Febrile Neutropenia Market Summary
Febrile Neutropenia Insights and Trends
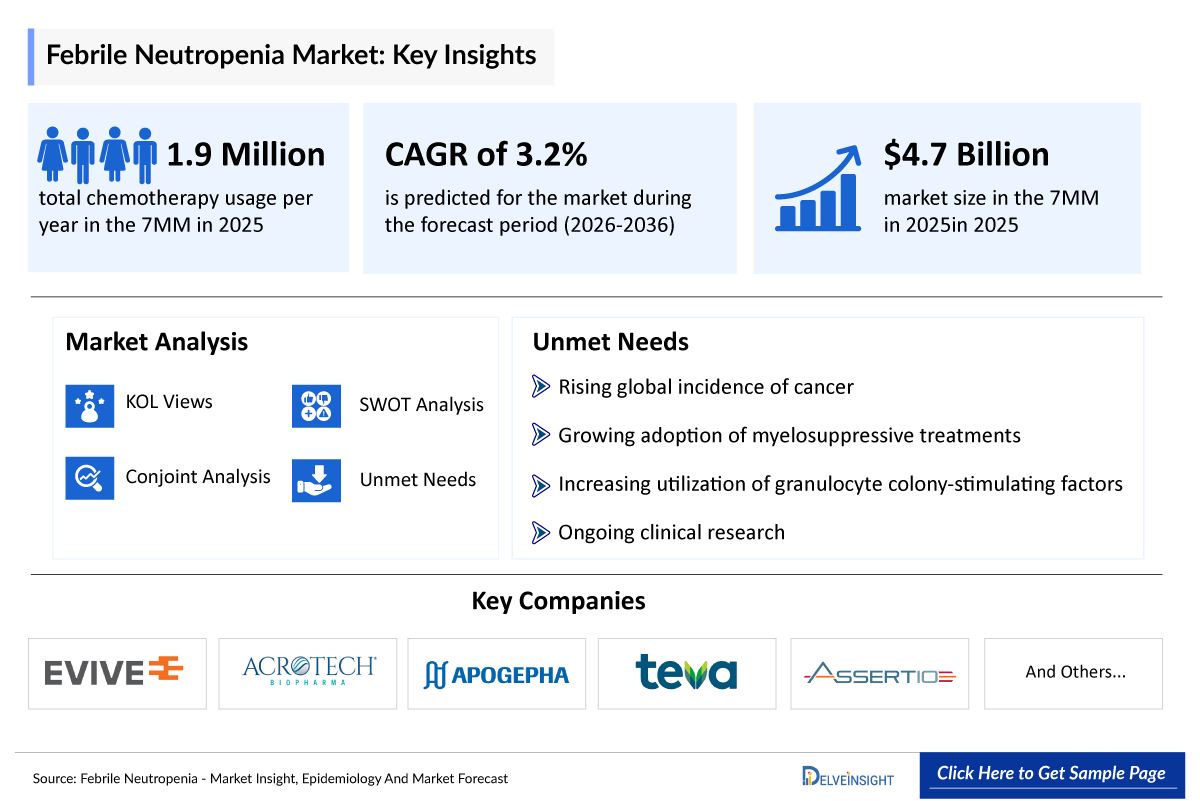
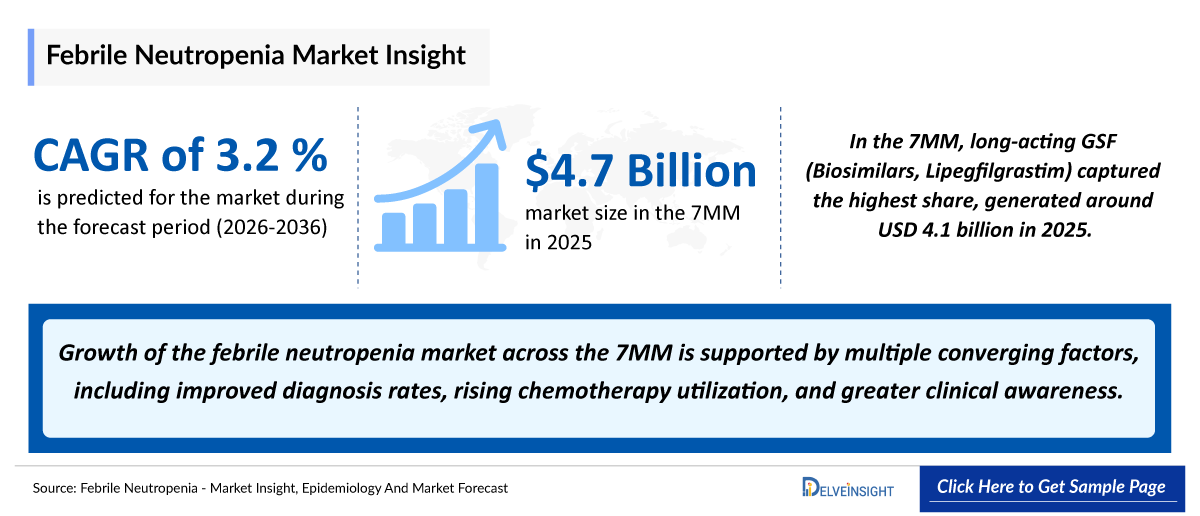
- The market size of febrile neutropenia in the 7MM is 4,750 million in 2025. The febrile neutropenia market is expected to experience continued expansion, driven by increasing cancer cases & chemotherapy treatment volumes, rising awareness of neutropenia risks, and the development of outpatient cancer care, entry & expansion of low-cost biosimilars and new emerging therapies.
- Febrile neutropenia shows a higher incidence rate in hematological malignancies than in solid tumors; however, the overall number of cases of febrile neutropenia is greater in solid tumors due to their higher patient population.
- Chemotherapy-induced neutropenia (CIN) and febrile neutropenia are common and potentially life-threatening complications of myelotoxic chemotherapy, particularly with regimens carrying a high risk or an intermediate risk in patients with additional risk factors.
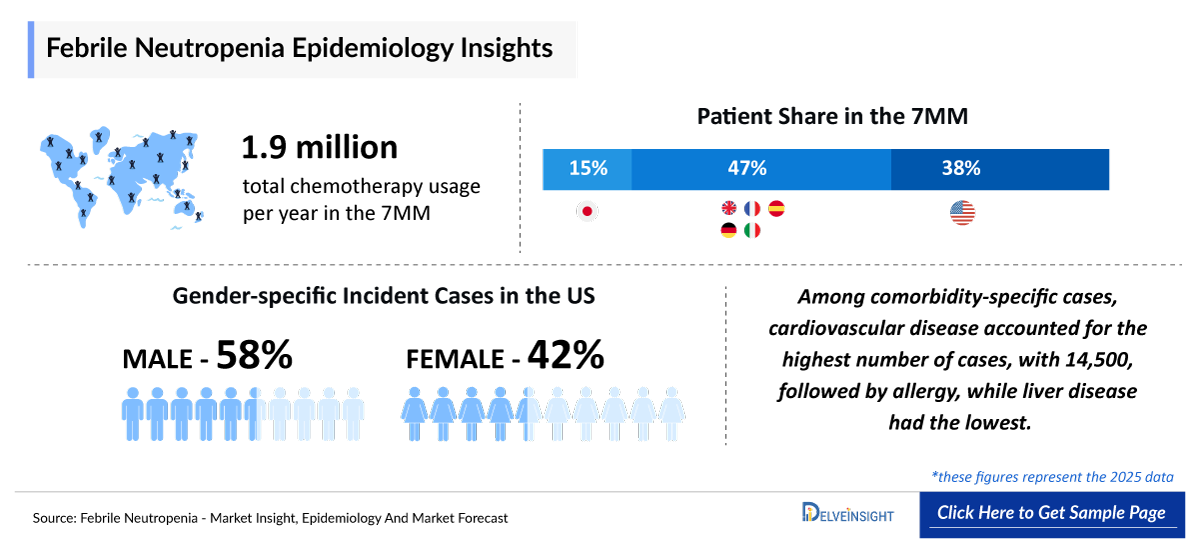
- In the US, the comorbidity-specific rate of febrile neutropenia is highest among patients with cardiovascular disease, followed by those with allergies and lung disease, indicating a greater burden in cardiovascular comorbid conditions.
- Febrile neutropenia is managed as a medical emergency with immediate empiric broad-spectrum antibiotics. Supportive measures such as Granulocyte-Colony Stimulating Factor (G-CSF) agents (Filgrastim, Pegfilgrastim, Lenograstim, Tbo-filgrastim, Lipegfilgrastim, and others) may be used in selected cases based on patient risk status.
- The febrile neutropenia rate observed in this real-world setting aligns closely with clinical trial findings, indicating consistency between controlled and routine practice outcomes.
- Delayed market entry of key supportive care therapies in Japan, driven by stringent regulatory requirements, has resulted in later adoption of globally established treatments such as piperacillin–tazobactam (ZOSYN) and pegfilgrastim (G-LASTA), influencing the pace of alignment with international febrile neutropenia management standards.
- Emerging pipeline development is constrained by regulatory delays and limited late-stage assets. Plinabulin (BeyondSpring Pharmaceuticals) has faced approval delays since 2021 due to insufficient data, while the pipeline remains sparse with AKT-011 (Akthelia Pharmaceuticals) in preclinical and SER-155 (Seres Therapeutics) in Phase I, indicating slower innovation and near-term growth challenges.
Febrile Neutropenia Market size and forecast in the 7MM
- 2025 Febrile Neutropenia Market Size: USD 4,750 million
- 2036 Projected Febrile Neutropenia Market Size: USD 6,700 million
- Febrile Neutropenia Growth Rate (2026–2036): 3.2% CAGR
DelveInsight's ‘Febrile Neutropenia Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Febrile Neutropenia, historical and forecasted epidemiology, as well as the Febrile neutropenia market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Febrile neutropenia market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Febrile neutropenia patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Febrile neutropenia and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Febrile neutropenia Market CAGR (Study period/Forecast period) |
3.9% (2025–2036) |
|
Febrile neutropenia Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Febrile neutropenia Companies |
|
|
Febrile neutropenia Therapies |
|
|
Febrile neutropenia Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Alcohol Use Disorder Market
Rising cancer incidence and chemotherapy use
Rising cancer incidence across global markets is a key driver of growth, primarily due to the increasing reliance on chemotherapy as a standard treatment approach. As the number of diagnosed cancer cases continues to grow driven by factors such as aging populations, improved screening programs, and enhanced diagnostic capabilities the overall use of chemotherapy is also expanding. This, in turn, elevates the risk of treatment-related complications such as neutropenia and associated infections, thereby increasing the demand for supportive care therapies and preventive interventions. Consequently, the growing cancer burden and subsequent chemotherapy utilization are significantly contributing to market expansion.
Demand of novel therapy in community oncology clinics with a competitive clinical profile
The growing demand for novel therapies in community oncology clinics is emerging as a significant market driver, driven by the need for treatments that offer a competitive clinical profile in terms of efficacy, safety, and convenience. As a large proportion of cancer patients are managed in community-based settings, there is increasing preference for therapies that are easy to administer, reduce hospital visits, and improve patient outcomes. Additionally, clinicians in these settings seek innovative options that can address unmet needs, minimize treatment-related complications, and enhance overall quality of care. This shift toward adopting advanced and differentiated therapies in community oncology clinics is expected to further accelerate market growth.
Febrile Neutropenia Understanding and Treatment Algorithm
Febrile Neutropenia Overview and Diagnosis
Despite advances in oncology, Febrile Neutropenia remains a significant clinical challenge, associated with high morbidity, mortality, and healthcare costs due to infectious complications driven by impaired immunity and evolving antimicrobial resistance; this underscores the critical need for rapid pathogen identification, timely empiric broad-spectrum therapy, and optimized treatment strategies supported by emerging diagnostics and antimicrobial stewardship approaches.
Effective management of Febrile Neutropenia requires a comprehensive and timely diagnostic approach, beginning with evaluation of prior and latent infections and thorough clinical assessment of common infection sites such as the lungs, skin, mucosa, catheter sites, and major organ systems. Risk stratification tools like PEN-FAST can help guide safe antibiotic selection in patients with reported penicillin allergy. At onset, prompt work-up including clinical examination, blood cultures, laboratory testing, and symptom-directed imaging is essential, with cultures ideally obtained before initiating antibiotics. While advanced diagnostics such as PCR and biomarkers like procalcitonin can aid in pathogen detection, they complement rather than replace standard methods. Imaging, particularly CT scans, should be guided by clinical presentation, as routine chest X-rays have limited sensitivity. In high-risk patients, additional targeted screening for fungal infections and selective use of invasive diagnostics can further support optimal management.
Further details are provided in the report.
Febrile Neutropenia Treatment
Febrile Neutropenia in patients undergoing chemotherapy is a medical emergency requiring rapid risk stratification and immediate intervention, as fever may be the only sign of a life-threatening infection. According to AGIHO guidelines, patients are categorized into high- and standard-risk groups, with high-risk cases (e.g., prolonged neutropenia, comorbidities) requiring urgent empiric intravenous antibacterial therapy within two hours of fever onset. Recommended first-line agents include broad-spectrum antibiotics targeting gram-negative pathogens such as Pseudomonas aeruginosa, with treatment tailored based on local resistance patterns and patient factors. If fever persists beyond 96 hours or in cases of prolonged neutropenia without antifungal prophylaxis, empiric antifungal therapy covering invasive infections like Aspergillosis should be initiated using agents such as amphotericin B formulations, echinocandins, or triazoles, with ongoing reassessment to optimize outcomes and ensure safe continuation of chemotherapy.
Further details related to country-based variations are provided in the report.
Febrile Neutropenia Unmet Needs
The section “Febrile Neutropenia Unmet Needs” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of epidemiological data
- Reimbursement-driven delayed febrile neutropenia access
- Rising antimicrobial resistance in febrile neutropenia
- Gaps in diagnosis and risk stratification in febrile neutropenia and others…..
Note: Comprehensive unmet needs insights in Febrile neutropenia and their strategic implications are provided in the full report.
Febrile Neutropenia Epidemiology
Key Findings from Febrile neutropenia Epidemiological Analysis and Forecast
- The total chemotherapy usage per year in the 7MM was 1,906,000 of which the United States accounted for the largest share; ~36% in 2025. Chemotherapy usage is expected to rise significantly in the coming years due to the increase in cancer incidence, expanding access to oncology care, increased patient demand and broader adoption of chemo in combination with other therapies.
- In 2025, chemotherapy-associated cases accounted for 692,000 and this number is projected to increase to 841,500 by 2036, growing at a CAGR of 1.8% in the US.
- Among the EU4 and the UK, Germany recorded the highest number of incident cases in 2025, accounting for ~24% of the total incident cases across these regions.
- The incidence of febrile neutropenia shows a higher trend among males compared to females, suggesting a relatively greater susceptibility in male patients.
- Febrile neutropenia patients are predominantly in the intermediate-risk category, followed by a considerably low-risk group, with only a small proportion classified as high-risk, reflecting an overall moderate-risk predominance.
Febrile Neutropenia Drug Analysis & Competitive Landscape
The Febrile neutropenia drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Febrile neutropenia treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Febrile neutropenia market.
Approved Therapies for Febrile Neutropenia
Eflapegrastim (ROLVEDON): Spectrum Pharmaceuticals/Assertio Holdings
Eflapegrastim-xnst (ROLVEDON) is a long-acting G-CSF formulated to reduce the risk of infection, as indicated by febrile neutropenia, in adult patients with non-myeloid malignancies receiving myelosuppressive chemotherapy. It is not intended for use in peripheral blood progenitor cell mobilization for hematopoietic stem cell transplantation.
In September 2022, Spectrum Pharmaceuticals announced that the FDA has approved eflapegrastim injection to decrease the incidence of infection, as manifested by febrile neutropenia, in adult patients with non-myeloid malignancies receiving myelosuppressive anti-cancer drugs associated with a clinically significant incidence of febrile neutropenia.
Efbemalenograstim alfa (RYZNEUTA): Evive Biotech/Acrotech Biopharma/APOGEPHA Arzneimittel GmbH
RYZNEUTA (F-627 [efbemalenograstim alfa]) is a recombinant fusion protein designed to manage chemotherapy-induced neutropenia. It comprises two G-CSF molecules linked to a human IgG2-Fc fragment, which extends its half-life comparable to PEGylated G-CSF therapies. Produced in Chinese Hamster Ovary (CHO) cells, it exhibits a mammalian glycosylation profile. The drug acts by binding to the G-CSF receptor, promoting the survival, proliferation, differentiation, and activation of neutrophil precursors and mature neutrophils, thereby enhancing neutrophil production and supporting immune function.
In April 2025, Evive Biotech and I'rom Group announced the start of collaboration to develop efbemalenograstim in Japan. The partnership will focus on advancing a same-day dosing regimen with chemotherapy supported by global clinical data and investigator-led studies showing comparable safety and efficacy to next-day dosing.
|
Competitive Landscape of Marketed Therapies | ||||||
|
Product |
Company |
Mechanism of Action |
Indication |
ROA |
Molecule Type |
Initial Approval |
|
RYZNEUTA [F-627 (efbemalenograstim alfa)] |
Evive Biotech/ Acrotech Biopharma/ APOGEPHA Arzneimittel GmbH |
G-CSF |
Febrile neutropenia |
SC |
Recombinant fusion proteins |
US: 2023 EU: 2024 |
|
Lipegfilgrastim (LONQUEX) |
Teva Pharmaceutical |
G-CSF |
Febrile neutropenia |
SC |
Recombinant fusion proteins |
EU: 2013 |
|
Filgrastim (GRANIX) |
Teva Pharmaceutical |
G-CSF |
Febrile neutropenia |
SC |
Recombinant fusion proteins |
US: 2012 |
|
Eflapegrastim-xnst (ROLVEDON) |
Assertio Holdings (Spectrum Pharmaceuticals) |
G-CSF |
Febrile neutropenia |
SC |
Recombinant fusion proteins |
US: 2022 |
|
Pegfilgrastim (G-LASTA) |
Kyowa Kirin/Terumo Corporation |
G-CSF |
Febrile neutropenia |
SC |
Recombinant fusion proteins |
JP: 2014 |
|
Note: In addition to above-mentioned therapies, several other biosimilars are being utilized. | ||||||
Febrile Neutropenia Pipeline Analysis
Plinabulin: BeyondSpring Pharmaceuticals
Plinabulin is a first-in-class, small-molecule selective immunomodulating microtubule-binding agent (SIMBA) that exerts both anti-cancer and hematopoietic protective effects. Unlike traditional G-CSFs, plinabulin works through a differentiated mechanism by promoting dendritic cell maturation, modulating immune response, and protecting hematopoietic stem/progenitor cells from chemotherapy-induced damage, thereby reducing the incidence of febrile neutropenia.
The drug has completed Phase III clinical trial for the prevention of chemotherapy-induced neutropenia in combination with G-CSF, demonstrating clinical benefit in reducing the duration of severe neutropenia and improving immune recovery in patients receiving myelosuppressive chemotherapy.
Note: Detailed emerging therapies assessment will be provided in the final report.
Febrile Neutropenia Key Players, Market Leaders and Emerging Companies
- Evive Biotech/Acrotech Biopharma/APOGEPHA Arzneimittel GmbH
- Teva Pharmaceutical
- Assertio Holdings (Spectrum Pharmaceuticals)
- Kyowa Kirin/Terumo Corporation)
- BeyondSpring Pharmaceuticals and others
Febrile Neutropenia Drug Updates
- In June 2025, Evive Biotech has signed an exclusive distribution agreement with Lenis farmacevtika to commercialize efbemalenograstim alfa across eight markets, including Slovenia, Croatia, Serbia, and others in the Adriatic region. The partnership aims to expand regional access to innovative oncology supportive care, leveraging Lenis’ strong distribution network across Central and Eastern Europe (Evive Biotech, 2025).
- In November 2022, Evive Biotech, announced that it has entered into a license agreement with Acrotech Biopharma, a New Jersey-based and wholly-owned subsidiary of Aurobindo Pharma USA Inc., to commercialize RYZNEUTA in the US.
Drug Class Insights
Febrile Neutropenia Market Outlook
Febrile neutropenia is a high-risk clinical condition that reflects a profound disruption of innate immunity following cytotoxic chemotherapy, making even minor infections potentially life-threatening. Its management is fundamentally driven by the principle that infection risk is inversely proportional to neutrophil count and duration of neutropenia, necessitating rapid clinical decision-making. The condition is microbiologically heterogeneous, with a shift from Gram-negative dominance to increasing Gram-positive and opportunistic infections, complicating empirical therapy choices. Early initiation of broad-spectrum agents such as Cefepime or Meropenem is therefore not only therapeutic but also preventive against clinical deterioration. Risk stratification frameworks are central to optimizing resource utilization, enabling tailored treatment intensity while minimizing unnecessary hospitalization. Increasing antimicrobial resistance and the expanding use of intensive cancer regimens continue to add complexity, reinforcing the need for dynamic, guideline-driven management strategies.
Over the past four decades, the understanding and management of febrile neutropenia in cancer patients have evolved substantially from a high-risk, low-control, and reactive care model to a more structured approach. Febrile neutropenia market was dominated by originator biologics such as filgrastim and pegfilgrastim, which benefited from strong patent protection and established clinical efficacy. This period was characterized by limited competition, premium pricing, and restricted access in cost-sensitive regions, as treatment relied heavily on these innovator therapies.
Following the patent expiry of filgrastim (mid-2000s) and pegfilgrastim (late 2010s), the entry of biosimilars significantly transformed the market. Biosimilars introduced price competition, improved accessibility, and expanded treatment adoption across global markets. This shift marked a transition from a high-cost, monopolistic landscape to a more competitive, volume-driven market, with biosimilars now playing a central role in driving growth and access.
According to the estimates, the United States will account for the largest market size of febrile neutropenia by 2036, owing to higher cases and comparatively higher treatment costs, followed by Japan and Germany.
Broader market dynamics are also limiting the commercial upside for legacy short-acting products (as seen with Eflapegrastim (ROLVEDON), with the overall short-acting G-CSF opportunity structurally shrinking over time. Eflapegrastim' net sales were USD 68 million for full-year 2025.
The G-LASTA (BodyPod) on-body injector has supported short-term revenue growth through improved convenience and adherence, but this benefit is expected to be gradually offset by rising biosimilar penetration, which is increasing price pressure and driving long-term revenue decline in Japan’s pegfilgrastim market.
In the febrile neutropenia market, empirical antibiotic therapy dominates as the first-line treatment due to its immediate initiation and survival importance, while targeted therapy is steadily growing with the rise of personalized care, antimicrobial resistance, and increasing febrile neutropenia incidence.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Febrile Neutropenia (2022–2036 Forecast)
The Febrile neutropenia market comprises targeted small molecules, and Recombinant fusion proteins alongside conventional and off-label therapies, each addressing distinct immunologic pathways and mechanisms underlying Febrile neutropenia.
Recombinant fusion proteins: ROLVEDON is a long-acting G-CSF formulated to reduce the risk of infection, as indicated by febrile neutropenia, in adult patients with non-myeloid malignancies receiving myelosuppressive chemotherapy.
Small molecules: Plinabulin is a first-in-class, small-molecule selective immunomodulating microtubule-binding agent (SIMBA) that exerts both anti-cancer and hematopoietic protective effects. It works by modulating immune response, promoting dendritic cell maturation, and protecting hematopoietic cells, thereby reducing febrile neutropenia risk.
Further details will be provided in the report….
Febrile Neutropenia Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Febrile neutropenia drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved Therapies in Febrile Neutropenia
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
The United States
|
US Reimbursement of Therapies Approved for Febrile Neutropenia | |
|
Drug/Therapy |
Access Program |
|
ROLVEDON |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Febrile Neutropenia Therapies Price Scenario & Trends
Pricing and analogue assessment of Febrile neutropenia therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Febrile Neutropenia Approved Drugs
Efbemalenograstim (RYZNEUTA) is administered as a 20 mg SC injection once per chemotherapy cycle. The wholesale acquisition cost (WAC) of USD 4,600 per dose and an assumed 4 chemotherapy cycles per year, the estimated annual treatment cost is USD 18,400.
Industry Experts and Physician Views for Febrile Neutropenia
To keep up with Febrile neutropenia market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the Febrile neutropenia emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Febrile neutropenia, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the University of California, Imperial College London, Hokkaido University, and Cholangiocarcinoma Foundation, etc. were contacted. Their opinion helps understand and validate current and emerging Febrile neutropenia therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Febrile neutropenia.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Febrile neutropenia is a critical, potentially life-threatening complication of cancer treatments that requires aggressive clinical management. While standard-of-care treatments like G-CSFs and broad-spectrum antibiotics remain effective, experts express a strong appetite for novel pipeline therapies that offer improved safety profiles, oral administration, and alternative mechanisms of action.” |
|
Italy |
“Febrile neutropenia is an oncologic emergency, highlighting that prompt initiation of broad-spectrum empiric antibiotics ideally within one hour of fever onset is critical for reducing mortality and improving patient outcomes.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Febrile neutropenia, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided..
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Febrile neutropenia, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Febrile neutropenia market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Febrile neutropenia market.
Report Insights
- Febrile Neutropenia Patient Population Forecast
- Febrile Neutropenia Therapeutics Market Size
- Febrile Neutropenia Pipeline Analysis
- Febrile Neutropenia Market Size and Trends
- Febrile Neutropenia Market Opportunity (current and forecasted)
Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year Forecast
- Febrile Neutropenia Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Febrile Neutropenia Treatment Addressable Market (TAM)
- Febrile Neutropenia Competitve Landscape
- Febrile Neutropenia Major Companies Insights
- Febrile Neutropenia Price Trends and Analogue Assessment
- Febrile Neutropenia Therapies and Drug Adoption/Uptake
- Febrile Neutropenia Therapies Peak Patient Share Analysis
Report Assessment
- Febrile Neutropenia Current Treatment Practices
- Febrile Neutropenia Unmet Needs
- Febrile Neutropenia Clinical Development Analysis
- Febrile Neutropenia Emerging Drugs Product Profiles
- Febrile Neutropenia Market Attractiveness
- Febrile Neutropenia Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Market Insights
- What was the Febrile neutropenia market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Febrile neutropenia?
- What are the disease risks, burdens, and unmet needs of Febrile neutropenia? What will be the growth opportunities across the 7MM concerning the patient population with Febrile neutropenia?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Febrile neutropenia? What are the current guidelines for treating Febrile neutropenia in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Febrile neutropenia market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets withing the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.