisomy of Homo sapiens chromosome 21 (HSA21). Individuals with trisomy 21 present with a distinct collection of symptoms and manifestations that affect multiple body systems, although variation exists between individuals. The management of patients with Down Syndrome is multidisciplinary. Newborns with suspicion of Down Syndrome should have a karyotyping done to confirm the diagnosis. The family needs to be referred to the clinical geneticist for the genetic testing and counseling of both parents.
There is no single standard treatment, but a range of therapies and interventions are available to help individuals with Down Syndrome reach their full potential. Treatment options include physical therapy that helps with motor skills, muscle strength, and coordination; occupational therapy that focuses on developing everyday skills like dressing, eating, and using technology; speech therapy, which aids in improving communication skills; special education services provided under federal law to ensure appropriate education for children with disabilities; behavioral counseling that addresses conditions like ADHD or autism that may coexist with Down Syndrome; social and recreational activities for promoting social interaction and engagement; and health monitoring to manage associated health conditions like heart defects, digestive issues, and thyroid problems.
Currently, several emerging down syndrome therapies, such as AEF0217, ACI-24.060, and others, are being evaluated in mid-stage and early-stage down syndrome clinical trials.
Detailed market assessment will be provided in the final report.
Down Syndrome Drug Uptake
This section focuses on the uptake rate of potential down syndrome drugs expected to be launched in the market during 2026–2036, which depends on the competitive landscape, safety, and efficacy data along with order of entry. It is important to understand that the key players evaluating their novel therapies in the pivotal and confirmatory trials should remain vigilant when selecting appropriate comparators to stand the greatest chance of a positive opinion from regulatory bodies, leading to approval, smooth launch, and rapid uptake.
Down Syndrome Pipeline Development Activities
The report provides insights into therapeutic candidates in different stages. It also analyzes key down syndrome companies involved in developing targeted therapeutics. Companies like Aelis Farma and AC Immune, and others actively engage in late and mid-stage research and development efforts for Down Syndrome. The Down Syndrome pipeline possesses a few potential drugs. However, there is a positive outlook for the down syndrome therapeutics market, with expectations of down syndrome market growth during the forecast period (2026–2036).
Pipeline Development Activities
The report covers information on collaborations, acquisitions and mergers, licensing, and patent details for Down Syndrome emerging therapy.
KOL Views
To keep up with current market trends, we take KOLs and SMEs’ opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts contacted for insights on Down Syndrome’ evolving treatment landscape, patient reliance on conventional therapies, patient therapy switching acceptability, and drug uptake, along with challenges related to accessibility, including MDs, radiologists, oncologists, and others.
DelveInsight’s analysts connected with 20+ KOLs to gather insights; however, interviews were conducted with 10+ KOLs in the 7MM. The opinion helps understand and validate current and emerging therapy treatment patterns or Down Syndrome market trends. This will support the clients in potential upcoming novel treatments by identifying the overall scenario of the market and the unmet needs.
Qualitative Analysis
We perform Qualitative and market Intelligence analysis using various approaches, such as SWOT and Conjoint Analysis. In the SWOT analysis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. These pointers are based on the Analyst’s discretion and assessment of the patient burden, cost analysis, and existing and evolving treatment landscape.
Market Access and Reimbursement
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of currently used therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Detailed market access and reimbursement assessment will be provided in the final report.
Scope of the Report
- The report covers a segment of key events, an executive summary, and a descriptive overview of Down Syndrome, explaining their causes, signs, symptoms, pathogenesis, and currently used therapies.
- Comprehensive insight into the epidemiology segments and forecasts, disease progression, and treatment guidelines has been provided.
- Additionally, an all-inclusive account of the emerging therapies and the elaborative profiles of late-stage and prominent therapies will impact the current treatment landscape.
- A detailed review of the Down Syndrome market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help shape and drive Down Syndrome.
Down Syndrome Report Insights
- Patient Population
- Therapeutic Approaches
- Down Syndrome Pipeline Analysis
- Down Syndrome Market Size and Trends
- Existing and Future Market Opportunity
Down Syndrome Report Key Strengths
- Eleven Years Forecast
- The 7MM Coverage
- Down Syndrome Epidemiology Segmentation
- Key Cross Competition
- Drugs Uptake and Key Market Forecast Assumptions
Down Syndrome Report Assessment
- Current Treatment Practices
- Unmet Needs
- Pipeline Product Profiles
- Market Attractiveness
- Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
- What was the Down Syndrome market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for down syndrome market growth?
- What can be the future treatment paradigm for Down Syndrome?
- What are the disease risks, burdens, and unmet needs of Down Syndrome? What will be the growth opportunities across the 7MM concerning the patient population with Down Syndrome?
- What are the current options for the Down Syndrome treament? What are the current guidelines for treating Down Syndrome in the 7MM?
- What are the recent novel therapies, targets, down syndrome mechanism of action (down syndrome MOA), and technologies being developed to overcome the limitations of existing therapies?
- What is the patient's share of Down Syndrome?
Reasons to Buy
- The report will help develop business strategies by understanding the latest trends and changing treatment dynamics driving Down Syndrome.
- Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis ranking of class-wise potential current and emerging therapies under the analyst view section to provide visibility around leading classes.
- Highlights of access and reimbursement policies of current therapies, barriers to accessibility of expensive off-label therapies, and patient assistance programs.
- To understand Key Opinion Leaders’ perspectives around the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
Get insights into our blog on "Which pipeline therapy has the potential to revolutionize the Down Syndrome Market?"