Retinopathy of Prematurity Market Summary
Retinopathy of Prematurity (ROP) Insights and Trends
- According to DelveInsight’s analysis, the total market size of Retinopathy of Prematurity (ROP) in the United States is approximately USD 9 million in 2025.
- Currently, the primary approach for ROP screening relies on binocular indirect ophthalmoscopy. However, determining the presence of ROP and the need for treatment heavily relies on experienced ophthalmologists. Hence, it is crucial to create a straightforward, precise, and efficient diagnostic method.
- The overall disease burden of ROP does not signify the actual number of patients going for treatment, as ˜10% of the total ROP patients, which represents the severe and treatable pool, in the US and European countries, and ˜30% of the total ROP patients in Japan are estimated to undergo any kind of treatment.
- Laser therapy is a standard of care for treating ROP and has emerged as a viable treatment option for ROP. This approach aims to counter the overproduction of VEGF within the retina, a key factor contributing to the condition. Anti-VEGF agents are useful in infants for whom laser photocoagulation is difficult or impossible.
- The FDA-approved drugs for treating ROP include LUCENTIS (ranibizumab) and EYLEA (aflibercept). Regeneron is leveraging its experience in retinal diseases with EYLEA’s label expansion in Retinopathy of Prematurity. This stems from the FDA's recent approval of EYLEA as a feasible treatment for ROP in preterm infants in 2023.
- Therapies that are both swift and efficient, enabling treatment at the bedside in local Neonatal Intensive Care Units (NICUs) without requiring lengthy procedures or specialized anesthesia, are more likely to achieve success compared to stressful and time-consuming approaches such as laser photocoagulation.
- Although preterm infant mortality is low, the proportion of patients with treatment-requiring retinopathy of prematurity is high in Japan. Thus, it is likely that compared to other countries, ophthalmologists in Japan treat infants at an earlier stage of ROP. Additionally, in recent times, anti-vascular endothelial growth factor (anti-VEGF) antibody treatment has become widespread in many facilities in Japan.
Retinopathy of Prematurity (ROP) Market Size and Forecast in the 7MM
- 2025 ROP Market Size: ~USD 14 million
- 2036 Projected ROP Market Size: ~USD 20 million
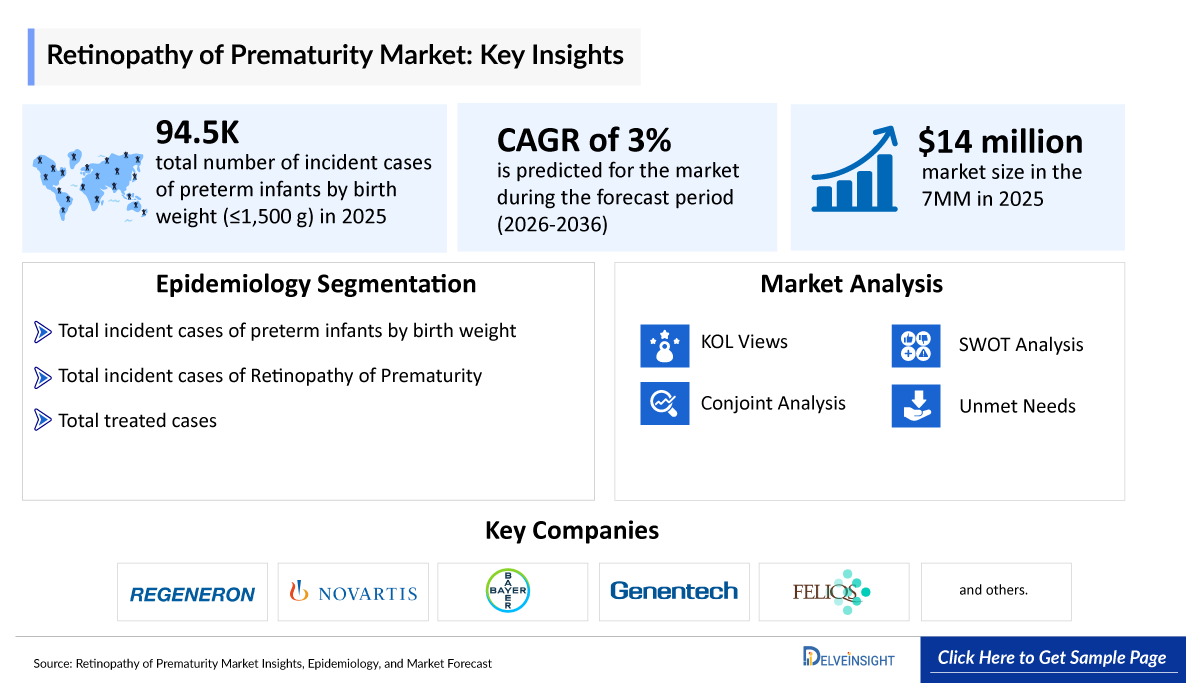
- ROP Growth Rate (2026–2036): 3% CAGR
DelveInsight's ‘Retinopathy of Prematurity (ROP) – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the ROP, historical and forecasted epidemiology, as well as the ROP market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The ROP market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates ROP patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in ROP and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
|
|
Historical Year |
|
|
Forecast Period |
|
|
Base Year |
|
|
Geographies Covered |
|
|
ROP Market CAGR (Study period/Forecast period) |
|
|
ROP Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
ROP Companies |
|
|
ROP Therapies |
|
|
ROP Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Retinopathy of Prematurity (ROP) Market
Rising ROP Incidence
The increasing incidence of ROP, particularly among preterm infants of very low birth weight (≤1,500 g), is a primary driver of ROP market expansion. In the US, in 2025, there were ~18,200 incident cases of ROP, which will further reach by 3% CAGR (2026-2036).
Rising Opportunities in ROP Therapies
Emerging evidence supporting the efficacy and safety of anti-VEGF agents highlights a growing opportunity for drug developers to focus on targeted therapies that inhibit abnormal retinal angiogenesis and improve long-term visual outcomes.
Emerging ROP Competitive Landscape
The ROP market is evolving with new therapies, advanced diagnostics, and a growing focus on early detection and prevention. Some of the ROP drugs in clinical trials include FLQ-101 (FeliQS Corporation), IBP-1118 (Infant Bacterial Therapeutics), and others.
Retinopathy of Prematurity (ROP) Understanding and Treatment Algorithm
Retinopathy of Prematurity (ROP) Overview and Diagnosis
ROP is an eye disease that can happen in premature babies (born early) or those who weigh less than 1,500 g at birth. ROP happens when abnormal blood vessels grow in the retina (the light-sensitive layer of tissue in the back of the eye). Some babies with ROP have mild cases and get better without treatment. However, some babies need treatment to protect their vision and prevent blindness.
Retinopathy of Prematurity (ROP) Diagnosis
ROP is diagnosed through a comprehensive eye exam by a specialized ophthalmologist. This exam involves dilating the infant’s pupils to assess the retina’s blood vessels. The disease is categorized into stages based on severity, location, and extent of abnormal vessel growth. Retinal imaging may aid in monitoring. Depending on the severity, treatment options such as laser therapy or medication are considered. Regular follow-up exams are crucial for timely intervention and to prevent vision loss.
Further details are provided in the report.
Retinopathy of Prematurity (ROP) Treatment
Many infants with ROP have mild cases that improve on their own. However, some infants require treatment to prevent the progression of ROP. Treatment options primarily encompass laser therapy, anti-VEGF injections, and ocular surgery. However, mainly two options are used for treatment once it is confirmed that a child has severe ROP. The first treatment type is the laser, applied to the immature or undeveloped part of the retina. This treatment is the current standard of care for the treatment of ROP. The second type of treatment is an injection of medication (bevacizumab, ranibizumab, and aflibercept have been used) into the eye. This medication injection stops a signal causing the abnormal blood vessels in ROP to form. These medications may be used as an alternative to, or in addition to, laser treatment. The injection is a newer treatment than the laser treatment. The effect of laser or injection treatment for ROP is usually good, with the disappearance of abnormal blood vessels from ROP. However, even with good diagnosis and timely treatment, the ROP sometimes worsens, and the retina pulls away from the back of the eye (a retinal detachment).
Further details related to country-based variations are provided in the report.
Retinopathy of Prematurity (ROP) Unmet Needs
The section “unmet needs of Retinopathy of Prematurity (ROP)” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
1. Lack of emerging therapies in the pipeline
2. Long-term follow-up and monitoring
3. Lack of long-term safety effects of anti-VEGF agents in infants
4. Limited treatment options and others…..
Comprehensive unmet needs insights in Retinopathy of Prematurity (ROP) and their strategic implications are provided in the full report.
Retinopathy of Prematurity (ROP) Epidemiology
Key Findings from Retinopathy of Prematurity (ROP) Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, the total number of incident cases of preterm infants by birth weight (≤1,500 g) in the 7MM was ~94,500 in 2025. As per the estimate, these cases are anticipated to increase during the forecast period of 2026–2036.
- Among the EU4 and the UK, France accounted for the highest number of incident cases of ROA, followed by Germany, whereas Spain accounted for the lowest number of cases in 2025.
- Among the 7MM, the United States has the highest number of patients undergoing treatment for ROP, followed by Japan, while Spain has the lowest in 2025.
Retinopathy of Prematurity (ROP) Drug Chapters & Competitive Analysis
The ROP drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the ROP treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the ROP therapeutics market.
Approved Therapies for Retinopathy of Prematurity (ROP)
LUCENTIS (ranibizumab): Novartis
LUCENTIS (ranibizumab) is a humanized, high-affinity antibody fragment that binds to vascular endothelial growth factor A (VEGF-A), a protein that can cause the growth of blood vessels in the eye, potentially leading to vision loss. LUCENTIS is an anti-VEGF therapy that is injected into the eye. It is approved in the EU, JP, and other countries to treat patients with ROP. LUCENTIS is indicated in preterm infants for the treatment of ROP with Zone I (Stage 1+, 2+, 3 or 3+), Zone II (Stage 3+), or AP-ROP (aggressive posterior Retinopathy of Prematurity) disease.
EYLEA (aflibercept): Regeneron/Bayer
EYLEA is a VEGF inhibitor formulated as an injection for the eye. It is designed to block the growth of new blood vessels and decrease the ability of fluid to pass through blood vessels (vascular permeability) in the eye by blocking VEGF-A and placental growth factor (PLGF), two growth factors involved in ocular angiogenesis. Regeneron and Bayer are jointly developing EYLEA. The lead sponsors of the trials were Regeneron for BUTTERFLEYE and Bayer for FIREFLEYE. Bayer and Regeneron are collaborating on the global development of EYLEA. Regeneron maintains exclusive rights to EYLEA in the United States. Bayer has licensed exclusive marketing rights outside the United States, where the companies share the profits from sales of EYLE equally.
|
ROP Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
LUCENTIS (ranibizumab) |
Novartis |
Retinopathy of prematurity with zone I (stage 1+, 2+, 3 or 3+), zone II (stage 3+) or AP ROP (aggressive posterior ROP) disease |
Monoclonal antibody |
Intravitreal Injection |
Vascular endothelial growth factor A inhibitors |
EU: September 2019; JP: October 2019 |
|
EYLEA (aflibercept) |
Regeneron/Bayer |
Retinopathy of prematurity in preterm infants |
Recombinant fusion protein |
Intravitreal injection |
Vascular endothelial growth factor A inhibitors |
US: February 2023; EU: December 2022; JP: September 2022 |
Note: Detailed marketed therapies assessment will be provided in the final report.
Retinopathy of Prematurity (ROP) Pipeline Analysis
There are only a few candidates in the pipeline for ROP at the moment. Among them, the majority of candidates are in the preclinical stage of investigation, focusing on the preventive options of ROP. Key players, such as FeliQS Corporation (FLQ-101) and Infant Bacterial Therapeutics (IBP-1118), are developing therapies for the prevention of ROP.
Comparison of Emerging Drugs Under Development | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in US |
|
FLQ-101 |
FeliQS Corporation |
I |
Prevention of retinopathy of prematurity |
Intravenous |
Lipid peroxidation inhibitor |
Small molecule |
- |
|
IBP-1118 |
Infant Bacterial Therapeutics |
Preclinical |
Pediatric and adult participants with Still's disease, including MAS |
- |
Undefined mechanism |
Dipeptide |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Note: A detailed emerging therapies assessment will be provided in the final report
Retinopathy of Prematurity (ROP) Key Players, Market Leaders and Emerging Companies
- Regeneron Pharmaceuticals
- Novartis
- Bayer
- Genentech
- FELIQS Corporation, and others
Retinopathy of Prematurity (ROP) Drug Updates
- In March 2026, SignaBlok announced that its TREM-1 peptide inhibitor received FDA Orphan Drug Designation for ROP.
- In February 2023, the FDA approved EYLEA (aflibercept) as the first drug for Retinopathy of Prematurity, a condition in premature infants where abnormal retinal blood vessel growth can cause retinal detachment and vision loss.
Retinopathy of Prematurity (ROP) Market Outlook
Currently, the standard of care for treating ROP is laser treatment. The other important treatment for ROP is the use of anti-VEGF injections. Currently, there are only two drugs, namely, EYLEA (aflibercept) and LUCENTIS (ranibizumab), approved for the treatment of Retinopathy of Prematurity.
When faced with severe ROP in a child, there are generally two avenues of treatment. The first method involves using a laser to target the underdeveloped regions of the retina. This time-tested approach has been the go-to solution for ROP for many years. Alternatively, the second treatment option involves injecting anti-VEGF injections (Bevacizumab, EYLEA, and LUCENTIS) directly into the eye. These injections intercept the signals that trigger abnormal blood vessel growth in ROP. Sometimes, these medications are used either in conjunction with or as an alternative to laser treatment. Unlike well-established laser therapy, medication injections are a relatively newer approach.
Both laser and injection treatments for ROP often yield positive outcomes, leading to the regression of the abnormal blood vessels associated with the condition. While effective, laser photocoagulation may not only be stressful but can also be time-consuming. In comparison, aflibercept works rapidly and provides an FDA-approved treatment that may be more accessible for some patients. Importantly, this approval improves access to care by allowing bedside treatment at the local NICU without the need for general anesthesia, pediatric anesthesia specialists, and investment in laser infrastructure.
However, even with swift diagnosis and timely intervention, there are instances where ROP stubbornly persists, and the retina detaches from the back of the eye—a condition known as retinal detachment. This distressing development can result in severely compromised vision. Nevertheless, there is hope: skilled retina surgeons are equipped to address retinal detachment caused by ROP through specialized surgery.
- The total market size of Retinopathy of Prematurity in the 7MM is approximately ~USD 14 million in 2025.
- Among EU4, Germany accounts for the largest market size in 2025, while Spain occupies the bottom of the ladder.
- In 2025, the highest revenue was generated by laser therapy in the 7MM.
- Among anti-VEGFs, bevacizumab is the most used therapy (approximately 80% of all VEGFs), followed by LUCENTIS as an approved therapy in Europe and Japan and off-label in the US. Whereas EYLEA is the most recent approval in all the countries. In terms of cost-effectiveness, bevacizumab is the most economical option.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Retinopathy of Prematurity (ROP) (2022–2036 Forecast)
- The ROP market comprises anti-VEGF biologics and emerging targeted approaches alongside conventional laser therapy and supportive care, each addressing abnormal retinal angiogenesis and vascular dysregulation in premature infants.
- Anti-VEGF: Aflibercept is a VEGF inhibitor that suppresses abnormal retinal neovascularization, reducing disease progression and establishing pharmacological intervention as a key treatment modality in ROP.
- Monoclonal antibody: Ranibizumab selectively inhibits VEGF-A, improving retinal outcomes and supporting the role of targeted biologics in managing ROP with a favorable safety profile.
- Emerging pathway-targeted therapies: IGF-1 analogs and novel anti-inflammatory agents (e.g., TREM-1 inhibitors) aim to restore normal vascular development and reduce inflammation, representing next-generation approaches in ROP management.
Biologics, particularly anti-VEGF agents, define the current innovation landscape in ROP, while emerging pathway-targeted therapies are expected to drive future pipeline expansion and improve long-term outcomes.
Further details will be provided in the report….
Retinopathy of Prematurity (ROP) Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the ROP market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in ROP is expected to vary across anti-VEGF biologics, emerging pharmacological agents, and conventional laser-based interventions. Recently approved therapies such as Aflibercept demonstrate increasing uptake, while Ranibizumab is anticipated to show moderate but steady adoption, supported by strong clinical efficacy, targeted VEGF inhibition, and growing physician confidence in pharmacologic management of ROP. Their adoption is largely driven by the limitations of laser photocoagulation, particularly in preserving peripheral vision and reducing long-term complications in premature infants.
In comparison, other therapies such as off-label Bevacizumab are expected to maintain significant but variable uptake due to cost advantages and real-world experience, while next-generation approaches (including sustained-release anti-VEGF agents and IGF-1–targeted therapies) are projected to show gradual but progressive uptake as clinical evidence evolves and these therapies become integrated into neonatal care practices.
Further detailed analysis of emerging therapies' drug uptake in the report…
Retinopathy of Prematurity Market Access and Reimbursement of ROP
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report….
Retinopathy of Prematurity (ROP) Therapies Price Scenario & Trends
Pricing and analogue assessment of ROP therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and the understanding of how pricing influences market access, adherence, and long-term uptake.
- Pricing of Retinopathy of Prematurity (ROP) Approved Drugs
The recommended dose of EYLEA is 0.4 mg via intravitreal injection. Treatment begins with a single injection per eligible eye, which may be administered bilaterally on the same day. Assuming US pricing, EYLEA is estimated to cost approximately USD 1,850 to USD 2,000 per injection.
Further details are provided in the final report….
Industry Experts and Physician Views for Retinopathy of Prematurity (ROP)
To keep up with ROP market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the ROP emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in ROP, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the Stanley Manne Children’s Research Institute, Lurie Children’s Hospital of Chicago, and National Eye Institute, National Institutes of Health, Bethesda, USA, etc., were contacted. Their opinion helps understand and validate current and emerging ROP therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in ROP.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Retinopathy of prematurity stands as a major contributor to childhood vision loss on a global scale. Until this point, the primary treatment method, laser photocoagulation, has been intricate and time-consuming. This procedure involves the permanent removal of retina tissue and adds stress to both the premature infants undergoing it and their families during this fragile period following preterm birth. In a significant breakthrough, healthcare providers will have access to aflibercept, an FDA-approved medication, to address this devastating condition in these extremely young patients.” |
|
Germany |
“Laser therapy for ROP is a safe and effective measure to control disease activity in most cases. However, the disadvantage of laser therapy is that the laser-treated retina is converted into scar tissue and is thus lost for possible future visual function.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Retinopathy of Prematurity (ROP), strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Retinopathy of Prematurity Market Report
- The Retinopathy of Prematurity Market report covers a segment of key events, an executive summary, a descriptive overview of ROP, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the ROP market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Retinopathy of Prematurity Market report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM ROP market.
Retinopathy of Prematurity Market Report Insights
- Retinopathy of Prematurity (ROP) Patient Population Forecast
- Retinopathy of Prematurity (ROP) Therapeutics Market Size
- Retinopathy of Prematurity (ROP) Pipeline Analysis
- Retinopathy of Prematurity (ROP) Market Size and Trends
- Retinopathy of Prematurity (ROP) Market Opportunity (Current and forecasted)
Retinopathy of Prematurity Market Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- Retinopathy of Prematurity (ROP) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Retinopathy of Prematurity (ROP) Treatment Addressable Market (TAM)
- Retinopathy of Prematurity (ROP) Competitive Landscape
- Retinopathy of Prematurity (ROP) Major Companies Insights
- Retinopathy of Prematurity (ROP) Price Trends and Analogue Assessment
- Retinopathy of Prematurity (ROP) Therapies Drug Adoption/Uptake
- Retinopathy of Prematurity (ROP) Therapies Peak Patient Share Analysis
Retinopathy of Prematurity Market Report Assessment
- Retinopathy of Prematurity (ROP) Current Treatment Practices
- Retinopathy of Prematurity (ROP) Unmet Needs
- Retinopathy of Prematurity (ROP) Clinical Development Analysis
- Retinopathy of Prematurity (ROP) Emerging Drugs Product Profiles
- Retinopathy of Prematurity (ROP) Market Attractiveness
- Retinopathy of Prematurity (ROP) Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Retinopathy of Prematurity Market Insights
- What was the Retinopathy of Prematurity (ROP) market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Retinopathy of Prematurity (ROP)?
- What are the disease risks, burdens, and unmet needs of Retinopathy of Prematurity (ROP)? What will be the growth opportunities across the 7MM concerning the patient population with Retinopathy of Prematurity (ROP)?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Retinopathy of Prematurity (ROP)? What are the current guidelines for treating Retinopathy of Prematurity (ROP) in the US, Europe, and Japan?
Reasons to Buy the Retinopathy of Prematurity Market Forecast Report
- The Retinopathy of Prematurity Market report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Retinopathy of Prematurity (ROP) market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)‑ enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.