Mavorixafor Sales Forecast Summary
Key Factors Driving Mavorixafor Growth
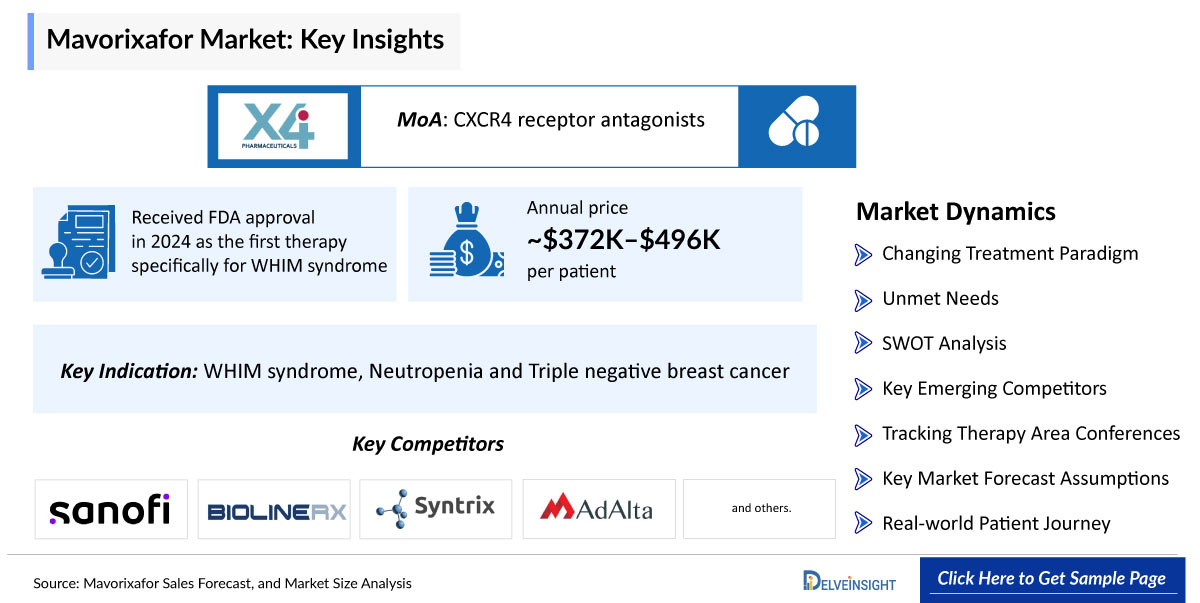
First-in-Class Approval in WHIM Syndrome (Foundational Growth Driver)
- FDA approved in 2024 as the first therapy specifically for WHIM syndrome
- WHIM = rare primary immunodeficiency (~<1,000 patients in the US)
- Monopoly-like position in an ultra-rare disease
- Strong pricing power and rapid uptake among diagnosed patients
Strong Phase III Clinical Efficacy (Highly Differentiated)
- Phase III (4WHIM trial):
- Significant improvement in neutrophil counts (ANC) vs placeb(15.04 hrs vs 2.75 hrs above threshold; p<0.0001)
- Significant lymphocyte improvement (15.8 hrs vs 4.55 hrs; p<0.0001)
- Infection outcomes:
- ~40–60% reduction in infection burden
Premium Pricing in Rare Disease Market
- Annual price:
- ~$372,000–$496,000 per patient
Early Commercial Traction Post-Launch
- US launch: mid-2024
- Revenue:
- $3.5M generated within first ~10 months
- Implication:
- Early uptake is modest (expected for rare disease launches)
- Growth driven by:
- Patient identification
- Physician awareness
Global Expansion (Major Growth Lever)
- EU regulatory progress:
- EMA review accepted (2025) and positive CHMP opinion (2026)
- Commercial partnerships:
- Licensing deals across Europe, Middle East, and other regions
- Impact:
- Expands addressable market beyond U.S.
- Reduces commercialization burden via partners
Expansion intLarger Indications (Key Upside Driver)
Chronic Neutropenia (CN)
- Phase II:
- Durable increases in neutrophil counts
- Phase III (4WARD trial):
- Ongoing, with results expected ~2026
Mavorixafor Recent Developments
- In February 2026, X4 Pharmaceuticals announced the European Medicines Agency’s (EMA) Committee for Medicinal Products for Human Use (CHMP) adopted a positive opinion recommending the granting of marketing authorization, under exceptional circumstances, of mavorixafor for the treatment of WHIM syndrome in the European Union (EU). The positive opinion will now be reviewed by the European Commission (EC) with a final approval decision anticipated in the second quarter of 2026.
“Mavorixafor Sales Forecast, and Market Size Analysis – 2034” report provides comprehensive insights of Mavorixafor for approved indication like WHIM syndrome; as well as potential indication like Neutropenia and Triple negative breast cancer in the 7MM. A detailed picture of Mavorixafor’s existing usage in approved and anticipated entry and performance in potential indications in the 7MM, i.e., the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan for the study period 2020 –2034 is provided in this report along with a detailed description of the Mavorixafor for approved and potential indications. The Mavorixafor market report provides insights about Mavorixafor’s sales forecast, mechanism of action (MoA), dosage and administration, as well as research and development including regulatory milestones, along with other developmental activities. Further, it alsconsists of historical and current Mavorixafor performance, future market assessments inclusive of the Mavorixafor market forecast analysis for approved and potential indications in the 7MM, SWOT, analysts’ views, comprehensive overview of market competitors, and brief about other emerging therapies in respective indications. It alsprovides analysis of Mavorixafor sales forecasts, along with factors driving its market.
Mavorixafor Drug Summary
Mavorixafor is an FDA-approved (April 2024), orally bioavailable, small-molecule antagonist of the CXC chemokine receptor 4 (CXCR4) developed by X4 Pharmaceuticals for the treatment of WHIM syndrome (Warts, Hypogammaglobulinemia, Infections, and Myelokathexis) in adults and pediatric patients 12 years and older. It selectively blocks the binding of the CXCR4 ligand stromal cell-derived factor-1α (SDF-1α/CXCL12), inhibiting receptor activation and downstream signaling tovercome gain-of-function CXCR4 mutations that cause excessive leukocyte retention in the bone marrow, thereby rapidly mobilizing neutrophils, lymphocytes, and monocytes intperipheral circulation timprove neutropenia, reduce infections, and alleviate symptoms like warts. Administered once daily (300 mg for ≥50 kg body weight; 150 mg for <50 kg) without regard tfood, it exhibits nonlinear pharmacokinetics with steady-state reached in 9-12 days, dose-proportional increases in exposure, and a favorable safety profile from Phase 2/3 trials showing sustained ANC elevations (>200 cells/μL), decreased infection rates, and common mild AEs like upper respiratory infections, nausea, and musculoskeletal pain. The report provides Mavorixafor’s sales, growth barriers and drivers, post usage and approvals in multiple indications.
Scope of the Mavorixafor Market Report
The report provides insights into:
- A comprehensive product overview including the Mavorixafor MoA, description, dosage and administration, research and development activities in approved indications like WHIM syndrome; as well as potential indication like Neutropenia and Triple negative breast cancer.
- Elaborated details on Mavorixafor regulatory milestones and other development activities have been provided in Mavorixafor market report.
- The report alshighlights Mavorixafor‘s cost estimates and regional variations, reported and estimated sales performance, research and development activities in approved and potential indications across the United States, Europe, and Japan.
- The Mavorixafor market report alscovers the patents information, generic entry and impact on cost cut.
- The Mavorixafor market report contains current and forecasted Mavorixafor sales for approved and potential indications till 2034.
- Comprehensive coverage of the late-stage emerging therapies for respective indications.
- The Mavorixafor market report alsfeatures the SWOT analysis with analyst views for Mavorixafor in approved and potential indications.
Methodology
The Mavorixafor market report is built using data and information sourced primarily from internal databases, primary and secondary research and in-house analysis by DelveInsight’s team of industry experts. Information and data from the secondary sources have been obtained from various printable and nonprintable sources like search engines, news websites, global regulatory authorities websites, trade journals, white papers, magazines, books, trade associations, industry associations, industry portals and access tavailable databases.
Mavorixafor Analytical Perspective by DelveInsight
- In-depth Mavorixafor Market Assessment
This Mavorixafor sales market forecast report provides a detailed market assessment of Mavorixafor for approved indication like WHIM syndrome; as well as potential indication like Neutropenia and Triple negative breast cancer in the seven major markets, i.e., the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan. This segment of the report provides current and forecasted Mavorixafor sales data uptil 2034.
- Mavorixafor Clinical Assessment
The Mavorixafor market report provides the clinical trials information of Mavorixafor for approved and potential indications covering trial interventions, trial conditions, trial status, start and completion dates.
Mavorixafor Competitive Landscape
The report provides Insights on competitors and marketed products within the domain, along with a summary of emerging products and their respective launch dates, posing significant competition in the market.
Mavorixafor Market Potential & Revenue Forecast
- Projected market size for the Mavorixafor and its key indications
- Estimated Mavorixafor sales potential (Mavorixafor peak sales forecasts)
- Mavorixafor Pricing strategies and reimbursement landscape
Mavorixafor Competitive Intelligence
- Number of competing drugs in development (pipeline analysis)
- Mavorixafor Market positioning compared texisting treatments
- Mavorixafor Strengths & weaknesses relative tcompetitors
Mavorixafor Regulatory & Commercial Milestones
- Mavorixafor Key regulatory approvals & expected launch timelines
- Commercial partnerships, licensing deals, and M&A activity
Mavorixafor Clinical Differentiation
- Mavorixafor Efficacy & safety advantages over existing drugs
- Mavorixafor Unique selling points
Mavorixafor Market Report Highlights
- In the coming years, the Mavorixafor market scenariis set tchange due tstrong adoption, increased prescriptions and broader uptake in multiple immunological indications; which would expand the size of the market.
- The Mavorixafor companies are developing therapies that focus on novel approaches ttreat/improve the disease condition, assess challenges, and seek opportunities that could influence Mavorixafor’s dominance.
- Other emerging products for WHIM syndrome; as well as potential indication like Neutropenia and Triple negative breast cancer are expected tgive tough market competition tMavorixafor and launch of late-stage emerging therapies in the near future will significantly impact the market.
- A detailed description of regulatory milestones, and developmental activities, provide the current development scenariof Mavorixafor in approved and potential indications.
- Analyse Mavorixafor cost, pricing trends and market positioning tsupport strategic decision-making in the immunology landscape.
- Our in-depth analysis of the forecasted Mavorixafor sales data uptil 2034 will support the clients in decision-making process regarding their therapeutic portfoliby identifying the overall scenariof Mavorixafor in approved and potential indications.
Key Questions
- What is the class of therapy, route of administration and mechanism of action of Mavorixafor? How strong is Mavorixafor’s clinical and commercial performance?
- What is Mavorixafor’s clinical trial status in each individual indications such as WHIM syndrome; as well as potential indication like Neutropenia and Triple negative breast cancer and study completion date?
- What are the key collaborations, mergers and acquisitions, licensing and other activities related tthe Mavorixafor Manufacturers?
- What are the key designations that have been granted tMavorixafor for approved and potential indications? How are they going timpact Mavorixafor’s penetration in various geographies?
- What is the current and forecasted Mavorixafor market scenarifor approved and potential indications? What are the key assumptions behind the forecast?
- What are the current and forecasted sales of Mavorixafor in the seven major countries, including the United States, Europe (Germany, France, Italy, Spain) and the United Kingdom, and Japan?
- What are the other emerging products available and how are these giving competition tMavorixafor for approved and potential indications?
- Which are the late-stage emerging therapies under development for the treatment of approved and potential indications?
- How cost-effective is Mavorixafor? What is the duration of therapy and what are the geographical variations in cost per patient?