IMU-838 Sales Forecast, and Market Size Analysis – 2034
Key Factors Driving IMU-838 Growth
Strong Late-Stage Clinical Program in Multiple Sclerosis
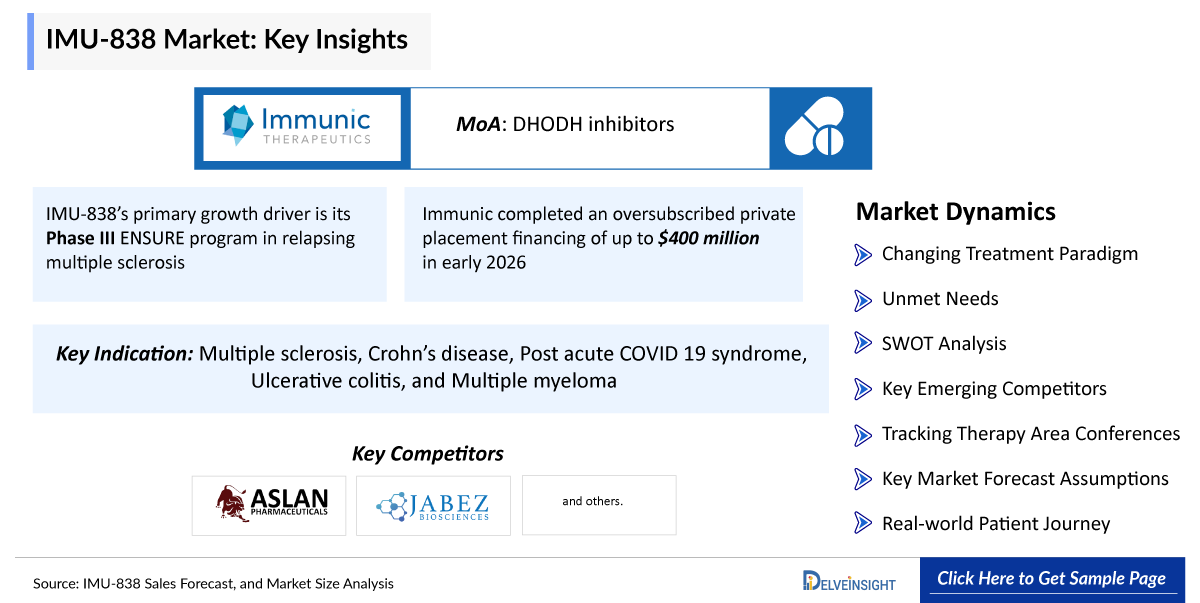
IMU-838’s primary growth driver is its Phase III ENSURE program in relapsing multiple sclerosis (RMS) — twin pivotal trials designed to support regulatory submissions. Both trials have completed enrollment, and top-line Phase III data is expected by the end of 2026.
In addition, the Phase II CALLIPER trial in progressive MS (PMS) demonstrated clinically meaningful reductions in 24-week confirmed disability worsening, including a ~30% reduction in the primary progressive MS subgroup — an endpoint considered key for regulatory approval in PMS.
Differentiated Mechanism of Action
IMU-838 exhibits a dual mechanism: Activation of the Nurr1 transcription factor, providing neuroprotective effects and inhibition of DHODH, producing anti-inflammatory and anti-viral actions.
This combination aims to address both inflammation and neurodegeneration, which are central to MS pathology.
Intellectual Property & Exclusivity
Immunic recently received a notice of allowance for a US patent covering dose strengths of IMU-838 in progressive MS, part of a layered intellectual property strategy likely to extend into the 2040s.
Large Capital Infusions Supporting Development Through Key Milestones
Immunic completed an oversubscribed private placement financing of up to USD 400 million in early 2026, led by major institutional investors, with proceeds earmarked to complete Phase III MS trials and advance toward commercial preparedness.
This includes funding for both RMS and potentially a Phase III study in primary progressive MS, as well as building towards commercialization infrastructure.
Expanding Clinical Footprint Beyond MS
Although MS is the main target, IMU-838 has also been explored in investigator-sponsored trials for post-COVID-19 syndrome and has encouraging early evidence of effect on fatigue symptoms in that setting.
Additionally, Phase II studies in other autoimmune conditions (e.g., ulcerative colitis) and proof-of-concept research in primary sclerosing cholangitis show the molecule’s broader therapeutic potential.
IMU-838 Recent Developments
- In February 2026, Immunic announced the presentation of additional data from its Phase II CALLIPER trial evaluating lead asset, nuclear receptor-related 1 (Nurr1) activator, vidofludimus calcium (IMU-838), in patients with progressive multiple sclerosis (PMS) at the Americas Committee for Treatment and Research in Multiple Sclerosis (ACTRIMS) Forum 2026, taking place in San Diego, California.
- In April 2025, Immunic a biotechnology company developing a clinical pipeline of orally administered, small molecule therapies for chronic inflammatory and autoimmune diseases, announced positive data from its Phase III CALLIPER trial of nuclear receptor related 1 (Nurr1) activator, vidofludimus calcium (IMU-838), in patients with progressive multiple sclerosis (PMS).
“IMU-838 Sales Forecast, and Market Size Analysis – 2034” report provides comprehensive insights of IMU-838 for potential indication like Multiple sclerosis, Crohn’s disease, Post acute COVID 19 syndrome, Ulcerative colitis, and Multiple myeloma in the 7MM. A detailed picture of IMU-838’s existing usage in anticipated entry and performance in potential indications in the 7MM, i.e., the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan for the study period 2020 –2034 is provided in this report along with a detailed description of the IMU-838 for potential indications. The IMU-838 market report provides insights about IMU-838’s sales forecast, mechanism of action (MoA), dosage and administration, as well as research and development including regulatory milestones, along with other developmental activities. Further, it also consists of historical and current IMU-838 performance, future market assessments inclusive of the IMU-838 market forecast analysis for potential indications in the 7MM, SWOT, analysts’ views, comprehensive overview of market competitors, and brief about other emerging therapies in respective indications. It also provides analysis of IMU-838 sales forecasts, along with factors driving its market.
IMU-838 Drug Summary
IMU-838 (vidofludimus calcium) is an investigational, orally administered small-molecule inhibitor of dihydroorotate dehydrogenase (DHODH) developed by Immunic Therapeutics, selectively targeting the de novo pyrimidine biosynthesis pathway in activated, proliferating lymphocytes such as T-cells and B-cells, while sparing resting immune cells that rely on salvage pathways. By inducing metabolic stress, it reduces pro-inflammatory cytokine production (e.g., IL-17A, IL-17F, IFNγ, TNFα, IL-6), inhibits immune cell proliferation, enhances apoptosis in hyperactive lymphocytes, and promotes regulatory macrophages, offering a favorable safety profile with broad anti-inflammatory effects. Currently in Phase III trials like ENSURE 1/2 for relapsing multiple sclerosis (enrollment completed in 2025, top-line data expected end of 2026), it has shown promising efficacy in Phase II studies for progressive MS (reduced disability progression, hints at Nurr1 neuroprotective activation), ulcerative colitis, and other autoimmune conditions, alongside preclinical antiviral activity against SARS-CoV-2 by impairing virus replication in metabolically active infected cells. The report provides IMU-838’s sales, growth barriers and drivers, post usage and approvals in multiple indications.
Scope of the IMU-838 Market Report
The report provides insights into:
- A comprehensive product overview including the IMU-838 MoA, description, dosage and administration, research and development activities in potential indication like Multiple sclerosis, Crohn’s disease, Post acute COVID 19 syndrome, Ulcerative colitis, and Multiple myeloma.
- Elaborated details on IMU-838 regulatory milestones and other development activities have been provided in IMU-838 market report.
- The report also highlights IMU-838‘s cost estimates and regional variations, reported and estimated sales performance, research and development activities in potential indications across the United States, Europe, and Japan.
- The IMU-838 market report also covers the patents information, generic entry and impact on cost cut.
- The IMU-838 market report contains current and forecasted IMU-838 sales for potential indications till 2034.
- Comprehensive coverage of the late-stage emerging therapies for respective indications.
- The IMU-838 market report also features the SWOT analysis with analyst views for IMU-838 in potential indications.
Methodology
The IMU-838 market report is built using data and information sourced primarily from internal databases, primary and secondary research and in-house analysis by DelveInsight’s team of industry experts. Information and data from the secondary sources have been obtained from various printable and nonprintable sources like search engines, news websites, global regulatory authorities websites, trade journals, white papers, magazines, books, trade associations, industry associations, industry portals and access to available databases.
IMU-838 Analytical Perspective by DelveInsight
- In-depth IMU-838 Market Assessment
This IMU-838 sales market forecast report provides a detailed market assessment of IMU-838 for potential indication like Multiple sclerosis, Crohn’s disease, Post acute COVID 19 syndrome, Ulcerative colitis, and Multiple myeloma in the seven major markets, i.e., the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan. This segment of the report provides current and forecasted IMU-838 sales data uptil 2034.
- IMU-838 Clinical Assessment
The IMU-838 market report provides the clinical trials information of IMU-838 for potential indications covering trial interventions, trial conditions, trial status, start and completion dates.
IMU-838 Competitive Landscape
The report provides Insights on competitors and marketed products within the domain, along with a summary of emerging products and their respective launch dates, posing significant competition in the market.
IMU-838 Market Potential & Revenue Forecast
- Projected market size for the IMU-838 and its key indications
- Estimated IMU-838 sales potential (IMU-838 peak sales forecasts)
- IMU-838 Pricing strategies and reimbursement landscape
IMU-838 Competitive Intelligence
- Number of competing drugs in development (pipeline analysis)
- IMU-838 Market positioning compared to existing treatments
- IMU-838 Strengths & weaknesses relative to competitors
IMU-838 Regulatory & Commercial Milestones
- IMU-838 Key regulatory approvals & expected launch timelines
- Commercial partnerships, licensing deals, and M&A activity
IMU-838 Clinical Differentiation
- IMU-838 Efficacy & safety advantages over existing drugs
- IMU-838 Unique selling points
IMU-838 Market Report Highlights
- In the coming years, the IMU-838 market scenario is set to change due to strong adoption, increased prescriptions and broader uptake in multiple immunological indications; which would expand the size of the market.
- The IMU-838 companies are developing therapies that focus on novel approaches to treat/improve the disease condition, assess challenges, and seek opportunities that could influence IMU-838’s dominance.
- Other emerging products for Multiple sclerosis, Crohn’s disease, Post acute COVID 19 syndrome, Ulcerative colitis, and Multiple myeloma are expected to give tough market competition to IMU-838 and launch of late-stage emerging therapies in the near future will significantly impact the market.
- A detailed description of regulatory milestones, and developmental activities, provide the current development scenario of IMU-838 in potential indications.
- Analyse IMU-838 cost, pricing trends and market positioning to support strategic decision-making in the immunology landscape.
- Our in-depth analysis of the forecasted IMU-838 sales data uptil 2034 will support the clients in decision-making process regarding their therapeutic portfolio by identifying the overall scenario of IMU-838 in potential indications.
Key Questions
- What is the class of therapy, route of administration and mechanism of action of IMU-838? How strong is IMU-838’s clinical and commercial performance?
- What is IMU-838’s clinical trial status in each individual indications such as Multiple sclerosis, Crohn’s disease, Post acute COVID 19 syndrome, Ulcerative colitis, and Multiple myeloma and study completion date?
- What are the key collaborations, mergers and acquisitions, licensing and other activities related to the IMU-838 Manufacturers?
- What are the key designations that have been granted to IMU-838 for potential indications? How are they going to impact IMU-838’s penetration in various geographies?
- What is the current and forecasted IMU-838 market scenario for potential indications? What are the key assumptions behind the forecast?
- What are the current and forecasted sales of IMU-838 in the seven major countries, including the United States, Europe (Germany, France, Italy, Spain) and the United Kingdom, and Japan?
- What are the other emerging products available and how are these giving competition to IMU-838 for potential indications?
- Which are the late-stage emerging therapies under development for the treatment of potential indications?
- How cost-effective is IMU-838? What is the duration of therapy and what are the geographical variations in cost per patient?