Severe Toxicities in Lymphoma Market
- As per DelveInsight, the Severe Toxicities in Lymphoma Market is expected to expand at a healthy growth rate during the forecast period (2023-2032), owing to the launch of new therapies in the market and the rise in the number of cases.
- Several major pharma and biotech companies, including Angiocrine Bioscience, among others, are actively working in the Severe Toxicities in Lymphoma Market.
Download the Sample PDF to Get More Insight @ Severe Toxicities in Lymphoma Market
DelveInsight's "Severe Toxicities in Lymphoma Market Insights, Epidemiology, and Market Forecast-2032" report delivers an in-depth understanding of the Severe Toxicities in Lymphoma, historical and forecasted epidemiology as well as the Severe Toxicities in Lymphoma therapeutics market trends in the United States, EU5 (Germany, Spain, Italy, France, and the United Kingdom) and Japan.
The Severe Toxicities in Lymphoma market report provides current treatment practices, emerging drugs, Severe Toxicities in Lymphoma market share of the individual therapies, current and forecasted Severe Toxicities in Lymphoma market size from 2019 to 2032 segmented by seven major markets. The Report also covers current Severe Toxicities in Lymphoma treatment practice/algorithm, market drivers, market barriers, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the Severe Toxicities in Lymphoma market.
|
Study Period |
2019 to 2032 |
|
Forecast Period |
2023-2032 |
|
Geographies Covered |
|
|
Severe Toxicities in Lymphoma Market |
|
|
Severe Toxicities in Lymphomas Market Size | |
|
Severe Toxicities in Lymphoma Companies |
Angiocrine Bioscience, and others. |
|
Severe Toxicities in Lymphoma Epidemiology Segmentation |
|
Severe Toxicities in Lymphoma Treatment Market
"Severe toxicities in lymphoma" generally refer to the adverse effects and complications that arise during or after lymphoma treatment, particularly chemotherapy or radiation therapy. These toxicities can vary in severity and impact patients' overall well-being and treatment outcomes. The causes and risk factors for severe toxicities in lymphoma often stem from the potent nature of cancer treatments, which can inadvertently affect healthy cells and tissues in addition to targeting cancer cells. Risk factors for severe toxicities include the type and dosage of chemotherapy drugs, the combination of treatments used, the patient's overall health and existing medical conditions, age, and genetic factors.
Severe Toxicities In Lymphoma Signs and Symptoms
The signs and symptoms of severe toxicities in lymphoma treatment can encompass a wide range of effects on various bodily systems. Common symptoms may include nausea, vomiting, fatigue, hair loss, anemia, lowered white blood cell count (neutropenia), increased risk of infections, nerve damage (peripheral neuropathy), heart problems, lung issues, and cognitive impairment, among others. The specific signs depend on the type of treatment and individual patient factors.
Severe Toxicities In Lymphoma Diagnosis
Diagnosing severe toxicities in lymphoma treatment involves close monitoring by healthcare professionals during and after therapy. Patients' symptoms are carefully assessed, and blood tests, imaging studies, or other diagnostic procedures may be performed to evaluate the extent and impact of treatment-related toxicities on various organs and bodily functions.
Treatment Options in the Severe Toxicities In Lymphoma Therapeutics Market
Managing severe toxicities in lymphoma treatment involves a range of strategies aimed at mitigating adverse effects and supporting patients through their treatment journey. Medications can be prescribed to alleviate specific symptoms, such as antiemetics for nausea or antibiotics for infections. Adjustments to treatment dosages or schedules may be necessary to reduce toxicity while maintaining treatment efficacy. Supportive care measures, including nutritional support, pain management, and counseling, play a crucial role in managing treatment-related toxicities.
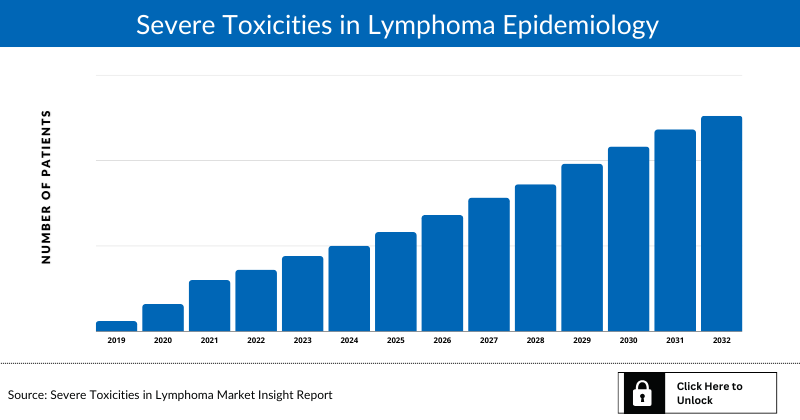
Severe Toxicities in Lymphoma Epidemiology
The Severe Toxicities in Lymphoma epidemiology section provides insights into the historical and current Severe Toxicities in Lymphoma patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the Severe Toxicities in Lymphoma market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings
The disease epidemiology covered in the report provides historical as well as forecasted Severe Toxicities in Lymphoma epidemiology scenario in the 7MM covering the United States, EU5 countries (Germany, Spain, Italy, France, and the United Kingdom), and Japan from 2019 to 2032.
Country Wise- Severe Toxicities in Lymphoma Epidemiology
The epidemiology segment also provides the Severe Toxicities in Lymphoma epidemiology data and findings across the United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan.
Get detailed insights into the historical as well as forecasted epidemiology trends in the 7MM, at: Severe Toxicities in Lymphoma Epidemiology Forecast
Severe Toxicities in Lymphoma Drug Chapters
The drug chapter segment of the Severe Toxicities in Lymphoma report encloses a detailed analysis of Severe Toxicities in Lymphoma marketed drugs and late-stage (Phase-III and Phase-II) Severe Toxicities in Lymphoma pipeline drugs. It also helps to understand the Severe Toxicities in Lymphoma clinical trial details, expressive pharmacological action, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug, and the latest news and press releases.
Severe Toxicities in Lymphoma Marketed Drugs
The report provides the details of the marketed products/off-label treatments available for Severe Toxicities in Lymphoma treatment.
Severe Toxicities in Lymphoma Emerging Drugs
The report provides the details of the emerging therapies under the late and mid-stage of development for Severe Toxicities in Lymphoma treatment.
Severe Toxicities in Lymphoma Market Outlook
The Severe Toxicities in Lymphoma market outlook of the report helps to build a detailed comprehension of the historic, current, and forecasted Severe Toxicities in Lymphoma market trends by analyzing the impact of current Severe Toxicities in Lymphoma therapies on the market, unmet needs, drivers and barriers, and demand for better technology.
This segment gives a thorough detail of Severe Toxicities in Lymphoma market trend of each marketed drug and late-stage pipeline therapy by evaluating their impact based on the annual cost of therapy, inclusion and exclusion criteria, mechanism of action, compliance rate, growing need of the market, increasing patient pool, covered patient segment, expected launch year, competition with other therapies, brand value, their impact on the market and view of the key opinion leaders. The calculated Severe Toxicities in Lymphoma market data are presented with relevant tables and graphs to give a clear view of the market at first sight.
According to DelveInsight, the Severe Toxicities in Lymphoma market in 7MM is expected to witness a major change in the study period 2019-2032.
Key Findings
This section includes a glimpse of the Severe Toxicities in Lymphoma market in 7MM.
The United States Market Outlook
This section provides the total Severe Toxicities in Lymphoma market size and market size by therapies in the United States.
EU-5 Countries: Market Outlook
The total Severe Toxicities in Lymphoma market size and market size by therapies in Germany, France, Italy, Spain, and the United Kingdom is provided in this section.
Japan Market Outlook
The total Severe Toxicities in Lymphoma market size and market size by therapies in Japan is also mentioned.
Severe Toxicities in Lymphoma Drugs Uptake
This section focuses on the rate of uptake of the Severe Toxicities in Lymphoma drugs recently launched in the Severe Toxicities in Lymphoma market or expected to get launched in the market during the study period 2019-2032. The analysis covers Severe Toxicities in Lymphoma market uptake by drugs; patient uptake by therapies; and sales of each drug.
Severe Toxicities in Lymphoma Drugs Uptake helps in understanding the drugs with the most rapid uptake, and reasons behind the maximal use of new drugs, and allow the comparison of the drugs on the basis of Severe Toxicities in Lymphoma market share and size which again will be useful in investigating factors important in market uptake and in making financial and regulatory decisions.
Severe Toxicities in Lymphoma Pipeline Development Activities
The Severe Toxicities in Lymphoma report provides insights into Severe Toxicities in Lymphoma clinical trials within Phase II and Phase III stages. It also analyses Severe Toxicities in Lymphoma key players involved in developing targeted therapeutics.
Pipeline Development Activities
The Severe Toxicities in Lymphoma report covers detailed information of collaborations, acquisitions, and mergers, licensing, patent details, and other information for Severe Toxicities in Lymphoma emerging therapies.
Severe Toxicities in Lymphoma Market Growth Factors
The market surrounding the management of severe toxicities in lymphoma treatment is driven by several factors. Ongoing research and development efforts focus on improving chemotherapy drugs and radiation therapy techniques to minimize adverse effects. The introduction of supportive care medications and therapies to alleviate specific toxicities contributes to market expansion. Additionally, initiatives to enhance patient education, improve healthcare infrastructure, and advance personalized medicine approaches to tailor treatments based on individual patient profiles further propel growth within the market for managing severe toxicities in lymphoma treatment.
Reimbursement Scenario in Severe Toxicities in Lymphoma
Approaching reimbursement proactively can have a positive impact both during the late stages of product development and well after product launch. In a report, we take reimbursement into consideration to identify economically attractive indications and market opportunities. When working with finite resources, the ability to select the markets with the fewest reimbursement barriers can be a critical business and price strategy.
KOL- Views
To keep up with current Severe Toxicities in Lymphoma market trends, we take KOLs and SMEs ' opinions working in the Severe Toxicities in Lymphoma domain through primary research to fill the data gaps and validate our secondary research. Their opinion helps to understand and validate current and emerging therapies treatment patterns or Severe Toxicities in Lymphoma market trends. This will support the clients in potential upcoming novel treatments by identifying the overall scenario of the market and the unmet needs.
Competitive Intelligence Analysis
We perform Competitive and Market Intelligence analysis of the Severe Toxicities in Lymphoma Market by using various Competitive Intelligence tools that include - SWOT analysis, PESTLE analysis, Porter's five forces, BCG Matrix, Market entry strategies, etc. The inclusion of the analysis entirely depends upon the data availability.
Scope of the Severe Toxicities In Lymphoma Market Report
- The report covers the descriptive overview of Severe Toxicities in Lymphoma, explaining its causes, signs and symptoms, pathophysiology, diagnosis and currently available therapies
- Comprehensive insight has been provided into the Severe Toxicities in Lymphoma epidemiology and treatment in the 7MM
- Additionally, an all-inclusive account of both the current and emerging therapies for Severe Toxicities in Lymphoma is provided, along with the assessment of new therapies, which will have an impact on the current treatment landscape
- A detailed review of the Severe Toxicities in Lymphoma market; historical and forecasted is included in the report, covering drug outreach in the 7MM
- The report provides an edge while developing business strategies, by understanding trends shaping and driving the global Severe Toxicities in Lymphoma market
Severe Toxicities In Lymphoma Market Report Highlights
- In the coming years, the Severe Toxicities in Lymphoma market is set to change due to the rising awareness of the disease, and incremental healthcare spending across the world; which would expand the size of the market to enable the drug manufacturers to penetrate more into the market
- The companies and academics are working to assess challenges and seek opportunities that could influence Severe Toxicities in Lymphoma R&D. The therapies under development are focused on novel approaches to treat/improve the disease condition
- Major players are involved in developing therapies for Severe Toxicities in Lymphoma. The launch of emerging therapies will significantly impact the Severe Toxicities in Lymphoma market
- A better understanding of disease pathogenesis will also contribute to the development of novel therapeutics for Severe Toxicities in Lymphoma
- Our in-depth analysis of the pipeline assets across different stages of development (Phase III and Phase II), different emerging trends and comparative analysis of pipeline products with detailed clinical profiles, key cross-competition, launch date along with product development activities will support the clients in the decision-making process regarding their therapeutic portfolio by identifying the overall scenario of the research and development activities
Severe Toxicities in Lymphoma Market Report Insights
- Severe Toxicities in Lymphoma Patient Population
- Severe Toxicities in Lymphoma Therapeutic Approaches
- Severe Toxicities in Lymphoma Pipeline Analysis
- Severe Toxicities in Lymphoma Market Size and Trends
- Severe Toxicities in Lymphoma Market Opportunities
- Impact of Upcoming Severe Toxicities in Lymphoma Therapies
Severe Toxicities in Lymphoma Market Report Key Strengths
- 11 Years Forecast
- 7MM Coverage
- Severe Toxicities in Lymphoma Epidemiology Segmentation
- Key Cross Competition
- Highly Analyzed Severe Toxicities in Lymphoma Market
- Severe Toxicities in Lymphoma Drugs Uptake
Severe Toxicities in Lymphoma Market Report Assessment
- Current Severe Toxicities in Lymphoma Treatment Practices
- Unmet Needs in the Severe Toxicities in Lymphoma Market
- Severe Toxicities in Lymphoma Pipeline Product Profiles
- Severe Toxicities in Lymphoma Market Attractiveness
- Severe Toxicities in Lymphoma Market Drivers and Barriers
Key Questions Answered In The Severe Toxicities In Lymphoma Market Report:
Severe Toxicities In Lymphoma Market Insights:
- What was the Severe Toxicities in Lymphoma drug class share (%) distribution in 2019 and how it would look like in 2032?
- What would be the Severe Toxicities in Lymphoma total market size as well as market size by therapies across the 7MM during the forecast period (2019-2032)?
- What are the key findings pertaining to the market across 7MM and which country will have the largest Severe Toxicities in Lymphoma market size during the forecast period (2019-2032)?
- At what CAGR, the Severe Toxicities in Lymphoma market is expected to grow by 7MM during the forecast period (2019-2032)?
- What would be the Severe Toxicities in Lymphoma market outlook across the 7MM during the forecast period (2019-2032)?
- What would be the Severe Toxicities in Lymphoma market growth till 2032, and what will be the resultant market Size in the year 2032?
- How would the unmet needs affect the market dynamics and subsequent analysis of the associated trends?
Severe Toxicities In Lymphoma Epidemiology Insights:
- What are the disease risk, burden, and regional/ethnic differences of the Severe Toxicities in Lymphoma?
- What are the key factors driving the epidemiology trend for seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What is the historical Severe Toxicities in Lymphoma patient pool in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- What would be the forecasted patient pool of Severe Toxicities in Lymphoma in seven major markets covering the United States, EU5 (Germany, Spain, France, Italy, UK), and Japan?
- Where will be the growth opportunities in the 7MM with respect to the patient population pertaining to Severe Toxicities in Lymphoma?
- Out of all 7MM countries, which country would have the highest prevalent population of Severe Toxicities in Lymphoma during the forecast period (2019-2032)?
- At what CAGR the patient population is expected to grow in 7MM during the forecast period (2019-2032)?
Current Severe Toxicities In Lymphoma Treatment Scenario, Marketed Drugs and Emerging Therapies
- What are the current options for the Severe Toxicities in Lymphoma treatment in addition to the approved therapies?
- What are the current treatment guidelines for the treatment of Severe Toxicities in Lymphoma in the USA, Europe, and Japan?
- What are the Severe Toxicities in Lymphoma marketed drugs and their respective MOA, regulatory milestones, product development activities, advantages, disadvantages, safety and efficacy, etc.?
- How many companies are developing therapies for the treatment of Severe Toxicities in Lymphoma?
- How many therapies are in-development by each company for Severe Toxicities in Lymphoma treatment?
- How many are emerging therapies in mid-stage, and late stage of development for Severe Toxicities in Lymphoma treatment?
- What are the key collaborations (Industry - Industry, Industry - Academia), Mergers and acquisitions, licensing activities related to the Severe Toxicities in Lymphoma therapies?
- What are the recent novel therapies, targets, mechanisms of action and technologies being developed to overcome the limitation of existing therapies?
- What are the clinical studies going on for Severe Toxicities in Lymphoma and their status?
- What are the current challenges faced in drug development?
- What are the key designations that have been granted for the emerging therapies for Severe Toxicities in Lymphoma?
- What are the global historical and forecasted market of Severe Toxicities in Lymphoma?
Reasons to buy Severe Toxicities In Lymphoma Market Forecast Report
- The report will help in developing business strategies by understanding trends shaping and driving the Severe Toxicities in Lymphoma market
- To understand the future market competition in the Severe Toxicities in Lymphoma market and Insightful review of the key market drivers and barriers
- Organize sales and marketing efforts by identifying the best opportunities for Severe Toxicities in Lymphoma in the US, Europe (Germany, Spain, Italy, France, and the United Kingdom) and Japan
- Identification of strong upcoming players in the market will help in devising strategies that will help in getting ahead of competitors
- Organize sales and marketing efforts by identifying the best opportunities for Severe Toxicities in Lymphoma market
- To understand the future market competition in the Severe Toxicities in Lymphoma market