Iron Deficiency Anemia (IDA) Insights and Trends
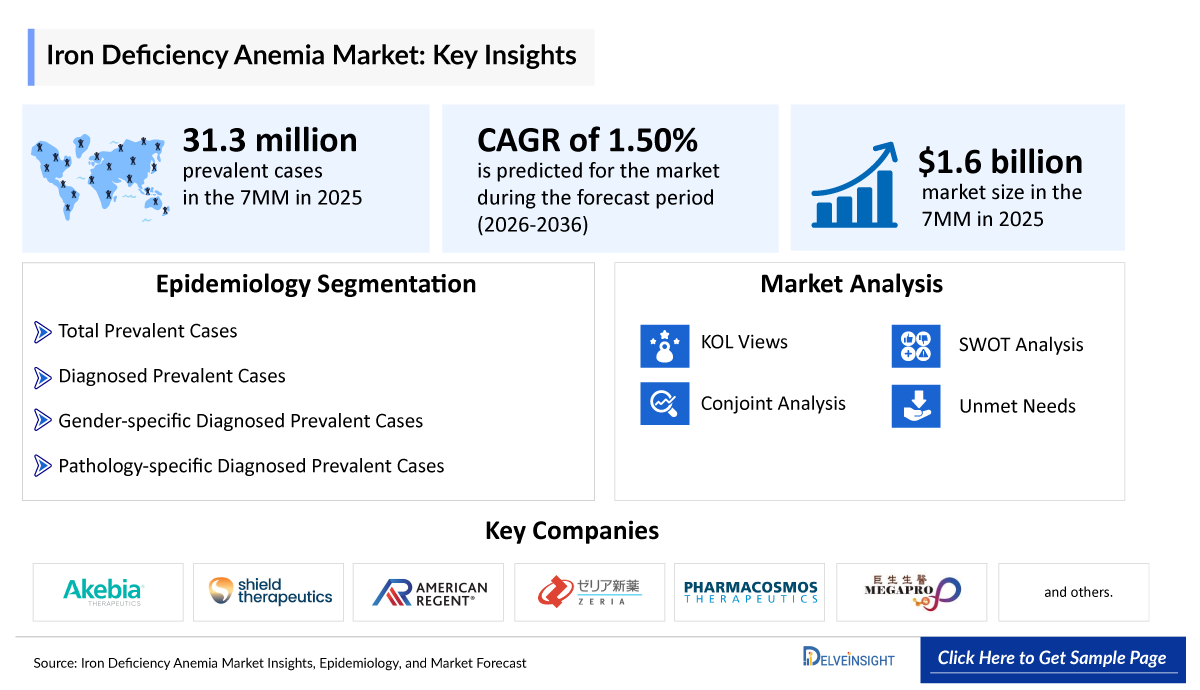
- According to DelveInsight’s analysis, IDA market size was found to be ~USD 1,600 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- A significant proportion of IDA burden is associated with comorbid conditions such as chronic kidney disease, cancer, chronic heart failure, inflammatory bowel disease, celiac disease, and women’s health-related factors, with chronic kidney disease contributing the largest share, highlighting the need for integrated disease management, early screening, and targeted treatment approaches to reduce complications and improve patient outcomes.
- In the IDA market across the 7MM, ferric carboxymaltose (INJECTAFER/FERINJECT) accounts for the largest share among treatment options, driven by its widespread use in moderate-to-severe patients requiring rapid and effective iron repletion.
- The current IDA market is characterized by widespread use of low-cost Oral Ferrous Products (OFPs) such as ferrous sulphate, ferrous fumarate, and ferrous gluconate, alongside a growing shift toward higher-cost advanced therapies including FERACCRU/ACCRUFER, AURYXIA/RIONA, and intravenous iron formulations like INJECTAFER/FERINJECT, and MONOFER, driven by the need for improved efficacy, faster replenishment, and better tolerability in moderate-to-severe patients.
- IDA treatment landscape includes a broad range of marketed iron replacement therapies across oral and intravenous formulations, developed by leading pharmaceutical companies, including Daiichi Sankyo (INJECTAFER/FERINJECT), Pharmacosmos (MONOFERRIC), and Rockwell Medical (TRIFERIC) reflecting continued innovation and competition in the IDA therapeutic space, with a growing focus on improved safety, rapid iron repletion, and patient convenience.
- Whereas the emerging IDA pipeline remains relatively limited, some of the IDA drugs in clinical trials include IDAX (IHAT-02) (Nemysis), MPB-1514 (IOP Injection) (MegaPro Biomedical) are being evaluated.
Iron Deficiency Anemia (IDA) Market Size and Forecast in the 7MM
- 2025 IDA Market Size: ~USD 1,600 million
- IDA Growth Rate (2026–2036): ~1.5% CAGR
DelveInsight's ‘Iron Deficiency Anemia (IDA) – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the IDA, historical and forecasted epidemiology, as well as the IDA market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The IDA market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, IDA patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in IDA and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
IDA Market CAGR (Study period/Forecast period) |
~1.5% (2026–2036) |
|
IDA Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
IDA Companies |
|
|
IDA Therapies |
|
|
IDA Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Iron Deficiency Anemia (IDA) Market
Rising IDA Incidence
The increasing prevalence of IDA, particularly among females, is a primary driver of IDA market expansion. In the US, in 2025, there were ~9,639,000 prevalent cases of IDA.
Rising Opportunities in Targeted Iron Therapies
Emerging evidence of improved efficacy and safety of novel iron formulations over conventional therapies highlights a growing opportunity for drug developers to focus on approaches that enhance iron absorption, optimize utilization, and ensure sustained correction of anemia in IDA.
Emerging IDA Competitive Landscape
Pipeline for ROP is limited. Some of the IDA drugs in clinical trials include IDAX (IHAT-02) (Nemysis), MPB-1514 (IOP Injection) (MegaPro Biomedical), and others.
Iron Deficiency Anemia (IDA) Understanding and Treatment Algorithm
Iron Deficiency Anemia (IDA) Overview and Diagnosis
IDA is the most common form of anemia, occurring when low iron levels reduce hemoglobin production and impair oxygen transport in the body. It can range from mild to severe, with symptoms such as fatigue, weakness, and shortness of breath, especially in advanced stages. IDA is mainly caused by poor iron intake, blood loss, or reduced absorption. Diagnosis is based on blood tests, and treatment typically involves oral or intravenous iron supplementation, with transfusion required in severe cases.
Iron Deficiency Anemia (IDA) Diagnosis
IDA is diagnosed through physical examination and blood tests, often detected during routine checkups. A complete blood count (CBC) is the primary test, showing low hemoglobin, hematocrit, and smaller red blood cells. Additional tests such as serum iron, ferritin, and total iron-binding capacity (TIBC) confirm iron deficiency. If needed, further investigations like stool tests or endoscopy are done to identify underlying causes such as internal bleeding.
Further details are provided in the report.
Iron Deficiency Anemia (IDA) Treatment
The primary goals of IDA treatment are to replenish iron stores, restore hemoglobin levels, alleviate symptoms such as fatigue and weakness, address underlying causes (e.g., blood loss or malabsorption), and prevent recurrence. Management is largely pharmacologic and follows a step-wise approach, with Oral Ferrous Products (OFPs) such as ferrous sulphate, ferrous fumarate, and ferrous gluconate used as first-line therapy due to their accessibility and cost-effectiveness. In patients with intolerance, poor absorption, or inadequate response, alternative oral agents such as FERACCRU/ACCRUFER and AURYXIA/RIONA may be considered. Intravenous iron formulations, including ferric carboxymaltose (INJECTAFER/FERINJECT), and ferric derisomaltose (Monofer) are utilized for rapid repletion in moderate-to-severe cases or in those with chronic conditions such as chronic kidney disease or inflammatory disorders. Supportive strategies, including dietary modification and treatment of underlying etiologies, are essential to ensure sustained hematologic stability and reduce the risk of relapse.
Further details related to country-based variations are provided in the report.
Iron Deficiency Anemia (IDA) Unmet Needs
The section “unmet needs of IDA” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Challenges in accurate and timely diagnosis
- Impacted patient’s quality of life (QoL)
- Pregnancy and effects of IDA
- Issues with existing treatment options, and others…..
Comprehensive unmet needs insights in Iron Deficiency Anemia (IDA) and their strategic implications are provided in the full report.
Iron Deficiency Anemia (IDA) Epidemiology
Key Findings from Iron Deficiency Anemia (IDA) Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, the total prevalent cases of IDA in the 7MM were approximately 31,370,000 in 2025.
- Among the EU4, Germany accounted for the highest number of prevalent cases of IDA, followed by Spain, whereas France accounted for the lowest number of cases in 2025.
- The observed female predominance and earlier onset of IDA highlight the importance of sex-specific screening and early intervention strategies, particularly in recognizing susceptibility patterns and addressing disease at onset to prevent long-term complications.
- The prevalence of anemia was 9.3% in the US population aged 2 years and older. Males had a lower prevalence of anemia than females.
Iron Deficiency Anemia (IDA) Drug Chapters & Competitive Analysis
The IDA drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the IDA treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the IDA therapeutics market.
Approved Therapies for Iron Deficiency Anemia (IDA)
Ferric Citrate (AURYXIA): Akebia Therapeutics
Ferric Citrate (AURYXIA), developed by Akebia Therapeutics (through its subsidiary Keryx Biopharmaceuticals), is an oral, non-calcium, iron-based small molecule with a dual mechanism of action, initially approved by the U.S. FDA in 2014 for hyperphosphatemia in chronic kidney disease and later in 2017 for the treatment of adult patients with IDA. By providing bioavailable iron while also acting as a phosphate binder, AURYXIA supports hemoglobin improvement and iron repletion, particularly in CKD patients, reflecting its differentiated therapeutic profile. In Japan, it is marketed as RIONA by Japan Tobacco and Torii Pharmaceutical, with approval for IDA granted in 2021, highlighting its global clinical utility and lifecycle expansion.
Ferric Maltol (FERACCRU/ACCRUFER): Shield Therapeutics
Ferric Maltol/ ST10 (FERACCRU/ACCRUFER), developed by Shield Therapeutics, is a novel oral iron therapy designed for patients with IDA who are intolerant to conventional oral ferrous products or seek an alternative to intravenous iron. Formulated as a stable ferric maltol complex containing 30 mg of iron, it enables targeted delivery and absorption in the duodenum while minimizing free iron–related gastrointestinal side effects. By remaining tightly bound, unabsorbed iron passes through the digestive system without dissociation, improving tolerability and safety. This mechanism supports effective iron repletion while offering a convenient, well-tolerated, and non-invasive alternative to hospital-based IV therapies, addressing a key unmet need in IDA management.
|
IDA Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Ferric Citrate (AURYXIA) |
Akebia Therapeutics |
In adult patients with CKD not on dialysis. |
Small molecule |
Oral |
Phosphate binding modulator |
US: 2017 Japan: 2021 |
|
Ferric Maltol (FERACCRU/ACCRUFER) |
Shield Therapeutics |
Adults with iron deficiency with or without anemia |
Small molecule |
Oral |
Iron replacement |
US: 2019 Europe: 2016 |
|
Ferric Derisomaltose (MONOFERRIC/ MONOFER) |
Pharmacosmos Therapeutics |
In adult patients: who have intolerance to oral iron or have non-hemodialysis dependent CKD |
Small molecule |
IV infusion |
Iron replacement |
Japan: 2022 US: 2020 Europe: 2009 |
Note: Detailed marketed therapies assessment will be provided in the final report.
Iron Deficiency Anemia (IDA) Pipeline Analysis
IDAX (IHAT-02): Nemysis
IDAX (IHAT-02), developed by Nemysis, HAT-02/IDAX (Nemysis) is a novel oral iron formulation (IHAT) designed as a natural ferritin mimic, enabling efficient iron absorption as intact nanoparticles without requiring gastric solubilization. It closely replicates physiological iron uptake, improves iron deficiency markers, and minimizes gastrointestinal side effects. By preventing the release of free iron in the gut, it helps preserve the microbiome while maintaining normal iron homeostasis after absorption.
Comparison of Emerging Drugs Under Development | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
IDAX (IHAT-02) |
Nemysis |
III |
Iron deficiency Anemia |
Oral |
Ferritin-mimetic iron nanoparticle absorbed intact without releasing free iron. |
Nanoparticulate ferric iron (Fe(III)) |
Information is available in the full report |
|
MPB-1514* |
MegaPro Biomedical |
II |
Iron Deficiency Anemia |
IV |
PEG-coated iron replacement |
PEGylated iron oxide nanoparticle |
Information is available in the full report |
|
* MegaPro Biomedical had planned to advance the drug into Phase IIb; however, no recent updates have been reported, although the asset continues to be listed in the company’s pipeline. | |||||||
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Note: A detailed emerging therapies assessment will be provided in the final report
Iron Deficiency Anemia (IDA) Key Players, Market Leaders and Emerging Companies
- Akebia Therapeutics
- Shield Therapeutics
- American Regent
- Zeria
- Pharmacosmos
- MegaPro Biomedical
- Nemysis, and others
Iron Deficiency Anemia (IDA) Drug Updates
- In March 2026, Shield Therapeutics announced that the European Medicines Agency (EMA), via Committee for Medicinal Products (CHMP) for Human Use, issued a positive opinion to extend the indication of FERACCRU to adolescents, now indicated for iron deficiency in patients ≥12 years, with all Pediatric Investigation Plan requirements fulfilled.
- In Dec 2025, the US FDA approved label expansion of ACCRUFER to include adolescents, now indicated for iron deficiency in patients ≥10 years.
- In November 2025, Nemysis Limited announced a Subscription Agreement under which Kolinpharma, via its parent IGEA Holding, invested in Nemysis’ capital.
Drug Class Insights
Iron Deficiency Anemia (IDA) Market Outlook
The IDA market is undergoing a notable transformation, moving beyond traditional oral iron salts and conventional supplementation toward more advanced, targeted, and patient-friendly therapies. The growing adoption of novel formulations such as ferric maltol and innovative approaches like nanoparticle-based iron delivery systems (e.g., IHAT) reflects a shift toward improved absorption, reduced gastrointestinal side effects, and enhanced patient compliance. These advancements validate mechanism-driven strategies focused on optimizing iron bioavailability and tolerability, while strengthening clinical and regulatory confidence in next-generation iron therapies and reshaping the competitive landscape.
With the continued development of novel agents, including intravenous iron formulations such as ferric carboxymaltose and pipeline candidates targeting improved safety and efficacy profiles, the IDA treatment landscape is evolving similarly to other high-burden chronic conditions, where innovation drives broader therapeutic expansion. The Japan remains the largest IDA market, supported by higher diagnosis rates, better access to advanced therapies, strong awareness, and faster adoption of novel treatments compared with the EU4, the United Kingdom, and the US.
Overall, the introduction of differentiated therapies, expanding treatment options across oral and intravenous segments, improved screening practices, and rising awareness particularly among high-risk populations such as women and patients with chronic diseases—are expected to drive steady growth in the 7MM IDA market from 2022–2036, with significant commercial opportunities for both approved products and emerging pipeline candidates.
- According to the estimates, the largest market size of IDA was captured by the United States, i.e., ~USD 1,300 million in 2025.
- The IDA market is evolving as treatment focus shifts from conventional oral ferrous products (e.g., ferrous sulphate, ferrous fumarate, and ferrous gluconate) toward more advanced and better-tolerated therapies, with novel oral agents such as FERACCRU/ACCRUFER and intravenous formulations including INJECTAFER/FERINJECT, and MONOFER driving adoption due to improved efficacy, tolerability, and broader use across patient populations.
- Regional market dynamics show the US leading in revenue, while Japan and EU markets demonstrate steady growth, reflecting increasing diagnosis rates, broader pediatric access, and rising adoption of novel mechanisms.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Iron Deficiency Anemia (IDA) (2022–2036 Forecast)
The IDA market comprises conventional oral iron therapies, novel oral agents, intravenous iron formulations, and supportive/non-drug products, each addressing distinct aspects of iron absorption, bioavailability, and tolerability in iron deficiency anemia.
- Conventional oral iron therapies: Agents such as ferrous sulphate, ferrous fumarate, and ferrous gluconate remain widely used first-line treatments, providing cost-effective iron replenishment but often limited by gastrointestinal side effects and poor adherence.
- Novel oral agents: FERACCRU / ACCRUFER represent next-generation oral therapies designed to enhance iron absorption and tolerability, validating the shift toward mechanism-driven formulations that improve patient compliance and expand use in broader populations.
- Intravenous iron therapies: Products such as INJECTAFER / FERINJECT, and MONOFER deliver rapid and efficient iron repletion, particularly in patients with severe deficiency, chronic diseases, or intolerance to oral therapies, reinforcing their role as key disease-management options.
- Supportive and non-drug products: Nutritional supplements and adjunctive therapies support iron restoration and maintenance, particularly in mild-to-moderate cases and preventive care settings.
Intravenous and novel oral therapies together define the core innovation landscape in IDA, with IV formulations well established clinically and commercially, while improved oral agents and emerging technologies are driving next-generation market growth.
Further details will be provided in the report….
Iron Deficiency Anemia (IDA) Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the IDA market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in IDA is expected to vary across oral iron formulations, intravenous (IV) iron therapies, and emerging novel agents. Recently approved or widely used IV iron therapies such as ferric carboxymaltose and ferric derisomaltose demonstrate moderate to high uptake, while newer agents like ferric pyrophosphate citrate (TRIFERIC) and investigational products are anticipated to show progressive uptake, supported by improved safety profiles, convenience of administration, and rapid replenishment of iron stores. Their adoption is likely to be driven by the limitations of conventional oral iron therapies, including poor gastrointestinal tolerability, low adherence, and delayed efficacy, particularly in patients with chronic conditions or severe anemia. In comparison, other next-generation therapies and novel delivery approaches are expected to witness gradual but steady uptake as clinical evidence expands and these agents are increasingly incorporated into treatment guidelines across diverse IDA patient populations.
Further detailed analysis of emerging therapies' drug uptake in the report…
Market Access and Reimbursement of IDA
The US Reimbursement for IDA Therapies | |
|
Drug |
Access Program |
|
Ferric Citrate (AURYXIA) |
Centers for Medicare & Medicaid Services, or CMS CoPay Program Patient Assistance Program (PAP) |
|
Ferric Maltol (FERACCRU/ACCRUFER) |
Northern (NHS) Treatment Advisory Group |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report….
Iron Deficiency Anemia (IDA) Therapies Price Scenario & Trends
Pricing and analogue assessment of IDA therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Further details are provided in the final report….
Industry Experts and Physician Views for Iron deficiency anemia (IDA)
To keep up with IDA market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the IDA emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in IDA, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the Harvard Medical School, University of Nottingham, and Japanese Red Cross Central Blood Institute, etc. were contacted. Their opinion helps understand and validate current and emerging IDA therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in IDA.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“The initial symptoms of IDA are so mild that they are mostly unnoticed, but as the disease progresses in the body, and the signs and symptoms intensify, worsening the anemia.” |
|
Japan |
“IDA diminishes working performance by constraining muscles to depend on anaerobic metabolism in order to greater attain muscle extent in contrast to healthy individuals.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Iron deficiency anemia (IDA), strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of IDA, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the IDA market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM IDA market.
Report Insights
- Iron Deficiency Anemia (IDA) Patient Population Forecast
- Iron Deficiency Anemia (IDA) Therapeutics Market Size
- Iron Deficiency Anemia (IDA) Pipeline Analysis
- Iron Deficiency Anemia (IDA) Market Size and Trends
- Iron Deficiency Anemia (IDA) Market Opportunity (Current and forecasted)
Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year Forecast
- Iron Deficiency Anemia (IDA) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Iron Deficiency Anemia (IDA) Treatment Addressable Market (TAM)
- Iron Deficiency Anemia (IDA) Competitive Landscape
- Iron Deficiency Anemia (IDA) Major Companies Insights
- Iron Deficiency Anemia (IDA) Price Trends and Analogue Assessment
- Iron Deficiency Anemia (IDA) Therapies Drug Adoption/Uptake
- Iron Deficiency Anemia (IDA) Therapies Peak Patient Share analysis
Report Assessment
- Iron Deficiency Anemia (IDA) Current Treatment Practices
- Iron Deficiency Anemia (IDA) Unmet Needs
- Iron Deficiency Anemia (IDA) Clinical Development Analysis
- Iron Deficiency Anemia (IDA) Emerging Drugs Product Profiles
- Iron Deficiency Anemia (IDA) Market Attractiveness
- Iron Deficiency Anemia (IDA) Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Market Insights
- What was the Iron Deficiency Anemia (IDA) market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Iron deficiency anemia (IDA)?
- What are the disease risks, burdens, and unmet needs of Iron deficiency anemia (IDA)? What will be the growth opportunities across the 7MM concerning the patient population with Iron deficiency anemia (IDA)?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Iron deficiency anemia (IDA)? What are the current guidelines for treating Iron Deficiency Anemia (IDA) in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Iron Deficiency Anemia (IDA) market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) ‑ enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.