Mastocytosis Market
Key Highlights
- Mastocytosis is a rare disorder characterized by the abnormal accumulation and activation of mast cells in the skin and/or extracutaneous organs, driven in most cases by activating mutations in the KIT gene, particularly KIT D816V.
- Diagnosis remains complex due to heterogeneous presentation and often requires a combination of clinical assessment, serum tryptase evaluation, and bone marrow examination with KIT mutation analysis.
- While systemic mastocytosis has seen therapeutic advancements with approved targeted agents such as AYVAKIT and RYDAPT, along with a growing pipeline of novel KIT inhibitors, cutaneous mastocytosis remains largely managed through symptomatic treatment, primarily antihistamines and supportive care.
DelveInsight's "Mastocytosis Market Insights, Epidemiology, and Market Forecast-2036" report delivers an in-depth understanding of the Mastocytosis, historical and forecasted epidemiology as well as the Mastocytosis market trends in the United States, EU5 (Germany, Spain, Italy, France, and United Kingdom) and Japan.
The Mastocytosis market report provides current treatment practices, emerging drugs, Mastocytosis market share of the individual therapies, current and forecasted Mastocytosis market Size from 2022 to 2036 segmented by seven major markets. The Report also covers current Mastocytosis treatment practice/algorithm, market drivers, market barriers and unmet medical needs to curate the best of the opportunities and assesses the underlying potential of the Mastocytosis market.
Geography Covered
- The United States
- EU5 (Germany, France, Italy, Spain, and the United Kingdom)
- Japan
Study Period: 2022-2036
Key Factors Driving the Mastocytosis Market
- Increasing Treatment Uptake Driven by Diagnostic Advancements: Historically, many patients remained undiagnosed or were treated symptomatically due to the heterogeneous presentation of the disease. As awareness among healthcare professionals grows and diagnostic procedures (such as serum tryptase evaluation and bone marrow examination) advance, more patients are expected to be accurately identified and enter the treatable pool, expanding the active market.
- Shift Toward Disease-Modifying Therapy in Adult Populations: Systemic mastocytosis predominantly affects adults, particularly those aged 40 and above. The recent approval of targeted agents like AYVAKIT (avapritinib) has marked a significant paradigm shift from purely supportive care to disease-modifying therapy. This transition ensures that the adult patient segment, which carries the highest disease burden, now has access to specialized treatments that target the underlying KIT D816V mutation.
- Improved Tolerability and Safety Profiles of Emerging Inhibitors: While current kinase inhibitors are effective, there is an ongoing need for therapies with better safety profiles. Emerging next-generation tyrosine kinase inhibitors, such as elenestinib (BLU-263), are specifically designed to be non-brain penetrant. This strategic design aims to minimize central nervous system (CNS) penetration, thereby reducing the risk of neurological side effects and allowing patients to remain on therapy longer.
- Orphan Disease Status and Regulatory Incentives: Mastocytosis is a rare disorder, with prevalence rates as low as 1 in 150,000 in certain regions. Because of this rare nature, pharmaceutical companies can leverage regulatory pathways for orphan drugs. For example, the US FDA recently granted a priority review to bezuclastinib for Non-Advanced Systemic Mastocytosis (NonAdvSM) with a PDUFA target date of December 2026, which accelerates market entry and provides a competitive edge.
- High Unmet Need in Specific Clinical Subtypes: While systemic forms have seen therapeutic progress, cutaneous mastocytosis and certain advanced subtypes still face limited specific treatment options. Cutaneous forms account for approximately 8.3% of cases and remain largely managed through antihistamines and supportive care. This creates a significant "first-mover" opportunity for any emerging therapy that can achieve a specific indication for these underserved patient populations
Mastocytosis Market Disease Understanding and Treatment Algorithm
Mastocytosis is a rare disorder characterized by the abnormal accumulation and activation of mast cells in the skin and/or extracutaneous organs, driven in most cases by activating mutations in the KIT gene, particularly KIT D816V. The disease presents as a spectrum broadly classified into cutaneous and systemic forms, each with distinct clinical and epidemiological features.
Cutaneous mastocytosis is more commonly observed in children and is typically confined to the skin, often showing a tendency for spontaneous regression over time. In contrast, systemic mastocytosis predominantly affects adults and involves multiple organs such as the bone marrow, liver, spleen, and gastrointestinal tract. Systemic mastocytosis encompasses a range of subtypes, including indolent and advanced forms, with varying degrees of severity and disease progression.
Mastocytosis Diagnosis
Diagnosis remains complex due to heterogeneous presentation and often requires a combination of clinical assessment, serum tryptase evaluation, and bone marrow examination with KIT mutation analysis.
Mastocytosis Treatment
The systemic mastocytosis market is mainly comprised of three major treatments i.e., RYDAPT (Midostaurin), AYVAKIT (Avapritinib), and Imatinib. In addition to these, supportive treatments are widely used.
Antihistamines are commonly used to manage symptoms such as itching, flushing, and abdominal discomfort. Corticosteroids may be prescribed to reduce inflammation and control severe allergic reactions. Monoclonal antibodies are under investigation for their potential to selectively target mast cells.
Symptom management also includes epinephrine auto-injectors for anaphylaxis. Regular monitoring is essential to manage disease progression and associated complications. Treatment approaches vary significantly depending on disease subtype and severity.
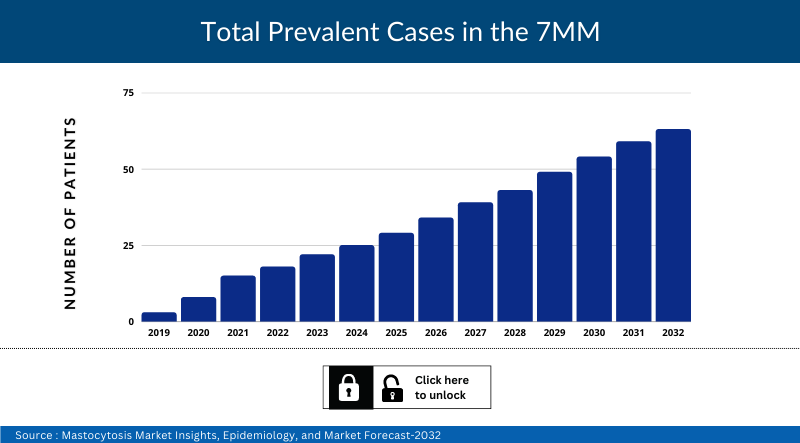
Mastocytosis Epidemiology
The Mastocytosis epidemiology section provides insights about the historical and current Mastocytosis patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the Mastocytosis market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings
The disease epidemiology covered in the report provides historical as well as forecasted Mastocytosis epidemiology scenario in the 7MM covering the United States, EU5 countries (Germany, Spain, Italy, France, and the United Kingdom), and Japan from 2022 to 2036.
- As per the IBM Watson Database, in US, the prevalence of mastocytosis in the general population is 10 per 100,000 individuals.
- According to a study conducted by the Spanish Network on Mastocytosis (REMA), the prevalence of systemic mastocytosis in Spain was estimated at approximately 10.6 per 100,000 individuals in the adult population.
- According to the National Institute for Health and Care Excellence, an article discussing Systemic Mastocytosis indicates that the estimated prevalence of this condition in England is approximately 1 in every 150,000 individuals. This statistic highlights the rarity of Systemic Mastocytosis within the general population, underscoring the need for heightened awareness and understanding of the disease among healthcare professionals.
Mastocytosis Drug Chapters
The drug chapter segment of the Mastocytosis report encloses the detailed analysis of Mastocytosis marketed drugs and late-stage (Phase-III and Phase-II) Mastocytosis pipeline drugs. It also helps to understand the Mastocytosis clinical trial details, expressive pharmacological action, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug and the latest news and press releases.
Mastocytosis Marketed Drugs
AYVAKIT (avapritinib): Blueprint Medicines/Sanofi
In June 2021, AYVAKIT is the first and only medicine approved by the FDA for adult patients with advanced systemic mastocytosis (AdvSM), including patients with aggressive systemic mastocytosis (ASM), systemic mastocytosis with an associated hematological neoplasm (SM-AHN), and mast cell leukemia (MCL), marking a shift in the treatment paradigm from supportive care to disease-modifying therapy. Avapritinib is a kinase inhibitor that is designed to target the KIT D816V mutation and has led to significant improvements in disease symptoms. Most adverse reactions were mild to moderate in severity and included eye edema, dizziness, peripheral edema, and flushing. In May 2023, the US FDA approved AYVAKIT as the first and only treatment for indolent systemic mastocytosis (ISM).
RYDAPT (midostaurin): Novartis
In September 2017, Novartis made a significant announcement regarding the approval of RYDAPT (midostaurin) by the US FDA for use in treating newly diagnosed acute myeloid leukemia (AML) with FLT3 mutations, as well as three distinct forms of systemic mastocytosis. RYDAPT is an oral medication that functions as a multi-targeted inhibitor, specifically designed to block various kinases, including FLT3 and KIT. These kinases play a crucial role in regulating numerous essential cellular processes, and by inhibiting their activity, RYDAPT disrupts the mechanisms that allow cancer cells to proliferate and survive.
Mastocytosis Emerging Drugs
Elenestinib (BLU-263): Blueprint Medicines Corporation/Sanofi
Elenestinib (BLU-263), developed by Blueprint Medicines Corporation, is a novel, investigational, oral, next generation tyrosine kinase inhibitor that is non-brain penetrant. It represents a groundbreaking advancement as a selective tyrosine kinase inhibitor specifically targeting both KIT and platelet-derived growth factor receptor alpha (PDGFRα). Elenestinib is designed for once-daily dosing, elenestinib demonstrates a favorable pharmacokinetic profile with limited central nervous system penetration, which may reduce the risk of neurological side effects commonly associated with other kinase inhibitors.
Bezuclastinib (CGT9486): Cogent Biosciences
Bezuclastinib (CGT9486), developed by Cogent Biosciences, is an investigational oral tyrosine kinase inhibitor that selectively targets mutations in the KIT receptor, particularly the KIT D816V mutation and other exon 17 mutations. This highly selective and potent inhibitor is designed to address the unmet medical needs of patients with these conditions, offering a new treatment option that aims to reduce mast cell burden and improve patient outcomes.
Latest Developments
- In February 2026, Blueprint Medicines reported that the HARBOR (NCT04910685) Phase II/III clinical study of elenestinib, is actively enrolling patients with ISM. The randomized, double-blind, placebo-controlled study includes dose-finding, registrational, and long-term extension phases, with key endpoints evaluating patient-reported symptoms, biomarkers, bone mineral density, anaphylaxis rates, and safety.
- In February 2026, Cogent Biosciences reported updated Phase III SUMMIT trial data for bezuclastinib in non-advanced systemic mastocytosis (NonAdvSM) at the American Academy of Allergy Asthma & Immunology Annual Meeting, demonstrating sustained, statistically significant improvements across endpoints and continued clinical benefit with longer treatment duration.
- In December 2025, Cogent Biosciences reported the submission of a NDA to the US FDA supported by the SUMMIT dataset for NonAdvSM. The company also reported topline APEX trial results in Advanced Systemic Mastocytosis (AdvSM), with NDA submission on track for 1H 2026.
Mastocytosis Market Outlook
The Mastocytosis market outlook of the report helps to build a detailed comprehension of the historic, current, and forecasted Mastocytosis market trends by analyzing the impact of current Mastocytosis therapies on the market, unmet needs, drivers and barriers, and demand for better technology.
This segment gives a thorough detail of Mastocytosis market trend of each marketed drug and late-stage pipeline therapy by evaluating their impact based on the annual cost of therapy, inclusion and exclusion criteria's, mechanism of action, compliance rate, growing need of the market, increasing patient pool, covered patient segment, expected launch year, competition with other therapies, brand value, their impact on the market and view of the key opinion leaders. The calculated Mastocytosis market data are presented with relevant tables and graphs to give a clear view of the market at first sight.
According to DelveInsight, the Mastocytosis market in 7MM is expected to witness a major change in the study period 2022-2036.
Key Findings
This section includes a glimpse of the Mastocytosis market in 7MM.
The United States Market Outlook
This section provides the total Mastocytosis market size and market size by therapies in the United States.
EU-5 Countries: Market Outlook
The total Mastocytosis market size and market size by therapies in Germany, France, Italy, Spain, and the United Kingdom is provided in this section.
Japan Market Outlook
The total Mastocytosis market size and market size by therapies in Japan is also mentioned.
Mastocytosis Drugs Uptake
This section focuses on the rate of uptake of the potential Mastocytosis drugs recently launched in the Mastocytosis market or expected to get launched in the market during the study period 2022-2036. The analysis covers Mastocytosis market uptake by drugs; patient uptake by therapies; and sales of each drug.
Mastocytosis Drugs Uptake helps in understanding the drugs with the most rapid uptake, reasons behind the maximal use of new drugs, and allow the comparison of the drugs on the basis of Mastocytosis market share and size which again will be useful in investigating factors important in market uptake and in making financial and regulatory decisions.
Mastocytosis Pipeline Development Activities
The Mastocytosis report provides insights into different therapeutic candidates in Phase II, and Phase III stage. It also analyses Mastocytosis key players involved in developing targeted therapeutics.
Mastocytosis Pipeline Development Activities
The Mastocytosis report covers the detailed information of collaborations, acquisition, and merger, licensing, patent details, and other information for Mastocytosis emerging therapies.
Reimbursement Scenario in Mastocytosis
Approaching reimbursement proactively can have a positive impact both during the late stages of product development and well after product launch. In a report, we take reimbursement into consideration to identify economically attractive indications and market opportunities. When working with finite resources, the ability to select the markets with the fewest reimbursement barriers can be a critical business and price strategy.
KOL- Views
To keep up with current Mastocytosis market trends, we take KOLs and SMEs ' opinion working in the Mastocytosis domain through primary research to fill the data gaps and validate our secondary research. Their opinion helps to understand and validate current and emerging therapies treatment patterns or Mastocytosis market trends. This will support the clients in potential upcoming novel treatment by identifying the overall scenario of the market and the unmet needs.
Competitive Intelligence Analysis
We perform Competitive and Market Intelligence analysis of the Mastocytosis Market by using various Competitive Intelligence tools that include - SWOT analysis, PESTLE analysis, Porter's five forces, BCG Matrix, Market entry strategies etc. The inclusion of the analysis entirely depends upon the data availability.
Scope of the Mastocytosis Market Report
- The report covers the descriptive overview of Mastocytosis, explaining its causes, signs and symptoms, pathophysiology, diagnosis and currently available therapies
- Comprehensive insight has been provided into the Mastocytosis epidemiology and treatment in the 7MM
- Additionally, an all-inclusive account of both the current and emerging therapies for Mastocytosis is provided, along with the assessment of new therapies, which will have an impact on the current treatment landscape
- A detailed review of the Mastocytosis market; historical and forecasted is included in the report, covering drug outreach in the 7MM
- The report provides an edge while developing business strategies, by understanding trends shaping and driving the global Mastocytosis market
Mastocytosis Market Report Highlights
- In the coming years, the Mastocytosis market is set to change due to the rising awareness of the disease, and incremental healthcare spending across the world; which would expand the size of the market to enable the drug manufacturers to penetrate more into the market
- The companies and academics are working to assess challenges and seek opportunities that could influence Mastocytosis R&D. The therapies under development are focused on novel approaches to treat/improve the disease condition
- Major players are involved in developing therapies for Mastocytosis. The launch of emerging therapies will significantly impact the Mastocytosis market
- A better understanding of disease pathogenesis will also contribute to the development of novel therapeutics for Mastocytosis
- Our in-depth analysis of the pipeline assets across different stages of development (Phase III and Phase II), different emerging trends and comparative analysis of pipeline products with detailed clinical profiles, key cross-competition, launch date along with product development activities will support the clients in the decision-making process regarding their therapeutic portfolio by identifying the overall scenario of the research and development activities
Mastocytosis Market Report Insights
- Mastocytosis Patient Population
- Therapeutic Approaches
- Mastocytosis Pipeline Analysis
- Mastocytosis Market Size and Trends
- Mastocytosis Market Opportunities
- Impact of upcoming Mastocytosis Therapies
Mastocytosis Market Report Key Strengths
- 11 Years Forecast
- 7MM Coverage
- Mastocytosis Epidemiology Segmentation
- Key Cross Competition
- Highly Analyzed Market
- Drugs Uptake
Mastocytosis Market Report Assessment
- Current Treatment Practices
- Unmet Needs
- Mastocytosis Pipeline Product Profiles
- Mastocytosis Market Attractiveness
- Market Drivers and Barriers
Key Questions
Market Market Insights:
- What was the Mastocytosis drug class share (%) distribution in 2022 and how it would look like in 2036?
- What would be the Mastocytosis total market size as well as market size by therapies across the 7MM during the forecast period (2022-2036)?
- What are the key findings pertaining to the market across 7MM and which country will have the largest Mastocytosis market size during the forecast period (2022-2036)?
- At what CAGR, the Mastocytosis market is expected to grow by 7MM during the forecast period (2022-2036)?
- What would be the Mastocytosis market outlook across the 7MM during the forecast period (2022-2036)?
- What would be the Mastocytosis market growth till 2036, and what will be the resultant market Size in the year 2036?
- How would the unmet needs affect the market dynamics and subsequent analysis of the associated trends?
Market Epidemiology Insights:
- What are the disease risk, burden, and regional/ethnic differences of the Mastocytosis?
- What are the key factors driving the epidemiology trend for seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What is the historical Mastocytosis patient pool in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What would be the forecasted patient pool of Mastocytosis in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- Where will be the growth opportunities in the 7MM with respect to the patient population pertaining to Mastocytosis?
- Out of all 7MM countries, which country would have the highest prevalent population of Mastocytosis during the forecast period (2022-2036)?
- At what CAGR the patient population is expected to grow in 7MM during the forecast period (2022-2036)?
Current Treatment Scenario, Market Marketed Drugs and Market Emerging Therapies:
- What are the current options for the Mastocytosis treatment in addition to the approved therapies?
- What are the current treatment guidelines for the treatment of Mastocytosis in the USA, Europe, and Japan?
- What are the Mastocytosis marketed drugs and their respective MOA, regulatory milestones, product development activities, advantages, disadvantages, safety and efficacy, etc.?
- How many companies are developing therapies for the treatment of Mastocytosis?
- How many therapies are in-development by each company for Mastocytosis treatment?
- How many are emerging therapies in mid-stage, and late stage of development for Mastocytosis treatment?
- What are the key collaborations (Industry - Industry, Industry - Academia), Mergers and acquisitions, licensing activities related to the Mastocytosis therapies?
- What are the recent novel therapies, targets, mechanisms of action and technologies being developed to overcome the limitation of existing therapies?
- What are the clinical studies going on for Mastocytosis and their status?
- What are the current challenges faced in drug development?
- What are the key designations that have been granted for the emerging therapies for Mastocytosis?
- What are the global historical and forecasted market of Mastocytosis?
Reasons to buy
- The report will help in developing business strategies by understanding trends shaping and driving the Mastocytosis market
- To understand the future market competition in the Mastocytosis market and Insightful review of the key market drivers and barriers
- Organize sales and marketing efforts by identifying the best opportunities for Mastocytosis in the US, Europe (Germany, Spain, Italy, France, and the United Kingdom) and Japan
- Identification of strong upcoming players in the market will help in devising strategies that will help in getting ahead of competitors
- Organize sales and marketing efforts by identifying the best opportunities for Mastocytosis market
- To understand the future market competition in the Mastocytosis market


.png)




