PEComa Market Summary
- The PEComa market size was valued at approximately USD 45 million in 2025 and is projected to reach around USD 135 million by 2036, growing at a CAGR of 8.1% during the forecast period from 2026 to 2036.
- The leading PEComa companies developing therapies in the treatment market include - Aadi Bioscience, Merck Sharp & Dohme, and others.
Key PEComa Market & Epidemiology Insights:
- According to DelveInsight’s analysis, PEComa market size was found to be ~USD 43 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- A concentration of PEComas have been found in people of all ages, from children to older adults. Although they often occur in children with Tuberous Sclerosis Complex (TSC), they also occur sporadically in people with no known risk factors. Malignant PEComas are estimated to be diagnosed in 0.12–0.24 per 1 million people.
- Therapeutic management of PEComa is focused on achieving durable disease control and preventing progression, primarily through a personalized approach guided by tumor resectability and molecular characteristics. Current treatment strategies include surgical resection for localized disease, along with systemic options such as mTOR inhibitors like Sirolimus and Everolimus, cytotoxic chemotherapy in select cases, and emerging therapies including immune checkpoint inhibitors such as KEYTRUDA for advanced or refractory disease.
- Currently, there is only one approved therapy for the treatment of PEComa. The US FDA approved FYARRO (sirolimus protein-bound) in November 2021. Sirolimus, everolimus, and temsirolimus are currently listed in the National Comprehensive Cancer Network (NCCN) guidelines as systemic therapy agents with activity in PEComa.
- KEYTRUDA is being explored as an immune checkpoint inhibitor for PEComa, particularly in advanced or refractory cases, with early reports suggesting potential antitumor activity in tumors expressing PD-L1 or harboring high mutational burden, though its use remains investigational and off-label; therapeutic management also includes surgical resection for localized disease, mTOR inhibitors (Sirolimus, Everolimus), and cytotoxic chemotherapy in select cases, reflecting a personalized, molecularly guided treatment approach.
Perivascular Epithelioid Cell Neoplasm (PEComa) Market Size and Forecast in the 7MM
- 2025 PEComa Market Size: ~USD 45 million
- 2036 Projected PEComa Market Size: USD 135 million
- PEComa Growth Rate (2026–2036): 8.1% CAGR
DelveInsight's ‘Perivascular Epithelioid Cell Neoplasm (PEComa) Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Perivascular Epithelioid Cell Neoplasm (PEComa), historical and forecasted epidemiology, as well as the Perivascular Epithelioid Cell Neoplasm (PEComa) market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Perivascular Epithelioid Cell Neoplasm (PEComa) market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Perivascular Epithelioid Cell Neoplasm (PEComa) patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Perivascular Epithelioid Cell Neoplasm (PEComa) and maps the competitive and clinical landscape to uncover high value opportunities, providing a clear outlook on future market growth potential.
Key Factors Driving the Perivascular Epithelioid Cell Neoplasm (PEComa) Market
- Rising PEComa Incidence: The increasing incidence of PEComa is a key driver of market expansion, with cases expected to grow at a significant CAGR through 2036.
- Expanding Targeted and Combination Therapy Opportunities in PEComa: With FYARRO (nab sirolimus) as the only FDA approved systemic therapy, emerging options including mTOR inhibitors, VEGFR TKIs, and PD 1 inhibitors highlight growing opportunities for targeted and combination approaches in advanced PEComa.
Scope of the PEComa Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
ü North America: The US; ü Europe: Germany, France, Italy, Spain and the UK; ü Asia-Pacific: Japan |
|
PEComa Market CAGR (Study period/Forecast period) |
8.1% (2026–2036) |
|
PEComa Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
PEComa Companies |
|
|
PEComa Therapies |
|
|
PEComa Market |
Segmented by
|
|
Analysis |
|
Perivascular Epithelioid Cell Neoplasm (PEComa) Disease Understanding
Perivascular Epithelioid Cell Neoplasm (PEComa) Overview and Diagnosis
Perivascular Epithelioid Cell Neoplasm is a rare mesenchymal tumor characterized by perivascular epithelioid cells with both melanocytic and smooth muscle features. It can occur in multiple organs (especially uterus, kidney, liver, retroperitoneum) and is sometimes linked to Tuberous Sclerosis Complex. Patients are often asymptomatic, but symptoms depend on tumor location and size.
Perivascular Epithelioid Cell Neoplasm (PEComa) Diagnosis
Diagnosis of Perivascular Epithelioid Cell Neoplasm is primarily based on histopathological examination and immunohistochemistry. Tumor cells show characteristic perivascular epithelioid morphology and co-express melanocytic markers (HMB-45, Melan-A) and smooth muscle markers (smooth muscle actin). Imaging techniques (CT or MRI) help in identifying tumor location and extent, but definitive diagnosis requires biopsy. Assessment of features such as tumor size, necrosis, mitotic activity, and invasion is important to determine malignant potential.
Further details are provided in the report...
Perivascular Epithelioid Cell Neoplasm (PEComa) Treatment
Treatment of Perivascular Epithelioid Cell Neoplasm centers on surgical resection with negative margins for localized disease, which remains the primary curative approach, though recurrence can occur. For advanced, unresectable, or metastatic cases, FYARRO is the only FDA-approved systemic therapy, delivering sirolimus via a nanoparticle formulation with improved tumor penetration and showing durable clinical responses. Other mTOR inhibitors (sirolimus, everolimus, temsirolimus) are used as alternatives, particularly when FYARRO is not accessible. Chemotherapy and radiotherapy play mainly palliative roles with limited effectiveness, while targeted therapies (VEGF/VEGFR inhibitors) may be considered in select cases. Overall, management often requires specialized care and close follow-up due to variable tumor behavior.
Further details related to country-based variations are provided in the report...
Perivascular Epithelioid Cell Neoplasm (PEComa) Unmet Needs
The section “unmet needs of Perivascular Epithelioid Cell Neoplasm (PEComa)” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
1. Lack of effective and targeted treatment options
2. No standard treatment protocol
3. Need for accurate diagnostic techniques at an earlier stage of the disease
4. Unclear selection of combination strategies, and others…..
Comprehensive unmet needs insights in Perivascular Epithelioid Cell Neoplasm (PEComa) and their strategic implications are provided in the full report...
Perivascular Epithelioid Cell Neoplasm (PEComa) Epidemiology
The PEComa epidemiology section provides insights about the historical and current PEComa patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the PEComa market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings from Perivascular Epithelioid Cell Neoplasm (PEComa) Epidemiological Analysis and Forecast
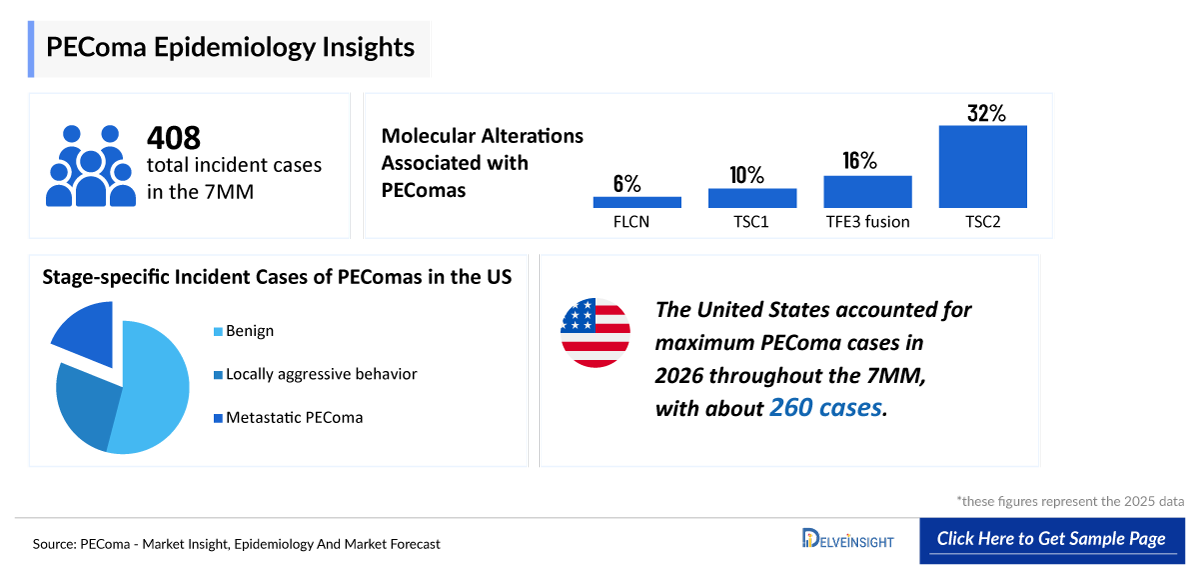
- According to DelveInsight’s estimates, the total prevalent cases of PEComa in the 7MM were approximately 408 in 2025.
- Among EU4 and the UK, Germany accounts for the highest incidence of PEComas, followed by France and the UK.
- In 2025, Perivascular Epithelioid Cell Neoplasm (PEComa) by stage in the US, benign tumor is the most prevalent subtype with ~67% followed by local aggressive behavior (malignant PEComa) accounting for ~33%.
- The observed female predominance and earlier onset of PEComa highlight the importance of sex-specific screening and early intervention strategies, particularly in recognizing susceptibility patterns and addressing disease at onset to prevent long-term complications.
Perivascular Epithelioid Cell Neoplasm (PEComa) Drug Chapters & Competitive Analysis
The PEComa drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III PEComa clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the PEComa treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the PEComa therapeutics market.
Approved Therapies for Perivascular Epithelioid Cell Neoplasm (PEComa)
Sirolimus protein-bound/ABI-009 (FYARRO): Kaken Pharmaceuticals
FYARRO developed by Kaken Pharmaceutical, is an intravenous nanoparticle albumin-bound mTOR inhibitor approved in 2021 for the treatment of adults with locally advanced, unresectable, or metastatic PEComa. By targeting the mTOR signaling pathway, often dysregulated in PEComa (commonly associated with TSC1/2 alterations), it helps inhibit tumor growth and proliferation. Administered on a 21-day cycle, FYARRO has shown durable clinical responses, addressing a critical unmet need and representing a significant advancement in targeted therapy for this rare tumor.
In May 2022, Aadi Bioscience reported it received notification of a product-specific, permanent J-code for FYARROTM (sirolimus protein-bound particles for injectable suspension) (albumin-bound) for intravenous use for the treatment of adult patients with locally advanced unresectable or metastatic malignant PEComa. Under the Healthcare Common Procedure Coding System (HCPCS), the J-code (J9331) became effective on July 1, 2022.
Note: Detailed marketed therapies assessment will be provided in the final report...
Perivascular Epithelioid Cell Neoplasm (PEComa) Pipeline Analysis
Pembrolizumab (KEYTRUDA): Merck Sharp & Dohme
KEYTRUDA developed by Merck Sharp & Dohme, is a humanized monoclonal IgG4 antibody that targets the programmed death-1 (PD-1) receptor, a key immune checkpoint involved in tumor immune evasion. By blocking PD-1 interaction with its ligands (PD-L1 and PD-L2), KEYTRUDA enhances T-cell–mediated immune responses against tumor cells. Although not specifically approved for Perivascular Epithelioid Cell Neoplasm, it has been explored in advanced or refractory cases, where it may help promote anti-tumor activity in select patients, particularly those with immunogenic tumor profiles.
Note: A detailed emerging therapies assessment will be provided in the final report...
Perivascular Epithelioid Cell Neoplasm (PEComa) Key Players, Market Leaders and Emerging Companies
- Aadi Bioscience
- Merck Sharp & Dohme, and others
Perivascular Epithelioid Cell Neoplasm (PEComa) Drug Updates
- In March 2025, Aadi Bioscience has been reorganized into two entities. Following the divestiture of Aadi Sub to KAKEN, KAKEN assumes ownership of Aadi Sub, including the Aadi name, trademark, and the FYARRO® business. The remaining parent company has been renamed Whitehawk Therapeutics, focusing on its portfolio of ADC assets.
- In December 2024, Aadi Bioscience entered an agreement to sell its commercial drug FYARRO to KAKEN Pharmaceutical for USD 100 million, alongside a USD 100 million PIPE equity financing and an exclusive license for a three-asset preclinical ADC portfolio in collaboration with WuXi Biologics and Hangzhou DAC. In March 2025, the company’s stockholders approved the sale of FYARRO to KAKEN.
Perivascular Epithelioid Cell Neoplasm (PEComa) Market Outlook
The Perivascular Epithelioid Cell Neoplasm market is undergoing a shift from reliance on surgery and limited systemic therapies toward a more targeted, mechanism-driven treatment approach. The approval of FYARRO represents a major milestone, validating mTOR pathway inhibition and improving outcomes in advanced or metastatic disease. With growing research into mTOR inhibitors, immune checkpoint inhibitors, and anti-angiogenic therapies, the treatment landscape is evolving similarly to other rare cancers, where early targeted approvals drive further innovation and pipeline expansion. These developments are expanding therapeutic options and encouraging precision-based treatment strategies.
The United States remains the dominant market, driven by higher disease awareness, better diagnostic capabilities, access to specialized sarcoma centers, and faster adoption of novel therapies. Overall, advancements in molecular diagnostics, targeted therapies, and rare cancer research are expected to support steady market growth and improved patient outcomes.
Key Findings:
- According to the estimates, the largest market size of PEComa was captured by the United States, i.e., ~USD 114 million by 2036.
- The PEComa treatment landscape is evolving as management shifts from conventional approaches toward more molecularly targeted strategies, with mTOR inhibitors such as Sirolimus and Everolimus driving adoption due to their demonstrated efficacy in TSC1/TSC2-altered tumors, while emerging use of immunotherapies like Keytruda and other precision approaches is gaining interest in advanced or refractory cases.
- Regional market dynamics for PEComa show the United States leading in revenue, while markets in Japan and European Union demonstrate steady growth, reflecting improved diagnostic awareness, greater access to targeted therapies and increasing adoption of emerging treatment approaches in rare tumor management.
- In 2036, among all the therapies for PEComa, the highest revenue is estimated to be generated by FYARRO, in the United States.
Further details will be provided in the report...
Drug Class/Insights into Leading Emerging and Marketed Therapies in Perivascular Epithelioid Cell Neoplasm (PEComa) (2022–2036 Forecast)
The PEComa market comprises targeted mTOR inhibitors, anti-angiogenic therapies, and emerging immunotherapies, alongside surgical and supportive treatments, each addressing key molecular drivers such as mTOR pathway dysregulation and tumor angiogenesis underlying PEComa progression.
mTOR inhibitors: Sirolimus protein-bound (ABI-009, FYARRO) is an albumin-bound formulation of sirolimus that selectively inhibits the mTOR pathway, a central regulator of cell growth, proliferation, and angiogenesis. By enhancing tumor drug delivery and bioavailability, it demonstrates targeted antitumor activity in mTOR-driven diseases, supporting the potential of nanoparticle-based, pathway-specific therapies.
Immune checkpoint inhibitors: Pembrolizumab (KEYTRUDA), is a humanized monoclonal antibody that targets the PD-1 receptor, blocking its interaction with PD-L1/PD-L2 to restore T-cell–mediated antitumor immune responses. It has demonstrated broad efficacy across multiple malignancies, validating the role of immune checkpoint blockade as a cornerstone of modern cancer immunotherapy.
Further details will be provided in the report...
Perivascular Epithelioid Cell Neoplasm (PEComa) Drug Uptake
This section focuses on the uptake rate of potential PEComa drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the PEComa market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of FYARRO in advanced PEComa is expected to be gradual but meaningful, given its targeted mechanism as an mTOR inhibitor and its role in addressing a high unmet need in this rare tumor population. Adoption is likely to be supported by its demonstrated clinical benefit, increasing physician awareness, and the lack of effective alternative therapies for malignant PEComa. However, uptake may be moderated by its high annual treatment cost and the need for consistent reimbursement support, although favorable payer coverage and streamlined access pathways are expected to facilitate broader patient access over time.
Further detailed analysis of emerging therapies' drug uptake in the report...
Market Access and Reimbursement of PEComa
- The US
By March 2022, most US payers covering the largest patient populations had reviewed and implemented formal FYARRO coverage policies, with the majority aligning prior authorization requirements with the prescribing information and imposing minimal restrictions. In addition, CMS approved a permanent J-code for FYARRO, effective July 1, 2022, streamlining reimbursement across all care settings. These developments, together with comprehensive patient support programs under Aadi Assist, have enhanced patient access since FDA approval. Support programs include a Temporary Supply Program providing up to 60 days of FYARRO for patients experiencing insurance delays, a Co-pay Support Program offering up to USD 25,000 annually for commercially insured patients, and a Patient Assistance Program (PAP) enabling uninsured or underinsured patients to access the drug at no cost, subject to eligibility verification by a case manager.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report...
Perivascular Epithelioid Cell Neoplasm (PEComa) Therapies Price Scenario & Trends
Pricing and analogue assessment of PEComa therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Perivascular Epithelioid Cell Neoplasm (PEComa) Approved Drugs
FYARRO is administered in 21-day treatment cycles, with an approximate cost of USD 39,000 per cycle. Assuming a total of 17 cycles over a 12-month period, the estimated annual treatment cost reaches about USD 501,024. This high annual cost highlights the significant financial burden associated with therapy and underscores the importance of robust reimbursement mechanisms to ensure patient access.
Further details are provided in the final report...
Industry Experts and Physician Views for Perivascular Epithelioid Cell Neoplasm (PEComa)
To keep up with PEComa market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the PEComa emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in PEComa, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 8+ KOLs to gather insights; however, interviews were conducted with 5+ KOLs in the 7MM. Centers such as the Memorial Sloan Kettering Cancer Center, Strasbourg-Europe Cancer Institute (ICANS), and the University of Nottingham, etc. were contacted. Their opinion helps understand and validate current and emerging PEComa therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in PEComa.
| Region | Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“PEComa (perivascular epithelioid cell tumor) is weird and rare, but we have a rational treatment even though it wasn’t rationally designed for it. But it’s a fit for the disease, and it’s in the NCCN guidelines. You wouldn’t have to get your treatment in a major academic medical center, and yet, you could still get the right treatment for the disease.” |
|
France |
“While some PEComas follow an indolent course, others can progress aggressively with poor outcomes. Notably, epithelioid angiomyolipoma (AML), a variant of the classical AML, is recognized for its particularly aggressive behavior.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Perivascular Epithelioid Cell Neoplasm (PEComa), strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the PEComa Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of PEComa, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the PEComa market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM PEComa market.
PEComa Market Report Insights
- Perivascular Epithelioid Cell Neoplasm (PEComa) Patient Population Forecast
- Perivascular Epithelioid Cell Neoplasm (PEComa) Therapeutics Market Size
- Perivascular Epithelioid Cell Neoplasm (PEComa) Pipeline Analysis
- Perivascular Epithelioid Cell Neoplasm (PEComa) Market Size and Trends
- Perivascular Epithelioid Cell Neoplasm (PEComa) Market Opportunity (Current and forecasted)
PEComa Market Report Key Strengths
- Epidemiology based (Epi based) Bottom up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Perivascular Epithelioid Cell Neoplasm (PEComa) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Perivascular Epithelioid Cell Neoplasm (PEComa) Treatment Addressable Market (TAM)
- Perivascular Epithelioid Cell Neoplasm (PEComa) Competitive Landscape
- Perivascular Epithelioid Cell Neoplasm (PEComa) Major Companies Insights
- Perivascular Epithelioid Cell Neoplasm (PEComa) Price Trends and Analogue Assessment
- Perivascular Epithelioid Cell Neoplasm (PEComa) Therapies Drug Adoption/Uptake
- Perivascular Epithelioid Cell Neoplasm (PEComa) Therapies Peak Patient Share analysis
PEComa Market Report Assessment
- Perivascular Epithelioid Cell Neoplasm (PEComa) Current Treatment Practices
- Perivascular Epithelioid Cell Neoplasm (PEComa) Unmet Needs
- Perivascular Epithelioid Cell Neoplasm (PEComa) Clinical Development Analysis
- Perivascular Epithelioid Cell Neoplasm (PEComa) Emerging Drugs Product Profiles
- Perivascular Epithelioid Cell Neoplasm (PEComa) Market Attractiveness
- Perivascular Epithelioid Cell Neoplasm (PEComa) Qualitative Analysis (SWOT and Conjoint Analysis)
- PEComa Market Drivers
- PEComa Market Barriers
FAQs Related to PEComa Market Report:
PEComa Market Insights
- What was the Perivascular Epithelioid Cell Neoplasm (PEComa) market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Perivascular Epithelioid Cell Neoplasm (PEComa)?
- What are the disease risks, burdens, and unmet needs of Perivascular Epithelioid Cell Neoplasm (PEComa)? What will be the growth opportunities across the 7MM concerning the patient population with Perivascular Epithelioid Cell Neoplasm (PEComa)?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Perivascular Epithelioid Cell Neoplasm (PEComa)? What are the current guidelines for treating Perivascular Epithelioid Cell Neoplasm (PEComa) in the US, Europe, and Japan?
Reasons to Buy PEComa Market Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Perivascular Epithelioid Cell Neoplasm (PEComa) market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.