Short Bowel Syndrome Market Summary
Short bowel syndrome (SBS) Insights and Trends
- Short bowel syndrome (SBS) is a complex condition caused by loss or dysfunction of parts of the small or large intestine, leading to poor absorption of nutrients, fluids, and electrolytes. Symptoms vary but often include severe diarrhea, dehydration, malnutrition, and weight loss. While there is no cure, SBS can usually be managed, though severe cases may be life-threatening.
- SBS is more common in adults due to chronic diseases, while children experience higher incidence due to neonatal conditions.
- TPN and dietary changes are the current standard of care treatment. SBS treatment is often tailored to the individual and depends on severity and symptoms.
- Growth factor therapy is used in SBS patients who are unable to achieve enteral independence despite optimal care. However, recombinant human growth hormone (ZORBTIVE), FDA-approved in 2003, has seen declining use due to limited efficacy and safety concerns.
- The prevalence of home parenteral nutrition for SBS is generally higher in the US than in Europe. US data often report annual prevalence rates, whereas European studies present point prevalence. This discrepancy partly reflects differences in reporting methods and the broader availability of home parenteral nutrition in the US, which encourages earlier transitions to home care for cost-saving reasons.
- In 2012, GATTEX was approved by the US FDA for the treatment of SBS. Human GLP-2 half-life is around 7 min, and GATTEX half-life is around 2–3 h. Despite its extended duration of action, GATTEX still requires daily subcutaneous injections owing to its relatively short half-life. Furthermore, its lyophilized powder formulation involves a complicated reconstitution process prior to administration
- Companies like Ironwood Pharmaceuticals (Apraglutide), Zealand Pharma (Glepaglutide), Hanmi Pharmaceutical (Sonefpeglutide), and Napo Therapeutics (MYTESI) are investigating their key products for SBS.
Short bowel syndrome (SBS) Market size and forecast of the 7MM
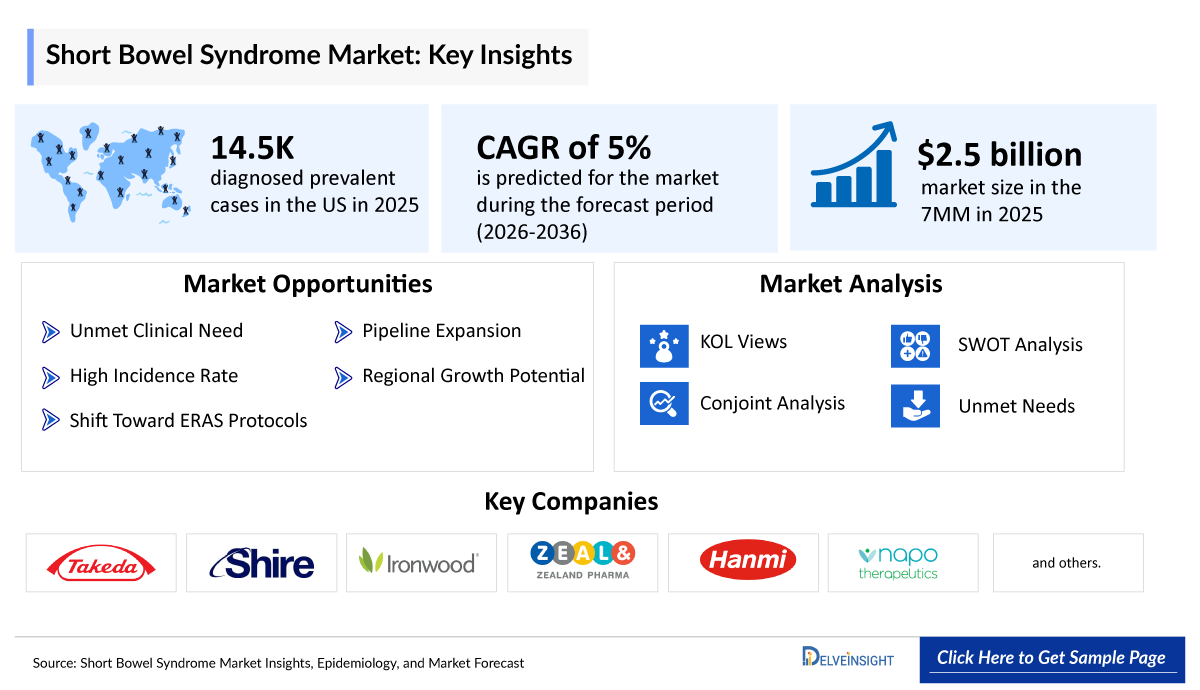
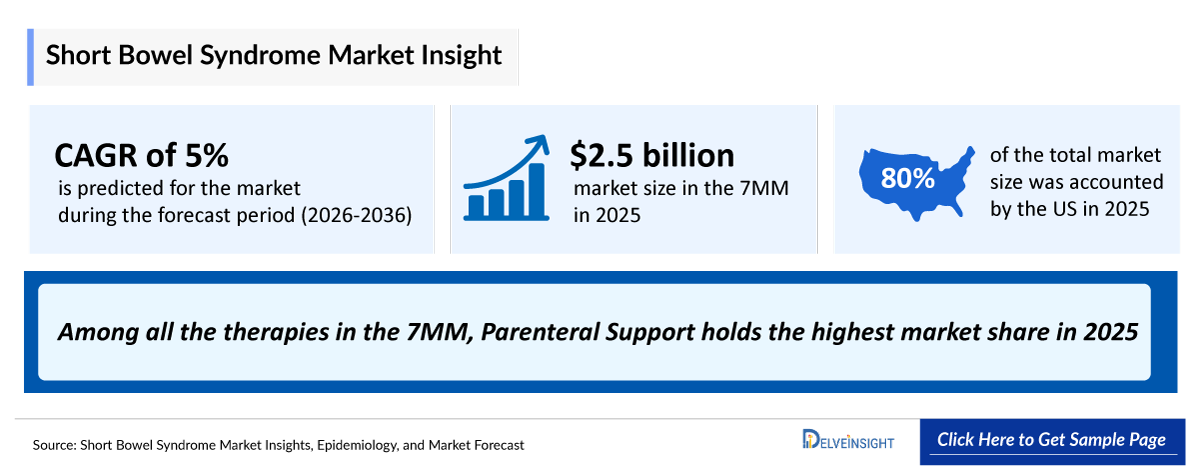
- 2025 SBS Market Size: ~USD 2,500 million
- 2036 Projected SBS Market Size: ~USD 4,100 million
- SBS Growth Rate (2026–2036): 5% CAGR
DelveInsight's ‘Short bowel syndrome (SBS) – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the SBS, historical and forecasted epidemiology, as well as the SBS market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The SBS market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, SBS patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in SBS and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
SBS Market CAGR (Study period/Forecast period) |
5% (2022–2036) |
|
SBS Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
SBS Companies |
|
|
SBS Therapies |
|
|
SBS Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Short bowel syndrome (SBS) Market
- Increasing cases of conditions like Crohn’s disease, necrotizing enterocolitis, and gastrointestinal surgeries, especially in neonates and the elderly, drive market growth.
- The launch of GLP-2 analogs such as apraglutide, glepaglutide, and HM15912 is expected to expand the SBS market by reducing Parenteral Nutrition dependence and improving patient outcomes
Short bowel syndrome (SBS) Understanding and Treatment Algorithm
SBS Overview and Diagnosis
Short bowel syndrome is a complex condition caused by the loss or dysfunction of portions of the small and/or large intestine, resulting in reduced absorption of nutrients, fluids, and electrolytes. It commonly presents with severe diarrhea, dehydration, malnutrition, and weight loss, and in some cases may become life-threatening, although it can often be managed despite having no cure. In adults, the normal small bowel length ranges from 275–850 cm, and SBS is typically defined as having less than 180–200 cm of remaining small intestine, often necessitating nutritional support. Most cases arise from major bowel resection, and prognosis largely depends on age and underlying disease, with around two-thirds of patients surviving the first year.
SBS is diagnosed through medical history, physical examination, and tests such as blood work, fecal fat analysis, and imaging, with additional procedures like endoscopy or liver biopsy to assess complications, while treatment is individualized and focuses on maintaining nutrition, promoting intestinal adaptation, and managing complications using total parenteral nutrition (TPN), enteral feeding, dietary modifications, medications, and, in severe cases, surgery or intestinal transplantation.
Further details are provided in the report.
Current SBS Treatment Landscape
Treatment of SBS is complex and individualized, focusing on maintaining nutrition, enhancing intestinal adaptation, and managing complications through approaches such as total parenteral nutrition (TPN), enteral feeding, dietary modifications, oral rehydration, medications, and, in severe cases, surgery or intestinal transplantation. Mild cases may be managed with diet and anti-diarrheal therapy, while many patients require TPN, though newer therapies like growth hormone and GLP-2 analogs have reduced long-term dependence. Nutritional management is central, with gradual transition from TPN to enteral or oral feeding as intestinal function improves, supported by tailored diets, small frequent meals, and supplementation. Medications include anti-diarrheals, acid-suppressing agents, and absorption-enhancing drugs, with antibiotics or enzyme therapy used when needed. Surgical options, reserved for refractory cases, include bowel-lengthening procedures (e.g., STEP, Bianchi) and, in severe cases, small bowel transplantation, particularly for patients with TPN-related complications or inadequate nutritional status.
Further details related to country-based variations are provided in the report.
Short bowel syndrome (SBS) Unmet Needs
The section “unmet needs of SBS” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Limited therapeutic options in SBS
- Therapeutic gaps across pediatric
- Long-term nutritional independence
- High cost and disturbed quality of life
and others…..
Note: Comprehensive unmet needs insights in SBS and their strategic implications are provided in the full report.
Short bowel syndrome (SBS) Epidemiology
Key Findings from SBS Epidemiological Analysis and Forecast
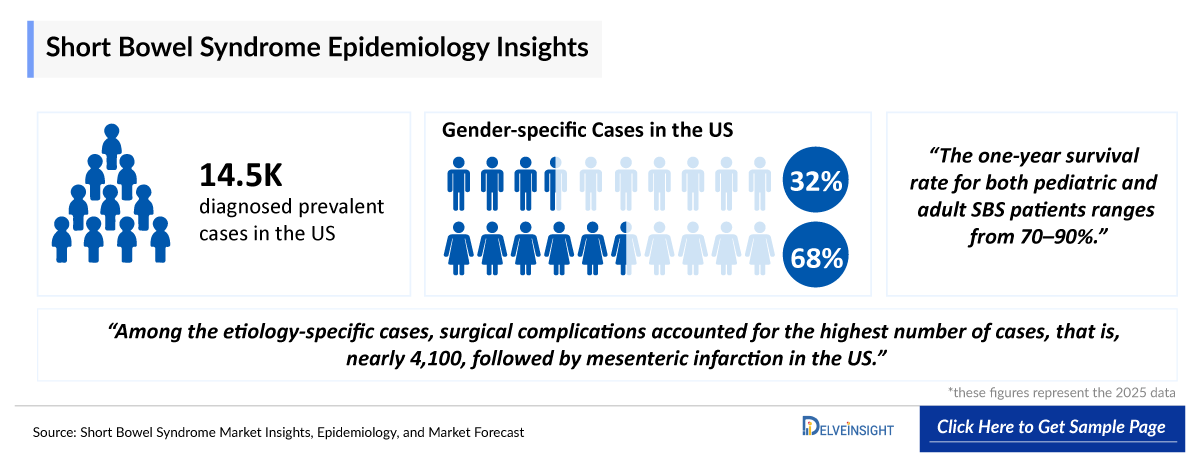
- US is recognized for having one of the world’s most advanced HPN programs, which plays a crucial role in supporting long-term survival for patients with chronic intestinal failure. In 2025, among the 7MM, the US had the highest number of diagnosed prevalent cases of SBS. According to DelveInsight estimates, there were 14,500 diagnosed prevalent cases of SBS in the US in 2025, which are projected to increase by 2036.
- SBS is more commonly diagnosed in women than men, with over 60% of cases occurring in females, compared to more than 30% in males in the US. Gender-specific diagnosed prevalence is expected to increase for both groups during the forecast period.
- SBS prevalence is higher in adults due to chronic conditions like Crohn’s disease and mesenteric ischemia. In contrast, pediatric SBS incidence is higher because it often results from acute congenital or neonatal conditions such as necrotizing enterocolitis and intestinal malformations, which occur more frequently early in life.
- In the US, adults now represent the largest share (~85%) of diagnosed prevalent SBS cases, a shift driven by rising surgical volumes, an aging population, and improved post-surgical survival over the past two decades. Pediatric SBS survivors transitioning into adulthood further expand the chronic adult SBS population, adding to prevalence beyond new adult-onset cases.
Short bowel syndrome (SBS) Drug Analysis & Competitive Landscape
The SBS drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the SBS treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the SBS market.
Approved Therapies for SBS
GATTEX/REVESTIVE (teduglutide): Takeda (Shire Pharmaceuticals)
Teduglutide is a novel, recombinant analogue of human GLP-2, a naturally occurring protein involved in the rehabilitation of the intestinal lining. It has been developed to reduce dependence on parenteral nutrition in adult patients with SBS. Teduglutide has received ODD for the treatment of SBS from the EMA and the US FDA.
Note: Detailed marketed therapies assessment will be provided in the final report.
Short bowel syndrome (SBS) Pipeline Analysis
Apraglutide: Ironwood Pharmaceuticals (VectivBio)
Apraglutide is an investigational, next-generation, long-acting synthetic GLP-2 analog being developed for a range of rare gastrointestinal diseases where GLP-2 can play a central role in addressing disease pathophysiology, including SBS-IF.
In August 2025, according to the Q2 2025 corporate highlights, following discussions with the US FDA in April 2025, the company is finalizing a confirmatory apraglutide Phase III trial design for patients with SBS who are dependent on parenteral support and plans to align with the FDA in Q4 of 2025. Pending alignment with the FDA, Ironwood expects to initiate a confirmatory Phase III trial in the 1H of 2026.
Glepaglutide (ZP1848): Zealand Pharma
Glepaglutide is a long-acting GLP-2 analog in development as a potential treatment option for SBS. Glepaglutide is being developed as a liquid product in an autoinjector designed for subcutaneous administration, aimed to reduce or eliminate the need for parenteral support in people living with SBS. The US FDA has granted ODD for glepaglutide for the treatment of SBS.
As per the Q2 2025 presentation in August 2025, the company had completed the Type A meeting with the US FDA, ensuring the alignment on the design of EASE-5, and anticipates the initiation of the Phase III trial (EASE-5) in H2 2025 to support resubmission in the US.
|
Competitive Landscape of Emerging Therapies | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
Apraglutide |
Ironwood Pharmaceuticals (VectivBio) |
III |
SBS-IF |
Subcutaneous |
GLP-2 receptor agonist |
Synthetic 33-amino-acid peptide |
2028 |
|
Glepaglutide (ZP1848) |
Zealand Pharma |
III |
SBS |
Subcutaneous |
GLP-2 receptor agonist |
Peptide |
Information is available in the full report |
|
Sonefpeglutide (HM15912) |
Hanmi Pharmaceutical |
II |
SBS-IF |
Subcutaneous |
GLP-2 analog that acts as a GLP-2 receptor agonist |
Peptide |
Information is available in the full report |
|
MYTESI (crofelemer) |
Napo Therapeutics (family company of Jaguar Health) |
II |
SBS-IF without CIC |
Oral |
Dual inhibitor of intestinal chloride channels (CFTR and CaCC) |
Plant-derived oligomeric proanthocyanidin |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Short bowel syndrome (SBS) Key Players, Market Leaders and Emerging Companies
- Takeda
- Ironwood Pharmaceuticals (VectivBio)
- Zealand Pharma
- Hanmi Pharmaceutical
- Napo Therapeutics (family company of Jaguar Health), and others
Short bowel syndrome (SBS) Drug Updates
- In April 2025, Ironwood Pharmaceuticals announced that, based on a recent discussion with the US FDA, a confirmatory Phase III trial is needed to seek approval of apraglutide for patients with SBS-IF who are dependent on parenteral support. While continuing to advance apraglutide, Ironwood has engaged Goldman Sachs to explore strategic alternatives for the company to maximize value for stockholders.
- In April 2025, Ironwood Pharmaceuticals announced that the company presented real-world data during the 2025 Digestive Disease Week (DDW) meeting. These data focused on the adoption and utilization of recently approved ICD-10 (International Statistical Classification of Diseases, 10th Revision) codes among SBS patients dependent on parenteral support.
- As per the Q2 2025 presentation in August 2025, the company had completed the Type A meeting with the US FDA, ensuring the alignment on the design of EASE-5, and anticipates the initiation of the Phase III trial (EASE-5) in H2 2025 to support resubmission in the US. The company is anticipating the potential approval of glepaglutide for the treatment of adult patients with SBS in H1 2026 in Europe. Additionally, the company is engaging in partnership discussions for future commercialization.
Short bowel syndrome (SBS) Market Outlook
The short bowel syndrome (SBS) market is evolving from reliance on total parenteral nutrition (TPN) the current cornerstone despite its cost, risks, and long-term burden toward advanced therapies that reduce dependence on parenteral support. Growing use of GLP-2 analogs such as GATTEX/REVESTIVE, along with emerging next-generation agents like apraglutide offering improved efficacy and convenient dosing, is driving this shift. With declining use of older growth hormone therapies and a strong focus on achieving intestinal autonomy, the SBS market is poised for significant growth, supported by innovation in biologics and ongoing pipeline advancements.
SBS treatment remains limited to parenteral support and GATTEX, with parenteral support dominating as the standard of care. Currently, it drives the highest revenue given its widespread use and comparatively lower cost, underscoring the market’s dependence on supportive rather than disease-modifying therapies.
- Among all the therapies in the 7MM, Parenteral Support holds the highest market share in 2025, By 2036, Parenteral Support is projected to maintain its dominant position, continuing to capture the largest market share.
- The SBS treatment market in the United States is experiencing steady growth, driven primarily by the rising prevalence of gastrointestinal surgeries, which is a major risk factor for SBS. In 2025, the United States market was valued at USD 2,000 million and is projected to increase by 2036.
- The SBS market across EU4 and the UK is projected to expand steadily from USD 200 million in 2025 through 2036, supported by favorable orphan drug incentives, well-established healthcare infrastructure, and a growing shift toward disease-modifying therapies in place of long-term parenteral nutrition.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in SBS (2022–2036 Forecast)
The SBS market comprises Peptides, and others, each targeting different aspects of tumor growth and progression.
Peptide: Peptide-based therapies are emerging as innovative treatment strategies, targeting key pathways involved in intestinal adaptation and absorption to reduce dependence on parenteral nutrition and improve clinical outcomes.
Short bowel syndrome (SBS) Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the SBS drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Short bowel syndrome (SBS)
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
The United States
|
US Reimbursement of Therapies Approved for SBS | |
|
Drug/Therapy |
Access Program |
|
GATTEX |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
SBS therapies Price Scenario & Trends
Pricing and analogue assessment of SBS therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of SBS Approved Drugs
GATTEX ranked as the 11th most expensive drug in the US in 2020, with a monthly cost of approximately USD 40,450 (as of March 2020) for 30 vials. Since GATTEX is typically administered on an ongoing basis, the total cost of treatment can be difficult to define. This becomes especially relevant when patients do not show the expected clinical response; however, it is important to consider that some individuals may experience a significant delayed response even after 24 months of therapy. For our analysis, we used a 12-month treatment duration, as patients typically remain on therapy for 6 to 24 months. Based on this timeframe and standard daily subcutaneous dosing, the estimated annual cost of therapy may reach approximately USD 485,400.
Industry Experts and Physician Views for Short bowel syndrome (SBS)
To keep up with SBS market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the SBS emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in SBS, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the University of California, Imperial College London, Hokkaido University, and Cholangiocarcinoma Foundation, etc. were contacted.Their opinion helps understand and validate current and emerging SBS therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in SBS.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“About 75% of cases of SBS develop after a single, massive resection of bowel; whereas, the remaining 25% occur after multiple resections. Around two-thirds of patients who develop SBS survive their initial hospitalization, and a similar number survive their first year after developing SBS. Age and underlying disease primarily determine a patient's long-term outcome.” |
|
Italy |
“SBS places a substantial economic and quality-of-life burden on patients and healthcare systems, highlighting the need for therapies that can reduce dependence on parenteral nutrition and improve long-term outcomes.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of SBS, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of SBS, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the SBS market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM SBS market.
Report Insights
- Short bowel syndrome (SBS) Patient population forecast
- Short bowel syndrome (SBS) Therapeutics market size
- Short bowel syndrome (SBS) Pipeline analysis
- Short bowel syndrome (SBS) Market size and trends
Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up forecasting
- Artificial Intelligence (AI)-enabled Market research report
- 11-year forecast
- Short bowel syndrome (SBS) Market outlook (North America, Europe, Asia-Pacific)
- Patient Burden trends (by geography)
- Short bowel syndrome (SBS) Treatment addressable Market (TAM)
- Short bowel syndrome (SBS) Competitve Landscape
- Short bowel syndrome (SBS) Major companies Insights
- Short bowel syndrome (SBS) Price trends and analogue assessment
- Short bowel syndrome (SBS) Therapies and Drug Adoption/Uptake
- Short bowel syndrome (SBS) Therapies Peak Patient Share Analysis
Report Assessment
- Short bowel syndrome (SBS) Current treatment practices
- Short bowel syndrome (SBS) Unmet needs
- Short bowel syndrome (SBS) Clinical development Analysis
- Short bowel syndrome (SBS) Emerging drugs product profiles
- Short bowel syndrome (SBS) Market attractiveness
- Short bowel syndrome (SBS) Qualitative analysis (SWOT and conjoint analysis)
FAQs
Market Insights
- What was the SBS market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of SBS?
- What are the disease risks, burdens, and unmet needs of SBS? What will be the growth opportunities across the 7MM concerning the patient population with SBS?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of SBS? What are the current guidelines for treating SBS in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the SBS market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets withing the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.