Endotracheal Tube Market Summary
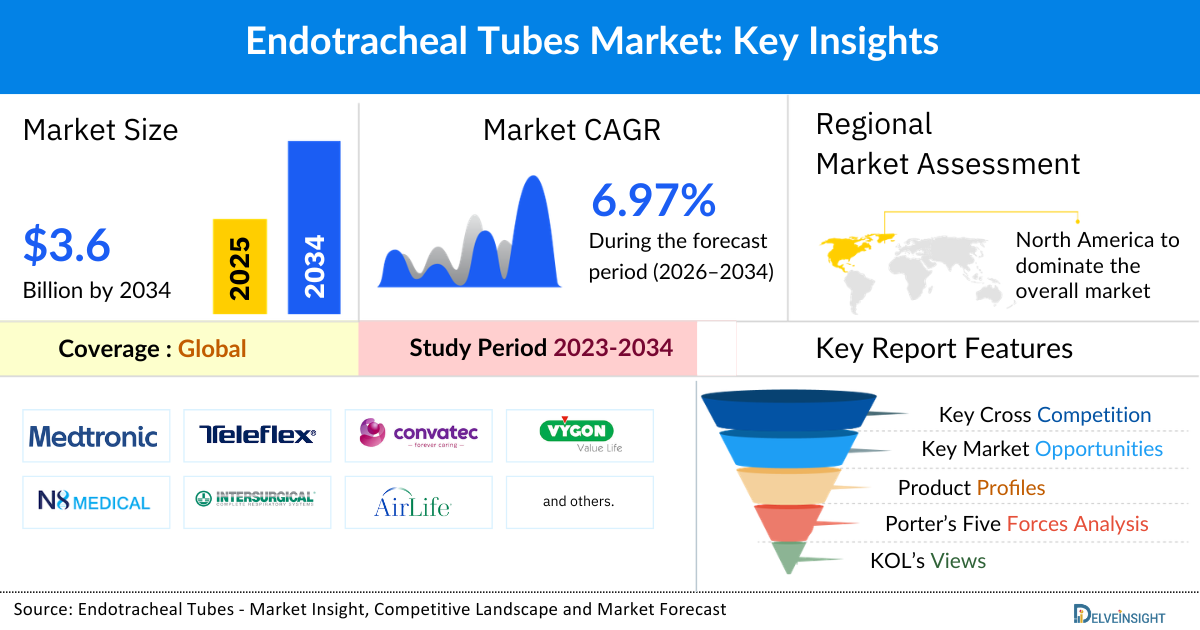
- The global endotracheal tubes market size is expected to increase from USD 2,021.76 million in 2025 to USD 3,691.85 million by 2034, reflecting strong and sustained growth.
- The global endotracheal tubes market is growing at a CAGR of 6.97% during the forecast period from 2026 to 2034.
- The market of endotracheal tubes is being primarily driven by the increasing prevalence of chronic respiratory disorders and cancer cases, the growing number of surgical procedures and ICU admissions, and the rising NICU and PICU admissions are collectively driving strong growth in the endotracheal tubes (ETTs) market by substantially increasing the demand for reliable airway management across all age groups. Chronic respiratory diseases and cancer often lead to respiratory failure or complex surgical interventions, requiring prolonged intubation and mechanical ventilation, while the rising volume of major surgeries and critical care admissions further elevates routine ETT usage in operating rooms and ICUs. At the same time, higher NICU and PICU admissions driven by preterm births, low birth weight, and congenital disorders are expanding demand for specialized neonatal and pediatric ETTs. Together, these factors are increasing both procedure volumes and per-patient ETT utilization, reinforcing the essential role of endotracheal tubes in acute, surgical, and critical care settings and accelerating overall market expansion.
- The leading companies operating in the endotracheal tubes market include Medtronic, Teleflex Incorporated, ConvaTec, Inc., Vygon, N8 Medical, LLC, Novo Klinik-Service GmbH, Intersurgical Ltd., AirLife, Angiplast Pvt Ltd., Guangzhou Orcl Medical Co.,Ltd., Sumi, Van Oostveen Medical B.V., DEAS S.R.L, ICU Medical Inc., Sterimed Group, Respicare., Hangzhou BEVER Medical Devices Co., Ltd., Bryan Medical, Flexicare (Group) Limited, Viggo Medical Devices, and others.
- North America is expected to dominate the overall endotracheal tubes market due to the growing prevalence of chronic respiratory disorders and cancer cases, increased surgical procedures, and rising number of trauma and accident cases have boosted the overall market across the region.
- In the product type segment of the endotracheal tubes market, the reinforced category is estimated to account for the largest market share in 2025.
Request for unlocking the report of the @ Endotracheal Tubes Market Trends
Endotracheal Tubes Market Size and Forecasts
|
Report Metrics |
Details |
|
2025 Market Size |
USD 2,021.76 million |
|
2034 Projected Market Size |
USD 3,691.85 million |
|
Growth Rate (2026-2034) |
6.97% CAGR |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia-Pacific |
|
Market Structure |
Moderately Concentrated |
Factors Contributing to the Growth of the Endotracheal Tubes Market
- Increasing prevalence of chronic respiratory disorders and cancer cases leading to a surge in endotracheal tubes: The rising prevalence of respiratory disorders is a major driver boosting the overall endotracheal tubes (ETT) market, as these conditions often necessitate airway management and mechanical ventilation. Diseases such as chronic obstructive pulmonary disease (COPD), asthma, pneumonia, acute respiratory distress syndrome (ARDS), lung cancer, and sleep apnea can lead to respiratory failure or severe hypoxemia, requiring immediate intervention through intubation of endotracheal tubes.
- Increasing number of surgical procedures and ICU admission esclating the market of endotracheal tubes: The increasing number of surgical procedures and ICU admissions is a major factor boosting the overall endotracheal tubes (ETT) market, as endotracheal intubation is a fundamental requirement in both operative and critical care settings. A growing volume of elective and emergency surgeries including orthopedic, cardiovascular, neurological, gastrointestinal, ENT, and trauma surgeries routinely require general anesthesia, during which endotracheal tubes are essential to secure the airway and ensure controlled ventilation.
- Rising NICU and PICU admissions: Rising admissions to Neonatal Intensive Care Units (NICUs) and Pediatric Intensive Care Units (PICUs) are significantly boosting the overall market for endotracheal tubes (ETTs) due to the growing need for advanced respiratory support in critically ill neonates and children. Premature births, low birth weight infants, neonatal respiratory distress syndrome (RDS), congenital anomalies, birth asphyxia, neonatal sepsis, and pediatric respiratory infections increasingly require invasive airway management and mechanical ventilation, making endotracheal intubation a cornerstone of care in NICUs and PICUs.
Endotracheal Tubes Market Report Segmentation
This endotracheal tubes market report offers a comprehensive overview of the global endotracheal tubes market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Product Type (Standard, Reinforced, Coated, and Others), Cuff Type (Cuffed and Uncuffed/Cuffless), Intubation Route (Oral and Nasal), Age Group (Adults and Pediatrics), End-Users (Hospitals, Ambulatory Surgical Centers, and Others), and geography. The report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing endotracheal tubes market.
Endotracheal tubes are medical devices inserted into the trachea to maintain an open airway and provide ventilation to patients, commonly used during surgery or in emergencies.
The overall market of endotracheal tubes is being collectively boosted by the increasing prevalence of chronic respiratory disorders and cancer cases, the growing number of surgical procedures and ICU admissions, and the rising NICU and PICU admissions are collectively driving strong growth in the endotracheal tubes (ETTs) market by substantially increasing the demand for reliable airway management across all age groups. Chronic respiratory diseases and cancer often lead to respiratory failure or complex surgical interventions, requiring prolonged intubation and mechanical ventilation, while the rising volume of major surgeries and critical care admissions further elevates routine ETT usage in operating rooms and ICUs. At the same time, higher NICU and PICU admissions driven by preterm births, low birth weight, and congenital disorders are expanding demand for specialized neonatal and pediatric ETTs. Together, these factors are increasing both procedure volumes and per-patient ETT utilization, reinforcing the essential role of endotracheal tubes in acute, surgical, and critical care settings and accelerating overall market expansion.
Get More Insights into the Report @ Endotracheal Tubes Market Insights
What are the latest endotracheal tubes market dynamics and trends?
The global market for endotracheal tubes has witnessed significant growth in recent years, largely driven by the increasing prevalence of chronic respiratory disorders and cancer cases, increasing number of surgical procedures and ICU admission, and rising NICU and PICU admissions.
According to the data provided by the World Health Organization (2025), approximately 80 million worldwide population was affected by the chronic respiratory disorders. In critical care settings, patients with exacerbations of chronic or acute respiratory illnesses frequently require short- or long-term mechanical ventilation, directly increasing demand for both standard and specialized ETTs, including cuffed, reinforced, and coated tubes.
Additionally, according to the data provided by the Lung Cancer Today (2025), lung cancer was estimated to represent 11% of all new cancer cases in men and 12% of all new cancer cases in women in 2024 worldwide. Endotracheal tubes are most commonly required in head and neck cancers, lung cancer, esophageal cancer, mediastinal tumors, thyroid cancer, and CNS cancers, as well as during major cancer surgeries across all tumor types. Their role is supportive and life-saving, ensuring airway patency, oxygenation, and safe delivery of anesthesia rather than treating the cancer itself.
Furthermore, according to the study published under National Library of Medicines (2024), estimated that, around 1 million cardiac surgeries were projected to occur each year. Procedures such as coronary artery bypass grafting (CABG), valve repair or replacement, congenital heart defect correction, and aortic surgeries involve long operative durations, cardiopulmonary bypass, and deep anesthesia, all of which necessitate the use of high-performance endotracheal tubes.
However, according to the data provided by the World Health Organization (2023), an estimated 13.4 million babies were born preterm. Preterm birth is a major factor boosting the overall market for endotracheal tubes (ETTs) because premature infants frequently suffer from underdeveloped lungs and immature respiratory control, making invasive airway support essential immediately after birth and during prolonged NICU stays. Conditions such as neonatal respiratory distress syndrome (RDS), apnea of prematurity, bronchopulmonary dysplasia (BPD), and pulmonary infections are highly prevalent among preterm neonates, often necessitating endotracheal intubation and mechanical ventilation.
Thus, the factors mentioned above are expected to boost the overall market of endotracheal tubes during the forecast period from 202 to 2034.
However, the risk of complications associated with the insertion and use of endotracheal tubes (ETTs) represents a key limitation that negatively impacts the overall ETT market, as these complications can directly affect patient safety, clinical outcomes, and clinician preference for airway management approaches. Additionally, the increasing number of product recalls due to safety concerns is also acting as a significant limiting barrier for the endotracheal tubes (ETTs) market by undermining clinician confidence, disrupting hospital procurement cycles, and increasing caution around product adoption.
Endotracheal Tubes Market Segment Analysis
Endotracheal Tubes Market by Product Type (Standard, Reinforced, Coated, and Others), Cuff Type (Cuffed and Uncuffed/Cuffless), Intubation Route (Oral and Nasal), Age Group (Adults and Pediatrics), End-Users (Hospitals, Ambulatory Surgical Centers, and Others), and Geography (North America, Europe, Asia-Pacific, and Rest of the World)
By Product Type: Reinforced Projected to Register Fastest Growth
In the product type segment of the endotracheal tubes market, the reinforced category, is projected to grow at the fastest CAGR of 7.73% during the forecast period from 2026 to 2034. The reinforced endotracheal tube (ETT) category is significantly boosting the overall endotracheal tubes market by addressing the growing demand for secure airway management in complex and high-risk surgical procedures. Reinforced ETTs, also known as armored tubes, are designed with embedded metal or nylon coils that prevent kinking and compression, ensuring uninterrupted airflow even when the patient’s head and neck are positioned at extreme angles.
This makes them particularly valuable in head and neck surgeries, neurosurgical procedures, maxillofacial operations, spine surgeries, and procedures performed in the prone position, where standard tubes are more prone to obstruction.
Additionally, the increasing volume of specialty and minimally invasive surgeries, along with the rising prevalence of conditions requiring complex airway management, has driven higher adoption of reinforced endotracheal tubes in operating rooms and specialized surgical centers.
Furthermore, reinforced ETTs enhance patient safety and surgical efficiency by reducing intraoperative airway complications, minimizing the need for tube repositioning, and lowering the risk of ventilation failure. This clinical reliability has led to strong clinician preference in specific indications, reinforcing consistent procurement.
As healthcare systems continue to expand advanced surgical capabilities and prioritize procedural safety, the reinforced endotracheal tube segment plays a key role in driving premiumization and value-based growth within the overall endotracheal tubes market.
Additionally, the increase in product development activities are further boosting the market of reinforced endotracheal tubes. For instance, in May 2025, Flexicare Medical Limited received FDA clearance for the ProVu Video Stylet and reinforced endotracheal tube system, a sterile, single-use, video-enabled stylet paired with a reinforced intubation tube designed for both oral and nasal intubation in adults. This integrated system aims to assist clinicians in both standard and difficult intubations by offering visualization and directional control during tube placement..
Overall, the factors mentioned above are expected to boost the overall market of reinforced ETTs thereby, escalating the overall market of endotracheal tubes market during the forecast period.
By Cuff Type: Cuffed Category Dominates the Market
The cuffed category in the endotracheal tube segment is expected dominate the market with a market share of 71.56% in 2025 and boosting the overall endotracheal tubes market. Cuffed ETTs provide an effective seal within the trachea, which helps prevent air leakage during mechanical ventilation and reduces the risk of aspiration of gastric contents, blood, or secretions, making them the preferred choice for adult patients and increasingly for pediatric and neonatal patients with appropriately designed low-pressure cuffs.
Additionally, the rising incidence of respiratory disorders such as chronic obstructive pulmonary disease (COPD), asthma exacerbations, pneumonia, acute respiratory distress syndrome (ARDS), and respiratory failure along with the growing number of complex surgical procedures requiring general anesthesia has increased the demand for reliable airway management solutions, directly favoring cuffed ETTs.
However, the expansion of intensive care units (ICUs), growth in emergency and trauma care, and increased use of long-term mechanical ventilation have further strengthened the preference for cuffed tubes, as they enable better ventilation control, accurate delivery of anesthetic gases, and improved monitoring of airway pressures.
Furthermore the technological advancements, including high-volume, low-pressure cuffs, subglottic suction ports, and antimicrobial coatings, have addressed earlier safety concerns such as tracheal injury, enhancing clinician confidence and accelerating adoption. Moreover, updated clinical guidelines and anesthesia practices increasingly recommend cuffed ETTs for both routine and high-risk procedures due to their enhanced safety and efficiency.
Moreover, the increase in product development activities among the key market players are further boosting the overall market of cuffed endotracheal tube market. For instance, in February 2025, the FDA cleared Rusch Endotracheal Tubes of Teleflex, including cuffed and reinforced versions, for oral or nasal intubation and airway management. This clearance covers adult and pediatric cuffed tubes, including reinforced variants that help reduce kinking and improve ventilation reliability across various clinical scenarios.
Collectively, these factors are driving higher utilization rates of cuffed endotracheal tubes across hospitals and surgical centers, making this category a key contributor to the sustained growth of the global endotracheal tubes market.
By Intubation Route: Oral Category Dominates the Market
The oral intubation route category is expected to domainates the market with a market share of 65.09% in 2025 and playing a significant role in boosting the overall endotracheal tube (ETT) market. The oral intubation route category is a major driver of growth in the global endotracheal tubes (ETT) market due to its widespread clinical preference, procedural versatility, and high procedural volumes across healthcare settings.
Oral intubation is the most commonly used route for securing the airway in operating rooms, emergency departments, and intensive care units because it allows rapid, direct access to the trachea, offers better visualization of the vocal cords, and is compatible with a broad range of endotracheal tube designs.
Additionally, the rising number of surgical procedures requiring general anesthesia, increasing trauma and emergency cases, and growing ICU admissions for respiratory failure and critical illnesses are directly translating into high demand for orally inserted endotracheal tubes, making this route the largest contributor to overall market volume.
From a product adoption perspective, oral intubation supports the use of advanced and specialized ETTs, further enhancing market value. For example, Medtronic’s Shiley™ Laser Oral Endotracheal Tube is specifically designed for laser-assisted airway and ENT surgeries performed via the oral route, offering enhanced resistance to laser strikes and reduced risk of airway fires.
The availability of such specialized oral ETTs expands the scope of oral intubation beyond routine anesthesia to high-risk, technology-driven surgical procedures, increasing average selling prices and driving premium segment growth.
Additionally, oral intubation is compatible with reinforced, coated, subglottic suction, and laser-resistant tubes, enabling manufacturers to innovate and diversify product portfolios while maintaining a common insertion route.
Furthermore, the dominance of oral intubation is reinforced by clinical training standards, established protocols, and guideline recommendations, which position it as the first-line airway management approach in most clinical scenarios. Its ease of use, lower complication rates compared to alternative routes, and adaptability across patient populations from pediatrics to adults ensure consistent demand across both developed and emerging healthcare markets.
Collectively, the combination of high procedural frequency, compatibility with advanced tube technologies, and expanding surgical and critical care infrastructure makes the oral intubation route a foundational and growth-sustaining segment that significantly boosts the overall endotracheal tubes market.
By Age Group: Adults Category Dominates the Market
The adults category category is expected to domainates the market with a market share of 84.06% in 2025 and significantly boosting the overall endotracheal tubes market. The adult endotracheal tube category is one of the strongest growth engines in the global endotracheal tubes market because adults represent the largest share of airway management procedures across surgical, emergency, and critical care settings.
Adults account for the majority of hospital intubations due to the high prevalence of respiratory diseases, chronic conditions requiring airway support, and the sheer volume of elective and emergency surgeries performed worldwide.
With an aging global population and an increasing burden of conditions such as COPD, cardiovascular disease, and lung cancer each often necessitating airway protection during surgery or critical illness the demand for adult ETTs continues to rise steadily, making this segment the foundation of overall market volume.
In intensive care units, adult patients frequently require prolonged mechanical ventilation, driving sustained consumption of standard, specialized, and advanced endotracheal tubes. This includes premium innovations designed to reduce complications associated with long-term intubation. For example, CeraShield™ by N8 Medical, Inc. is a biofilm-resistant endotracheal tube developed specifically for adult patients expected to be intubated for more than 24 hours. Its unique antimicrobial coating is engineered to inhibit bacterial adhesion and biofilm formation, addressing major drivers of ventilator-associated respiratory infections and improving patient outcomes in critically ill adults.
Moreover, the adult category benefits from wide clinical acceptance and standardized protocols that prioritize secure airway management for procedures ranging from general anesthesia to trauma resuscitation and ICU respiratory support. Adult ETTs including standard cuffed tubes, reinforced designs, and advanced technologies like antimicrobial or subglottic suction variants are routinely procured in large volumes by hospitals, driving steady baseline revenue and enabling manufacturers to scale production.
As healthcare systems expand surgical capacity and critical care infrastructure worldwide, the robust utilization of adult endotracheal tubes continues to be a key pillar of market growth, ensuring both high unit demand and adoption of increasingly sophisticated product innovations.
By End-Users: Hospitals Category Dominates the Market
Hospitals are the primary growth driver of the global endotracheal tubes market because they represent the central hub for surgical care, emergency treatment, and critical care services, all of which rely heavily on secure airway management. Hospitals conduct a high volume of elective and emergency surgeries that require general anesthesia, where endotracheal intubation is a standard practice to maintain airway patency and ensure controlled ventilation.
In addition, hospitals house intensive care units (ICUs) that manage patients with respiratory failure, sepsis, trauma, neurological disorders, and post-operative complications, many of whom require short- or long-term mechanical ventilation using endotracheal tubes. The continuous expansion of hospital infrastructure, particularly in emerging economies, along with rising investments in ICU beds and advanced respiratory care, is directly increasing the consumption of endotracheal tubes.
Moreover, hospitals are the largest adopters of advanced and specialized endotracheal tube technologies, further boosting market value beyond volume growth. Tertiary and quaternary hospitals increasingly use cuffed, reinforced, coated, subglottic suction, and laser-resistant endotracheal tubes to reduce complications, improve patient safety, and support complex surgical procedures.
Hospitals also follow strict infection-control protocols and quality benchmarks, which encourages the adoption of premium products designed to lower ventilator-associated pneumonia (VAP) and airway trauma.
Centralized procurement systems, bulk purchasing, and long-term supplier contracts ensure consistent demand, while clinician training and standardized intubation protocols reinforce routine usage.
Collectively, the combination of high procedural frequency, ICU dependence, adoption of technologically advanced tubes, and expanding hospital capacity worldwide positions hospitals as the dominant and most influential end-user segment driving sustained growth in the global endotracheal tubes market.
Endotracheal Tubes Market Regional Analysis
North America Endotracheal Tubes Market Trends
North America is expected to account for the highest proportion of 35.69% of the endotracheal tubes market in 2025, out of all regions. This domination is due to the growing prevalence of chronic respiratory disorders and cancer cases, increased surgical procedures, and rising number of trauma and accident cases have boosted the overall market across the region.
As per the data from GLOBOCAN (2025) the projected number of new cases of cancer in the United States was projected to occur 2.54 million, with further projections suggesting an increase to 3.38 million cases by 2045.
The rising number of cancer surgeries, including tumor resections and reconstructive procedures, has increased the routine use of ETTs in operating rooms to maintain a secure airway under general anesthesia. Additionally, cancer patients are more susceptible to respiratory complications, airway obstruction, infections, and treatment-related toxicities from chemotherapy, radiation therapy, or immunotherapy, which often necessitate ICU admission and ventilatory support, further driving endotracheal tube utilization.
Additionally, according to the data provided by the American Lung Association (2025), approximately 11.1 million adults in the United States were living with COPD. Patients with advanced COPD frequently experience acute exacerbations, often triggered by infections or environmental factors, which can result in severe hypoxemia and hypercapnia requiring emergency airway management and invasive mechanical ventilation using endotracheal tubes.
Moreover, leading industry players in the region are further engaged in boosting the overall market of endotracheal tubes. For example, in May 2025, the ProVu Video Stylet with Reinforced Endotracheal Tube was cleared by FDA as a video-enabled intubation system that combines a reinforced ETT with a video stylet. It assists clinicians with oral and nasal intubations and enables visual verification of tube placement, improving accuracy in both routine and difficult airway procedures.
Therefore, the factors mentioned above are expected to significantly propel the growth of the endotracheal tube market in the United States.
Europe Endotracheal Tubes Market Trends
In Europe, the growth of the Endotracheal Tubes market is being strongly driven by the growing prevalence of chronic respiratory disorders, along with the increasing surgical procedures and increasing cases of preterm birth across the region.
According to the data provided by the Government of UK (2025), the rate of emergency admissions for respiratory disease in England increased by 7% from the financial year ending 2023 to the financial year ending 2024.
Additionally, in parallel, the volume of surgeries performed in the UK continues to rise. The British Heart Foundation (2022), reported that over 30,000 adult heart surgeries are performed annually.
During such emergency admissions, patients frequently require intubation and mechanical ventilation, making endotracheal tubes essential medical devices in both ICU and emergency settings.
Furthermore, according to the data provided by the International Agency for research on Cancer (2025), the number of new cases of brain and CNS cancer are projected to occur in 2025 was 5,930 in the United Kingdom and the projection were further estimated to increase 6,970 cases by 2045.
Patients with brain tumors or CNS malignancies often require complex surgeries, such as tumor resections or decompressive procedures, which necessitate general anesthesia and mechanical ventilation. During these procedures, endotracheal intubation is essential to secure the airway, maintain oxygenation, and ensure precise control over ventilation, particularly in cases where intracranial pressure management is critical.
Thus, the factors mentioned are expected to boost the overall market of endotracheal tubes in the United Kingdom during the forecast period.
Asia-Pacific Endotracheal Tubes Market Trends
The Asia Pacific region is emerging as a significant growth driver for the endotracheal tubes market, fueled by a chronic respiratory diseases, an increase in cancer cases, and continuous advancements in medical technology. According to a patient survey conducted by the Japanese Ministry of Health, Labour and Welfare (2023), approximately, 200,000 people in Japan were diagnosed with COPD, representing an estimated prevalence of 0.2% - 0.4%. Additionally, according to the data provided by the GLOBOCAN (2025), in 2025, the estimated new cases of trachea, bronchus, and lung cancer in China was projected to occur 1.14 million with projections further estimating that these new cases would rise to 1.72 million by 2045.
Patients commonly undergo bronchoscopy, lung biopsies, tumor resections, and thoracic surgeries, all of which require secure intubation and mechanical ventilation. In advanced cases, airway obstruction, respiratory failure, and ICU admissions further increase the use of endotracheal tubes.
Thus, the factors mentioned above are expected to boost the overall market of endotracheal tubes in the Asia-Pacific region.
Who are the major players in the endotracheal tubes market?
The following are the leading companies in the endotracheal tubes market. These companies collectively hold the largest market share and dictate industry trends.
- Medtronic
- Teleflex Incorporated
- ConvaTec, Inc.
- Vygon
- N8 Medical, LLC
- Novo Klinik-Service GmbH
- Intersurgical Ltd.
- AirLife
- Angiplast Pvt Ltd.
- Guangzhou Orcl Medical Co.,Ltd.
- Sumi
- Van Oostveen Medical B.V.
- DEAS S.R.L
- ICU Medical Inc.
- Sterimed Group
- Respicare.
- Hangzhou BEVER Medical Devices Co., Ltd.
- Bryan Medical
- Flexicare (Group) Limited,
- Viggo Medical Devices, and others
How is the competitive landscape shaping the endotracheal tubes market?
The competitive landscape of the endotracheal tubes market is shaped by a combination of global medical device leaders and numerous regional players, resulting in a moderately concentrated industry where established firms hold significant market influence but smaller manufacturers also play an important role.
Major multinational companies such as Medtronic, Teleflex Incorporated, Smiths Medical, Becton, Dickinson and Company (BD), and Well Lead Medical are among the frontrunners in the market. These players maintain strong positions through extensive product portfolios that include regular, reinforced, and technologically advanced endotracheal tubes. They leverage global distribution networks, established brand recognition, continuous innovation, and strategic collaborations to secure contracts with large hospitals and healthcare systems in developed regions like North America and Europe.
Beyond these leaders, a diverse set of mid-sized and regional manufacturers including ANGIPLAST, Intersurgical, Fuji Systems, Medline, Mercury Medical, Sterimed, and various Asian producers contribute to market dynamics by catering to local demands, offering cost-competitive products, or focusing on niche segments such as pediatric, reinforced, or antimicrobial-coated tubes. Smaller players often compete on pricing, regional partnerships, and distribution agreements rather than sheer scale.
Competitive strategies in the market emphasize product differentiation and innovation, with firms investing in advanced tube designs (e.g., antimicrobial coatings, subglottic suction ports, video compatibility) to enhance patient outcomes and meet stringent clinical requirements. Mergers, acquisitions, and strategic alliances are also used to expand geographic reach and bolster technological capabilities.
Recent Developmental Activities in the Endotracheal Tubes Market
- In May 2025, Flexicare Medical Limited received FDA clearance for the ProVu Video Stylet and reinforced endotracheal tube system, a sterile, single-use, video-enabled stylet paired with a reinforced intubation tube designed for both oral and nasal intubation in adults.
- In May 2025, the ProVu Video Stylet with Reinforced Endotracheal Tube was cleared by FDA as a video-enabled intubation system that combines a reinforced ETT with a video stylet. It assists clinicians with oral and nasal intubations and enables visual verification of tube placement, improving accuracy in both routine and difficult airway procedures.
- In April 2025, Teleflex LMA® Fastrach™ ETT Single Use received FDA 510(k) clearance as a single‑use endotracheal tube designed for use with the LMA Fastrach airway, providing a standard airway device with atraumatic tip and standard 15 mm connector for clinical intubations.
- In February 2025, the FDA cleared Rusch Endotracheal Tubes of Teleflex, including cuffed and reinforced versions, for oral or nasal intubation and airway management. This clearance covers adult and pediatric cuffed tubes, including reinforced variants that help reduce kinking and improve ventilation reliability across various clinical scenarios.
|
Report Metrics |
Details |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Endotracheal Tubes Market CAGR (2026-2034) |
6.97% |
|
Key Companies in the Endotracheal Tubes Market |
Medtronic, Teleflex Incorporated, ConvaTec, Inc., Vygon, N8 Medical, LLC, Novo Klinik-Service GmbH, Intersurgical Ltd., AirLife, Angiplast Pvt Ltd., Guangzhou Orcl Medical Co.,Ltd., Sumi, Van Oostveen Medical B.V., DEAS S.R.L, ICU Medical Inc., Sterimed Group, Respicare., Hangzhou BEVER Medical Devices Co., Ltd., Bryan Medical, Flexicare (Group) Limited, Viggo Medical Devices, and others. |
|
Endotracheal Tubes Market Segments |
by Product Type, by Cuff Type, by Intubation Route, by Age Group, by End-Users, and by Geography |
|
Endotracheal Tubes Regional Scope |
North America, Europe, Asia Pacific, Middle East, Africa, and South America |
|
Endotracheal Tubes Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Endotracheal Tubes Market Segmentation
- Endotracheal Tubes by Product Type Exposure
- Standard
- Reinforced
- Coated
- Others
- Endotracheal Tubes Cuff Type Exposure
- Cuffed
- Uncuffed/Cuffless
- Endotracheal Tubes Intubation Route Exposure
- Oral
- Nasal
- Endotracheal Tubes Age Group Exposure
- Adults
- Pediatrics
- Endotracheal Tubes End-Users Exposure
- Hospitals
- Ambulatory Surgical Centers
- Others
Endotracheal Tubes Geography Exposure
- North America Endotracheal Tubes Market
- United States Endotracheal Tubes Market
- Canada Endotracheal Tubes Market
- Mexico Endotracheal Tubes Market
- Europe Endotracheal Tubes Market
- United Kingdom Endotracheal Tubes Market
- Germany Endotracheal Tubes Market
- France Endotracheal Tubes Market
- Italy Endotracheal Tubes Market
- Spain Endotracheal Tubes Market
- Rest of Europe Endotracheal Tubes Market
- Asia-Pacific Endotracheal Tubes Market
- China Endotracheal Tubes Market
- Japan Endotracheal Tubes Market
- India Endotracheal Tubes Market
- Australia Endotracheal Tubes Market
- South Korea Endotracheal Tubes Market
- Rest of Asia-Pacific Endotracheal Tubes Market
- Rest of the World Endotracheal Tubes Market
- South America Endotracheal Tubes Market
- Middle East Endotracheal Tubes Market
- Africa Endotracheal Tubes Market
Endotracheal Tubes Market Recent Industry Trends and Milestones (2022-2026):
|
Category |
Key Developments |
|
Endotracheal Tubes Regulatory Approvals |
Flexicare Medical Limited - ProVu Video Stylet and reinforced endotracheal tube system (FDA), Teleflex - LMA® Fastrach™ ETT (FDA), Teleflex - Rusch Endotracheal Tubes (FDA) |
|
Acquisition in the Endotracheal Tubes Market |
AirLife, formerly SunMed, announced the completion of its acquisition of Avanos Medical’s respiratory health business, including the BALLARD®, MICROCUFF®, and endOclear® product lines and SunMed announced that it had entered into a definitive agreement to acquire Avanos Medical’s respiratory health business, including the BALLARD®, MICROCUFF®, and endOclear® product lines. |
|
Company Strategy |
Smiths Medical (now part of ICU Medical), introduced new taper-cuff designs and advanced markers for improved sealing and reduced tracheal trauma. Medtronic expanded antimicrobial-coated tube series targeting infection control in ICU settings. |
|
Emerging Technology |
Smart & sensor-integrated endotracheal tubes, antimicrobial & advanced coating technologies, ai-assisted & predictive technologies, robotic and autonomous intubation systems, and others |
Impact Analysis
AI-Powered Innovations and Applications:
AI-powered innovations in endotracheal tubes (ETTs) are focused on improving patient safety, intubation accuracy, and ventilation management. Emerging AI applications combine sensor-enabled ETTs with machine-learning algorithms to continuously monitor cuff pressure, tube position, airway pressure, and ventilation patterns, enabling real-time alerts for tube displacement, over-inflation, or airway obstruction. AI is also being integrated with airway management systems to predict difficult intubations, support optimal tube size and placement decisions, and guide cuff inflation based on patient-specific anatomy and physiology. In advanced settings, AI-assisted and robotic intubation platforms use data from cameras, force sensors, and historical cases to improve placement precision and reduce airway trauma, particularly in critical care and emergency scenarios. Overall, AI is transforming ETTs from passive devices into intelligent, decision-support tools that enhance clinical outcomes and workflow efficiency.
U.S. Tariff Impact Analysis on Endotracheal Tubes Market:
A U.S. tariff impact analysis on the endotracheal tubes market shows that broader changes to U.S. trade policy especially higher import duties on medical devices and their components could significantly influence costs, supply chains, and competitiveness within the sector. Recent policy moves have introduced baseline tariffs on imported medical products (e.g., a universal 10% duty with higher region-specific rates) and proposals for even steeper levies on goods from China and other regions, potentially driving up the landed cost of critical devices that are manufactured abroad. Because a substantial portion of medical devices and supplies used in the U.S. healthcare system is imported, tariffs can increase production and procurement costs, which manufacturers may pass on to hospitals and payors, leading to higher prices for endotracheal tubes and related airway management products. This cost pressure is compounded by potential supply chain disruptions as companies seek alternative suppliers or relocate production to tariff-friendly regions to mitigate duty burdens, a process that is both time-intensive and subject to regulatory approvals such as FDA clearance. Moreover, uncertainty around tariff policy can dampen investment in R&D and complicate long-term planning, particularly for firms dependent on globally sourced components. Collectively, these tariff dynamics suggest the endotracheal tubes market in the U.S. could face increased prices, supply realignments, and strategic shifts toward domestic manufacturing or diversified sourcing to navigate tariff risks.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
- Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges.
- Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies.
Startup Funding & Investment Trends:
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
University of Nottingham, UK |
£1.1M |
iTraXS Project |
- |
Smart fibre-optic sensor-equipped endotracheal tube with integrated pressure & placement monitoring aimed to prevent cuff injury and improve placement accuracy. |
|
Surgicure Technologies |
$1.785 million |
Horseshoe™ |
Seed |
An innovative airway management device designed to secure the endotracheal tube more reliably than traditional tape methods, prevent unintended extubations, reduce facial pressure injuries, and improve oral care. |
Key takeaways from the Endotracheal Tubes market report study
- Market size analysis for the current endotracheal tubes market size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the endotracheal tubes market.
- Various opportunities available for the other competitors in the endotracheal tubes market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current endotracheal tubes market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the endotracheal tubes market growth in the future?