helicobacter pylori infections 6mm market
Helicobacter pylori (H. pylori) Insights and Trends
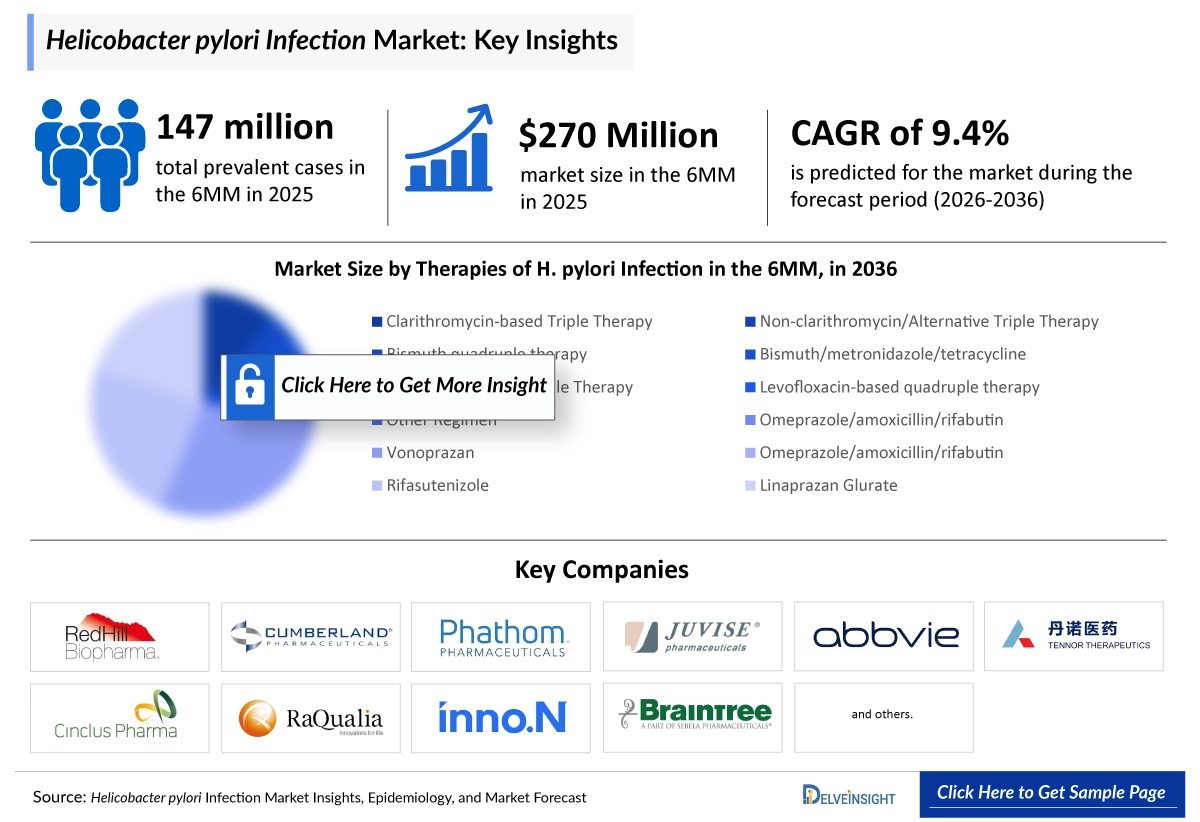
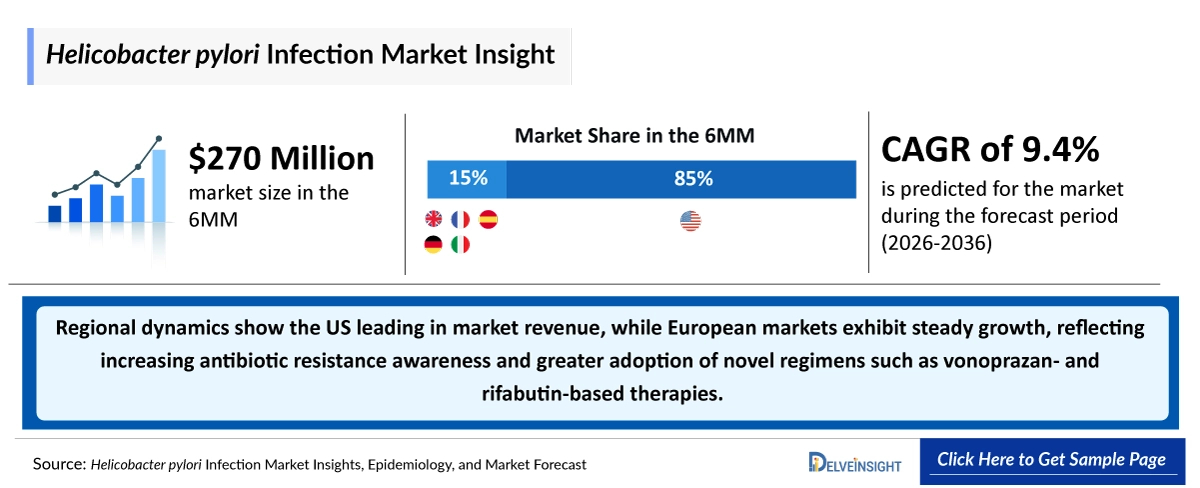
- According to DelveInsight’s analysis, H. pylori infection market size was found to be ~USD 270 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom) in 2025.
- H. pylori infection burden is decreasing overall due to improved hygiene, sanitation, living conditions, and widespread antibiotic use, which have reduced transmission, especially in younger generations. However, prevalence remains high in Spain in both children and adults because of persistent intrafamilial transmission, historical high infection rates in older cohorts, and regional socioeconomic and household crowding factors that continue to support early-life exposure and ongoing spread.
- There is a growing emphasis on "precision medicine" for H. pylori infection, utilizing molecular testing (e.g., PCR) to detect resistance mutations (such as 23S rRNA for clarithromycin) prior to treatment, allowing for tailored therapy rather than empirical guessing.
- The persistence of high eradication failure rates has significant clinical and public health implications, prolonging exposure to H. pylori-associated disease and exacerbating downstream effects of repeated treatment, particularly the emergence of antibiotic resistance.
- The current H. pylori treatment landscape includes key combination therapies such as omeprazole magnesium, amoxicillin, and rifabutin (TALICIA), vonoprazan-based dual and triple regimens (VOQUEZNA DUAL/VOQUEZNA TRIPLE PAK), and bismuth, metronidazole, and tetracycline (PYLERA), reflecting ongoing optimization of eradication strategies and adaptation to resistance patterns.
- While the emerging H. pylori infection pipeline remains limited, with only a few therapies currently in development, including ifasutenizol (TNP-2198; TenNor Therapeutics), Linaprazan glurate (Cinclus Pharma), and Tegoprazan/RQ-00000004 (RaQualia Pharma/HK-inno.N/Braintree).
- A major unmet need in H. pylori infection is the lack of updated epidemiological and antibiotic resistance data, particularly in developed countries, limiting accurate disease burden assessment and hindering optimized prevention and treatment strategies.
Helicobacter pylori (H. pylori) Infection Market Size and Forecast (in the 6MM)
- 2025 H. pylori Infection Market Size: ~USD 270 million
- 2036 H. pylori Infection Market Size: ~USD 720 million
- H. pylori Infection Growth Rate (2026–2036): 9.4% CAGR
DelveInsight's ‘Helicobacter pylori (H. pylori) Infection – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the H. pylori infection, historical and forecasted epidemiology, as well as the H. pylori infection market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom.
The Helicobacter pylori (H. pylori) Infection market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, H. pylori Infection patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in H. pylori Infection and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
H. pylori infection Market CAGR (Forecast period) |
9.4% (2026–2036) |
|
H. pylori infection Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
H. pylori infection Companies |
|
|
H. pylori infection Therapies |
|
|
H. pylori infection Therapies |
Segmented by
|
|
Analysis |
|
Key Factors Driving of the Helicobacter pylori (H. pylori) Infection
- Transition from low-cost generics to premium branded therapies
- Low eradication rates of current SoC create significant market opportunity
- Expanding use of non-invasive diagnostic tests improving accessibility
Helicobacter pylori (H. pylori) Infection Understanding and Treatment Algorithm
Helicobacter pylori (H. pylori) Infection Overview and Diagnosis
Helicobacter pylori (H. pylori) is a spiral-shaped, flagellated, gram-negative, microaerophilic bacterium that infects the stomach lining. The infection is usually acquired in childhood and can persist for years if untreated, often leading to chronic gastritis and peptic ulcer disease, and increasing the risk of gastric cancer and MALT lymphoma.
The diagnosis of active H. pylori infection requires detection of the bacterium and its activity, using noninvasive or invasive methods. Non-invasive tests, primarily the UBT and SAT, are highly sensitive and specific, whereas serologic IgG testing cannot reliably distinguish active from past infection. Invasive approaches, including upper endoscopy with biopsy, provide confirmatory evidence through histology, RUT, culture, or PCR; culture offers definitive specificity and enables antibiotic susceptibility testing, but is technically demanding and less sensitive. Standard clinical practice confirms infection using either two positive noninvasive tests or at least two positive invasive tests (e.g., histology plus urease).
Further details are provided in the report….
Current Helicobacter pylori (H. pylori) Infection Treatment
Treatment of H. pylori infection in children and adolescents aims for high eradication rates and is guided by bacterial susceptibility to clarithromycin (CLA) and metronidazole (MET), as well as the child’s age and weight. For CLA- and MET-susceptible strains, standard first-line triple therapy with a PPI, amoxicillin, and clarithromycin for 14 days is preferred, with clarithromycin replaceable by metronidazole after treatment failure. In areas with high or unknown clarithromycin resistance, optimized bismuth quadruple therapy (PPI, bismuth, tetracycline or amoxicillin in younger children, and metronidazole) is recommended, while alternative regimens include rifabutin-based triple therapy (TALICIA) or vonoprazan–amoxicillin dual therapy (VOQUEZNA). Sequential therapy may be considered for fully susceptible strains but should be avoided in resistant infections. For strains resistant to both CLA and MET, or when susceptibility is unknown, bismuth quadruple therapy is preferred, with rescue therapy guided by susceptibility testing to maximize eradication.
Further details related to country-based variations are provided in the report.
Helicobacter pylori (H. pylori) Infection Unmet Needs
The section “unmet needs of H. pylori Infection” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Increasing treatment resistance and limited effective therapies
- Need for expanded susceptibility testing and surveillance
- Diagnostic limitations and test accessibility
- Stagnation in translational vaccine research
- Scarcity of recent epidemiology data, and others…..
Note: Comprehensive unmet needs insights in H. pylori Infection and their strategic implications are provided in the full report.
Helicobacter pylori (H. pylori) Infection Epidemiology
Key Findings from H. pylori Infection Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, the total diagnosed prevalent cases of H. pylori infection in the 6MM were approximately ~148 million in 2025.
- In 2025, Spain accounted the highest number of diagnosed prevalent cases of H. pylori infection while the UK accounted for the least number of cases of the total diagnosed prevalent population in EU4 and the UK.
- In the US, age-specific trends in H. pylori infection show a clear correlation with increasing age, with prevalence generally rising among older adults. The infection is often acquired in childhood, and prevalence increases across adult age groups, reflects historical exposure and slower decline in older cohorts.
Helicobacter pylori (H. pylori) Infection & Competitive Landscape
The H. pylori Infection drug chapters provide a detailed, market-focused review of approved therapies and the emerging pipeline across Phase III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the H. pylori Infection treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the H. pylori Infection therapeutics market.
Approved Therapies for H. pylori Infection
Omeprazole Magnesium, Amoxicillin, and Rifabutin (TALICIA): RedHill BioPharma/Cumberland Pharmaceuticals
TALICIA is an oral, three-in-one capsule containing omeprazole, a PPI, amoxicillin, a penicillin-class antibacterial, and rifabutin, a rifamycin antibacterial that is indicated for the treatment of H. pylori infection in adults to improve eradication rates in the setting of increasing resistance to traditional clarithromycin-based regimens.
|
Competitive Landscape: Marketed Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Omeprazole magnesium, amoxicillin, and rifabutin (TALICIA) |
RedHill BioPharma/ Cumberland Pharmaceuticals |
H. pylori infection |
Small molecule |
Oral |
Omeprazole magnesium: PPI Amoxicillin: Cell wall synthesis inhibitor Rifabutin: DNA-dependent RNA-polymerase inhibitor |
US: 2019 |
|
Vonoprazan, amoxicillin, clarithromycin (VOQUEZN TRIPLE) and Vonoprazan, amoxicillin (VOQUEZNA DUAL) PAKs |
Phathom Pharmaceuticals |
H. pylori infection |
Small molecule |
Oral |
Potassium-competitive Acid Blocker (PCAB) |
US: 2022 |
H. pylori Infection Pipeline Analysis
Rifasutenizol (TNP-2198): TenNor Therapeutics
Rifasutenizol is an investigational, oral, multi-target antibacterial drug candidate developed by TenNor Therapeutics, designed as a novel rifamycin-nitroimidazole conjugate with a synergistic mechanism of action against H. pylori and other microaerophilic/anaerobic bacteria; it aims to address unmet needs in treating H. pylori infection, including antibiotic-resistant strains, by combining dual pharmacophores in a single molecule. As per the company’s latest update, it plans to initiate a Phase IIb clinical trial in the US in the second half of 2026, followed by a Phase III study. Based on this timeline, DelveInsight assumes Phase III completion by 2029 and, considering ~1 year for regulatory review, the therapy could potentially launch in 2030.
|
Competitive Landscape: Emerging Therapies | |||||||
|
Drug Name |
Company |
Indication |
Highest Phase |
Molecule Type |
RoA |
MoA |
Anticipated Launch in the US |
|
Rifasutenizol (TNP-2198) |
TenNor Therapeutics |
H. pylori infection |
III |
Small molecule |
Oral |
Anti-bacterial (activity against microaerophilic and anaerobic bacterial pathogens) |
2030 |
|
Linaprazan Glurate |
Cinclus Pharma |
H. pylori infection |
I/III |
Small molecule |
Oral |
P-CAB |
Information is available in the full report |
|
Tegoprazan/ RQ-00000004 |
RaQualia Pharma/HK-inno.N/ Braintree (Sebela Pharmaceuticals) |
H. pylori infection |
III |
Small molecule |
Oral |
P-CAB |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
H. pylori Infection Key Players, Market Leaders and Emerging Companies
- RedHill BioPharma
- Cumberland Pharmaceuticals
- Phathom Pharmaceuticals
- Juvisé Pharmaceuticals
- AbbVie
- TenNor Therapeutics
- Cinclus Pharma
- RaQualia Pharma
- HK-inno.N
- Braintree (Sebela Pharmaceuticals), and others
H. pylori Infection Drug Updates
- As per company’s 2026 presentation, RedHill BioPharma has submitted a UK Marketing Authorization Application (MAA) for TALICIA, which is currently under review by the Medicines and Healthcare products Regulatory Agency (MHRA). The company is actively pursuing regulatory approval and is engaging with potential regional partners to support broader commercialization following the anticipated authorization.
- In November 2025, Cinclus Pharma reported that it plans to begin clinical studies on H. pylori in this year, with the expected results available in 2027. It aims to submit applications for regulatory approval to the FDA and EMA in 2027, with the goal of obtaining approval by 2028 or 2029.
- In October 2025, RedHill BioPharma entered into a strategic partnership with Cumberland Pharmaceuticals, which includes a US TALICIA co-commercialization agreement to further enhance commercial activities.
Helicobacter pylori (H. pylori) Infection Market Outlook
The H. pylori infection treatment landscape is undergoing a gradual shift as rising antibiotic resistance challenges traditional eradication regimens based on proton pump inhibitors and standard antibiotics. Recently approved therapies such as TALICIA (rifabutin-based triple therapy) and VOQUEZNA (vonoprazan-based dual and triple regimens) represent key advancements in eradication strategies. TALICIA targets resistant strains through rifabutin’s bactericidal activity by inhibiting bacterial RNA polymerase, while VOQUEZNA introduces a PCAB mechanism that provides stronger and more sustained acid suppression compared to conventional PPIs, thereby improving eradication outcomes and expanding treatment options.
- With advancing candidates such as rifasutenizol (TenNor Therapeutics), the field is evolving toward targeted antibacterial therapies designed to overcome resistant H. pylori strains and address unmet eradication needs.
- Overall, launch of novel eradication therapies, increasing awareness of antibiotic resistance, and improved diagnostic practices are expected to drive steady growth in the 6MM H. pylori infection market from 2022–2036, with strong commercial implications for both marketed products and the emerging pipeline.
- Among the 6MM, the United States accounted for the largest market size of H. pylori infection. i.e., ~USD 230 million in 2025.
- In 2036, among all the therapies for H. pylori Infection, the highest revenue is estimated to be generated by Vonoprazan (VOQUEZNA Dual & Triple Pak), in the United States.
- Regional dynamics show the US leading in market revenue, while European markets exhibit steady growth, reflecting increasing antibiotic resistance awareness and greater adoption of novel regimens such as vonoprazan- and rifabutin-based therapies.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in H. pylori infection (2022–2036 Forecast)
The H. pylori infection market comprises targeted combination therapies, including PPI-based regimens, bismuth-based regimens, and novel dual- or triple-drug formulations, each designed to eradicate the bacterium, overcome antibiotic resistance, and optimize gastric mucosal healing.
Targeted small-molecule therapies: Rifasutenizol (TNP-2198) is an investigational rifamycin-nitroimidazole conjugate that provides synergistic activity against resistant H. pylori strains, highlighting the potential of mechanism-driven oral therapies to address unmet needs in eradication.
Established combination therapies: TALICIA demonstrates high eradication rates in patients with prior treatment failure or resistant strains, validating the clinical and commercial potential of targeted triple therapy.
Further details will be provided in the report….
Helicobacter pylori (H. pylori) Infection Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the H. pylori market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in H. pylori infection is expected to vary based on treatment positioning, mechanism of action, and resistance profiles. Recently introduced or emerging therapies such as TALICIA in the UK are expected to demonstrate relatively faster uptake, supported by its targeted activity against resistant strains, limited competition in the rifabutin-based regimen space, and strong clinical need in patients with prior treatment failure.
In comparison, emerging candidates such as rifasutenizole (TNP-2198) and linaprazan glurate are expected to show moderate uptake trajectories, reflecting their gradual adoption as clinical evidence expands and physicians gain familiarity with these novel mechanisms aimed at improving eradication rates and addressing antibiotic resistance.
Further detailed analysis of emerging therapies' drug uptake will be provided in the report…
Market Access and Reimbursement of Helicobacter pylori (H. pylori) Infection
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
The United States
|
US Reimbursement of Therapies Approved for H. pylori Infection | |
|
Drug/Therapy |
Access Program |
|
TALICIA |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further Details are provided in the final report….
H. pylori Infection therapies Price Scenario & Trends
Pricing and analogue assessment of H. pylori Infection therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of H. pylori Infection Approved Therapies
TALICIA is a fixed-dose, three-drug combination consisting of omeprazole, a proton pump inhibitor; amoxicillin, a penicillin-class antibacterial agent; and rifabutin, formulated as delayed-release capsules. The recommended dosage is four capsules taken three times daily for 14 days. Based on the 2022 WAC of USD 334.32 and the prescribed thrice-daily regimen, the estimated treatment cost of therapy is approximately USD 697.
Industry Experts and Physician Views for H. pylori infection
To keep up with H. pylori Infection market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the H. pylori Infection emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in H. pylori Infection, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the California Northstate University, the University of Occupational and Environmental Health, and Newcastle University, etc. were contacted. Their opinion helps understand and validate current and emerging H. pylori Infection therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in H. pylori infection.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Conducting pre-treatment assessments, such as evaluating for a history of antibiotic allergies or drug intolerances, can prevent complications from H. pylori treatment. Clinicians should educate patients about potential adverse effects, such as nausea, diarrhea, dysgeusia, and fatigue, encourage timely reporting for effective management, and emphasize the importance of medication adherence to prevent antibiotic resistance and treatment failure.” |
|
Germany |
“As resistance patterns evolve, H. pylori management must move toward patient-centric decision-making that balances efficacy, safety, cost, and ease of use. Adjunctive probiotics can mitigate side effects and support adherence, and second-line therapy should be tailored to individual patient factors, including allergies and prior exposure. In refractory cases, repurposed agents such as rifabutin offer a valuable salvage option and may play an increasingly important role in treatment-failure populations.” |
Qualitative Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of H. pylori Infection, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. These pointers are based on the analyst’s discretion and assessment of the patient burden, cost analysis, and existing and evolving treatment landscape.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of H. pylori Infection, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the H. pylori Infection market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 6MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 6MM H. pylori Infection market.
Helicobacter pylori (H. pylori) Infection Report Insights
- Report Insights
- H. pylori Infection Patient Population Forecast
- H. pylori Infection Therapeutics Market Size
- H. pylori Infection Pipeline Analysis
- H. pylori Infection Market Size and Trends
- H. pylori Infection Market Opportunity (Current and Forecasted)
Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year Forecast
- H. pylori Infection Market Outlook (North America, Europe)
- Patient Burden Trends (by geography)
- H. pylori Infection Treatment Addressable Market (TAM)
- H. pylori Infection Competitive Landscape
- H. pylori Infection Major Companies Insights
- H. pylori Infection Price Trends and Analogue Assessment
- H. pylori Infection Therapies Drug Adoption/Uptake
- H. pylori Infection Therapies Peak Patient Share analysis
Report Assessment
- H. pylori Infection Current Treatment Practices
- H. pylori Infection Unmet Needs
- H. pylori Infection Clinical development analysis
- H. pylori Infection Emerging Drugs Product Profiles
- H. pylori Infection Market Attractiveness
- H. pylori Infection Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Market Insights
- What was the H. pylori Infection market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of H. pylori Infection?
- What are the disease risks, burdens, and unmet needs of H. pylori Infection? What will be the growth opportunities across the 6MM concerning the patient population with H. pylori Infection?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of H. pylori Infection? What are the current guidelines for treating H. pylori infection in the US, and Europe?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the H. pylori Infection market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.