Systemic Lupus Erythematosus Market Summary
Systemic Lupus Erythematosus (SLE) Insights and Trend
- According to DelveInsight’s analysis, SLE market size was found to be ~USD 3,100 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- Biologic therapies have traditionally been reserved for patients who show an inadequate response to conventional treatments or who are dependent on corticosteroids. However, the 2023 EULAR guidelines have shifted away from this stepwise “add-on” approach, recognizing that biologics may be introduced earlier in the treatment course when clinically appropriate.
- Until 2021, the treatment landscape for Systemic Lupus Erythematosus remained largely stagnant, with belimumab (BENLYSTA) as the only widely approved biologic, now expanded to include adult and pediatric patients (=5 years) with active lupus nephritis, across IV and SC formulations, while more recently, additional FDA-approved biologics such as anifrolumab (SAPHNELO) have begun to broaden therapeutic options.
- The SLE investigational pipeline is rich with mechanistically diverse agents at various stages of clinical development. Key late-stage and emerging therapies include obinutuzumab, ianalumab, dapirolizumab pegol, upadacitinib, deucravacitinib, litifilimab, enpatoran, and others.
- Furthermore, CAR T-cell therapies are emerging in SLE as a potentially transformative approach, enabling targeted B-cell depletion and immune reset with the possibility of durable, drug-free remission in refractory disease. Key candidates include rapcabtagene autoleucel (YTB-323), zolacabtagene autoleucel, resecabtagene autoleucel (CABA-201), and obecabtagene autoleucel (obe-cel, AUCATZYL), marking a shift away from lifelong immunosuppression toward one-time, disease-modifying intervention.
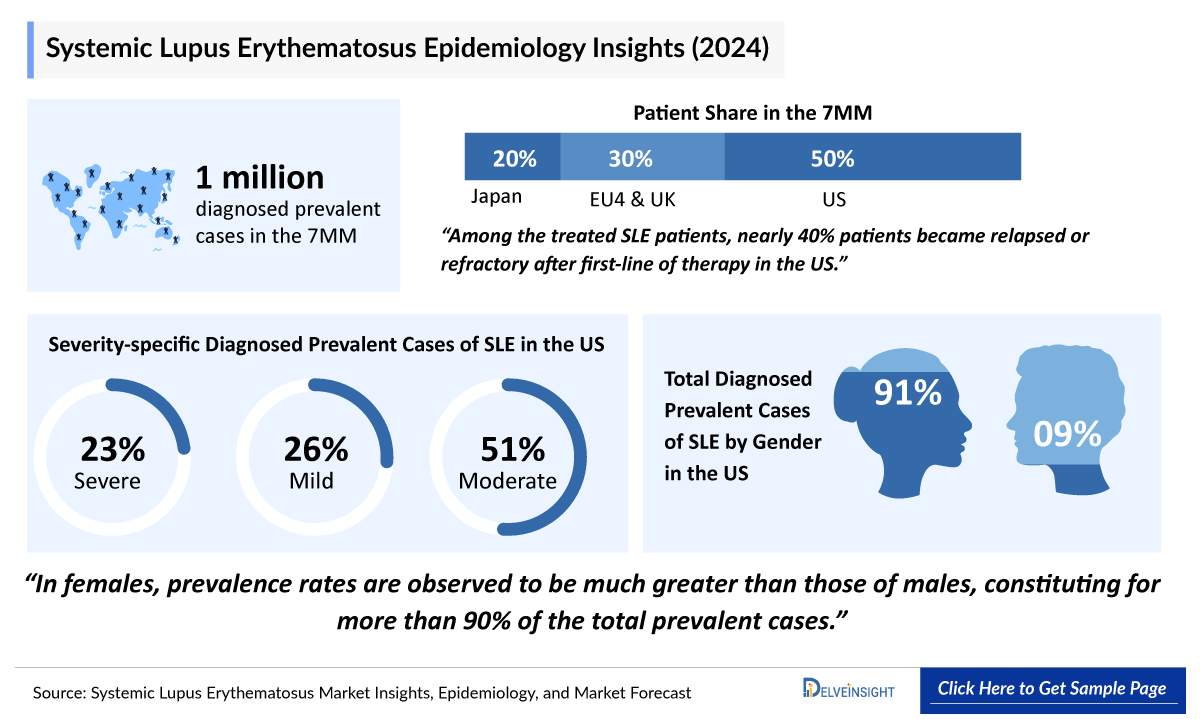
- In 2026, gender-based diagnosed prevalent SLE cases across the USA, are expected to include 9% cases in males and 91% cases in females, reinforcing the disease’s strong female predominance and highlighting the role of hormonal and immune-mediated mechanisms in shaping SLE susceptibility, driving its population-level burden.
- Delayed diagnosis remains a key unmet need in SLE due to heterogeneous, non-specific early symptoms and the lack of reliable biomarkers for early detection, disease stratification, and flare prediction.
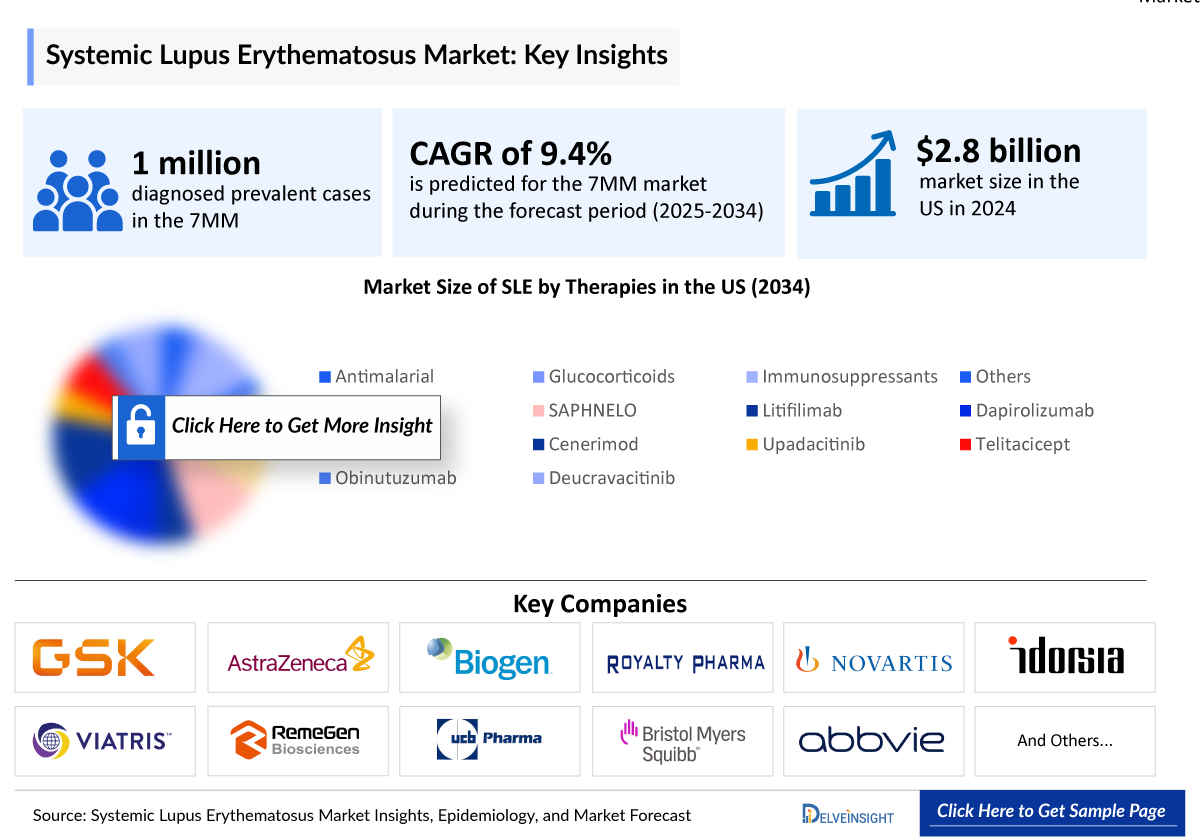
Systemic Lupus Erythematosus (SLE) Market Size and Forecast in the 7MM
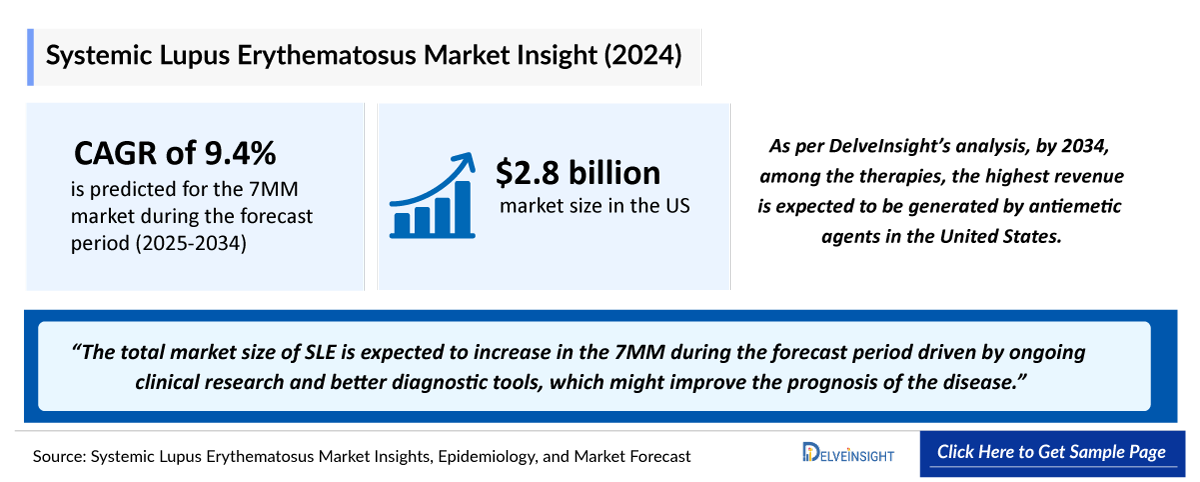
- 2025 Systemic Lupus Erythematosus (SLE) Market Size: ~USD 3,100 million
- 2036 Projected Systemic Lupus Erythematosus (SLE) Market Size: ~USD 8,400 million
- Systemic Lupus Erythematosus (SLE) Growth Rate (2026–2036): 9.8% CAGR
DelveInsight's ‘Systemic Lupus Erythematosus (SLE)– Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the SLE, historical and forecasted epidemiology, as well as the SLE market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The SLE market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates SLE patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in SLE and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Systemic Lupus Erythematosus (SLE) Market CAGR (Forecast period) |
9.8% (2026–2036) |
|
Systemic Lupus Erythematosus (SLE) Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Systemic Lupus Erythematosus (SLE) Companies |
|
|
Systemic Lupus Erythematosus (SLE) Therapies |
|
|
Systemic Lupus Erythematosus (SLE) Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Systemic Lupus Erythematosus (SLE) Market
Rising Systemic Lupus Erythematosus (SLE) Prevalence
The overall prevalence of SLE is increasing, particularly among females, driven by improved disease awareness, advances in diagnostic practices, and better survival rates resulting from earlier diagnosis and improved treatment.
Rising Opportunities in SLE
Advances in targeted biologics and small molecules including B-cell inhibitors, interferon inhibitors, and JAK/TYK2/S1P modulators are reshaping the SLE treatment landscape, increasing competition while addressing unmet needs in moderate-to-severe and refractory disease.
Systemic Lupus Erythematosus (SLE) Understanding and Treatment Algorithm
Systemic Lupus Erythematosus (SLE) Overview and Diagnosis
SLE is a chronic autoimmune disease that can affect virtually any organ system, with manifestations ranging from mild skin and joint involvement to severe neurological and systemic complications. SLE is the most common form of lupus, accounting for nearly 70% of all cases. The disease has a multifactorial origin, with genetic susceptibility and environmental triggers contributing to immune dysregulation, where the immune system mistakenly attacks the body’s own tissues. While skin and joints are most commonly affected, organ involvement varies widely among patients. SLE typically follows a relapsing–remitting course, with periods of flares alternating with remission or low disease activity.
The diagnosis of SLE is complex due to its heterogeneous clinical presentation and lack of a single definitive diagnostic marker. Diagnosis relies on a comprehensive evaluation of clinical manifestations and serological testing, including antinuclear antibodies (ANA), anti-dsDNA, and anti-Smith antibodies, supported by complement levels and other laboratory findings. Imaging may be used to assess organ involvement, while biopsies particularly renal biopsy in suspected lupus nephritis—provide histopathological confirmation and help guide management decisions.
Note: Further details are provided in the report.
Systemic Lupus Erythematosus (SLE) Treatment
The management of SLE aims to achieve remission or low disease activity, prevent organ damage, and improve quality of life through a personalized, multidisciplinary approach. Hydroxychloroquine is recommended for all patients as foundational therapy, while NSAIDs may be used for mild symptoms. Glucocorticoids are reserved for disease flares and are used at the lowest effective dose with early tapering to reduce long-term toxicity.
Treatment is escalated based on disease severity and organ involvement using immunosuppressants such as mycophenolate mofetil, azathioprine, methotrexate, and cyclophosphamide. Biologic therapies are increasingly important in refractory or moderate-to-severe disease, including belimumab (B-cell targeted) and anifrolumab (type I interferon receptor inhibitor), with rituximab used off-label in resistant cases. In lupus nephritis, voclosporin is also used as part of combination therapy to improve renal outcomes. Organ-specific treatment is guided by clinical manifestations, with more intensive immunosuppression required for severe systemic, hematologic, or neuropsychiatric involvement.
Note: Further details related to country-based variations are provided in the report.
Systemic Lupus Erythematosus (SLE) Unmet Needs
The section “unmet needs of SLE” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Heterogeneous and unpredictable nature of SLE
- Delayed diagnosis and lack of validated biomarkers
- Limited FDA-approved therapies in SLE
- Restricted reimbursement and access barriers and others…..
Note: Comprehensive unmet needs insights in SLE and their strategic implications are provided in the full report.
Systemic Lupus Erythematosus (SLE) Epidemiology
Key Findings from Systemic Lupus Erythematosus (SLE) Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, the total diagnosed prevalent cases of SLE in the 7MM were approximately 911,600 in 2025.
- It has been observed that the prevalence of SLE is higher in adults aged 40-59 years, ~42% as compared to other age groups (0-18 years, 19-39 years and above 60+ years) in the 7MM.
- In 2025, among severity of SLE in the US, moderate SLE was the most prevalent subtype (~261,600 cases), followed by mild SLE cases.
- The total diagnosed prevalent population of SLE across EU4 and the UK has shown a steady upward trend which accounts for ~309,400 cases in 2025.
- The SLE is found to be more prevalent in females than in males.
Systemic Lupus Erythematosus (SLE) Drug Chapters & Competitive Analysis
The SLE drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the SLE treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the SLE therapeutics market.
Approved Therapies for Systemic Lupus Erythematosus (SLE)
Belimumab (BENLYSTA): GlaxoSmithKline
Belimumab (BENLYSTA) is a human IgG1? monoclonal antibody that inhibits BLyS, thereby reducing the survival of autoreactive B cells and autoantibody production in lupus. It is indicated as an add-on therapy for patient’s =5 years with active SLE and lupus nephritis, helping to reduce disease activity and flares and enabling steroid sparing. It is administered as an intravenous infusion or subcutaneous injection and is not recommended for severe active CNS lupus. It is the first and only biologic approved for both the chronic autoimmune disease SLE and lupus nephritis.
In November 2017, GSK announced that the European Commission has approved a new SC formulation of belimumab as an add-on therapy in adult patients with active autoantibody-positive SLE despite standard therapy. In March 2011, the US FDA first approved belimumab for the treatment of patients with SLE of adult patients with active, autoantibody-positive SLE who are receiving standard therapy.
|
Marketed/Approved Competitive Landscape of SLE | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Belimumab (BENLYSTA) |
GlaxoSmithKline |
SLE |
Monoclonal antibody |
SC and IV |
BLyS-specific inhibitor |
US: 2020 EU: 2021 JP: 2021 |
|
Anifrolumab (SAPHNELO) |
AstraZeneca |
Moderate to severe SLE |
Monoclonal antibody |
IV infusion |
Type I interferon receptor antagonist |
US: 2021 EU: 2022 JP: 2021 |
SLE Pipeline Analysis
Obinutuzumab (GAZYVA/GAZYVARO): Genentech (Roche)
Obinutuzumab injection is an engineered monoclonal antibody that targets a protein called CD20 on the surface of the lymphoma and leukemia cells. It binds to Type 2 CD20 antibodies. This allows obinutuzumab to have a much higher induction of antibody-dependent cytotoxicity and a higher direct cytotoxic effect than the classic CD20 antibodies. Upon binding to CD20, it mediates B-cell lysis through the engagement of immune effector cells by directly activating intracellular death signaling pathways and/or activation of the complement cascade. GAZYVA is marketed as GAZYVARO in Europe. In February 2026, Roche filed regulatory applications in both the US and Europe for obinutuzumab in SLE.
|
Emerging Competitve Landscape of SLE | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
Upadacitinib (RINVOQ) |
AbbVie |
III |
SLE |
Oral |
JAK 1 inhibitors |
Small molecule |
2028 |
|
Obinutuzumab (GAZYVA/GAZYVARO) |
Genentech (Roche) |
III |
SLE |
IV infusion |
TSLP inhibitor |
Monoclonal antibody |
Information is available in the full report |
|
Rapcabtagene autoleucel (YTB323) |
Novartis |
III |
SLE |
IV infusion |
CD19-targeted |
CAR-T cell therapy |
Information is available in the full report |
|
Dapirolizumab Pegol |
UCB Pharma/Biogen |
III |
SLE |
IV infusion |
CD40L Inhibitor |
Monoclonal antibody |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Systemic Lupus Erythematosus (SLE) Key Players, Market Leaders and Emerging Companies
- GlaxoSmithKline
- AstraZeneca
- Genentech (Roche)
- AbbVie
- Bristol Myers Squibb
- Novartis
- MorphoSys
- UCB Pharma
- Biogen
- Juno Therapeutics
- Johnson & Johnson Innovative Medicine
- Idorsia Pharmaceuticals
- Viatris
- Merck and others
Systemic Lupus Erythematosus (SLE) Drug Updates
- In April 2026, Roche announced that the US FDA has accepted the company sBLA for obinutuzumab for the treatment of SLE. The submission is supported by positive Phase III ALLEGORY study results, which demonstrated a statistically significant and clinically meaningful improvement in the primary endpoint. The FDA is expected to make a decision on an approval by December 2026.
- As per Novartis’s 2026 interim financial report, the company anticipates Phase III trials (SIRIUS-SLE 1 and SIRIUS-SLE 2) readouts in 2027, with regulatory submission of Ianalumab for SLE in 2028.
- In March 2026, Johnson & Johnson announced the US FDA gave FTD for nipocalimab in SLE treatment.
- As of May 2025, Viatris anticipates Phase III data readouts for Cenerimod in SLE, with a potential regulatory submission expected by 2027 for both studies, OPUS-1 and OPUS-2
Drug Class Insights
Systemic Lupus Erythematosus (SLE) Market Outlook
SLE is a chronic autoimmune disease characterized by immune dysregulation leading to systemic inflammation and damage across multiple organs, including the skin, joints, kidneys, blood vessels, heart, brain, and lungs. The disease follows a relapsing–remitting course with periods of flares and remission, making long-term management complex and highly individualized. Its heterogeneous presentation often requires a combination of clinical assessment and targeted therapy based on disease severity and organ involvement.
Management is guided by EULAR/ACR recommendations, with hydroxychloroquine as the foundational therapy for most patients due to its ability to reduce flares and long-term disease activity. Mild disease is additionally managed with NSAIDs and low-dose corticosteroids for symptomatic control. In moderate-to-severe disease, treatment is escalated to immunosuppressants such as mycophenolate mofetil, azathioprine, methotrexate, and cyclophosphamide, which help control immune-mediated organ damage. Glucocorticoids are used for acute flares but are tapered early due to long-term toxicity concerns.
Biologic therapies are increasingly used in patients with inadequate response to conventional treatment. Belimumab, a B-cell targeted agent, helps reduce autoantibody production and disease activity, while anifrolumab, an interferon receptor inhibitor, targets type I interferon signaling and is particularly effective in cutaneous and musculoskeletal manifestations. Rituximab is also used off-label in refractory cases. Despite these advances, treatment remains individualized, with many patients requiring combination therapy and ongoing adjustment based on disease activity and organ involvement.
Emerging therapies in systemic lupus erythematosus (SLE) are increasingly focused on targeted immune modulation aimed at improving efficacy while reducing steroid dependence. Key late-stage and investigational agents include B-cell directed therapies such as obinutuzumab and ianalumab, type I interferon pathway inhibitors like litifilimab, and FcRn blockade via nipocalimab. In addition, novel oral and pathway-specific agents such as deucravacitinib (TYK2 inhibitor) and cenerimod (S1P receptor modulator) are being evaluated for broader immune regulation and disease control. CAR-T cell therapies targeting CD19-positive B cells are also emerging as a potential disease-modifying approach in refractory SLE, showing early promise in achieving deep and sustained remission.
- According to the estimates, the largest market size of SLE was captured by the United States, i.e., ~USD 3,100 million in 2025.
- The SLE market is evolving as treatment focus shifts from broad immunosuppressants and corticosteroids toward targeted therapies, with biologics and emerging pathway-specific agents (including B-cell and interferon-targeted therapies) driving adoption due to improved disease control, steroid-sparing potential, and expanding use across moderate-to-severe disease.
- Regional market dynamics show the US leading in SLE treatment uptake and innovation, while Europe and Japan demonstrate steady growth, supported by improved diagnosis, wider access to biologics, and increasing adoption of novel targeted mechanisms across both adult and selected pediatric populations.
Note: Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Systemic Lupus Erythematosus (SLE) (2022–2036 Forecast)
The SLE market comprises corticosteroids, antimalarials, conventional immunosuppressants, biologics, and emerging targeted therapies, each addressing distinct immune pathways such as B-cell activation (BLyS/BAFF pathway), type I interferon signaling, and broader T- and B-cell mediated immune dysregulation underlying systemic disease activity.
Monoclonal Antibodies: Monoclonal antibodies such as Belimumab, a BLyS inhibitor, and Anifrolumab, a type I interferon receptor antagonist, target key B-cell and interferon pathways in SLE, improving disease control with differing efficacy and safety profiles.
Small Molecules: Oral small molecules such as cenerimod (S1P1 modulator), deucravacitinib (TYK2 inhibitor), and upadacitinib (JAK1 inhibitor) target multiple immune pathways in SLE, reflecting a shift toward oral multi-pathway immune modulation in treatment.
CAR T therapies: CD19-directed CAR-T therapies, including rapcabtagene autoleucel, zolacabtagene autoleucel, and resecabtagene autoleucel, show strong potential in SLE by inducing deep B-cell depletion and durable remission, though safety risks such as CRS and ICANS require intensive monitoring.
Note: Further details will be provided in the report….
Systemic Lupus Erythematosus (SLE) Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the SLE market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in SLE is expected to increase with the growing adoption of targeted biologics such as Belimumab and Anifrolumab, driven by their ability to address key immune pathways and reduce disease activity and steroid dependence. Belimumab is likely to maintain broader uptake due to its established safety profile and flexible formulations, while anifrolumab may see more selective use in patients with high interferon–driven disease.
Note: Further detailed analysis of emerging therapies' drug uptake in the report…
Market Access and Reimbursement of Systemic Lupus Erythematosus (SLE)
The United States
|
The US Reimbursement for Systemic Lupus Erythematosus (SLE) Therapies | |
|
Drug |
Access Program |
|
Belimumab (BENLYSTA) |
|
|
Anifrolumab (SAPHNELO) |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Systemic Lupus Erythematosus (SLE) Therapies Price Scenario & Trends
Pricing and analogue assessment of SLE therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Systemic Lupus Erythematosus (SLE) Approved Drugs
Belimumab is available in two formulations, SC and IV, and the average cost of both was considered for analysis. For the SC formulation, the dosing schedule is 400 mg weekly for the first month, followed by 200 mg weekly thereafter. In the United States, the unit price for a 200 mg SC dose was USD 1,153. The IV formulation is dosed at 10 mg/kg every 2 weeks for the first three doses, then every 4 weeks. The unit price for 400 mg IV was USD 2,161, and costs were calculated separately for pediatric and adult patients before averaging. All US cost data were originally based on 2023 prices and adjusted to reflect 2022 values.
Further details are provided in the final report….
Industry Experts and Physician Views for Systemic Lupus Erythematosus (SLE)
To keep up with SLE market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the SLE emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in SLE, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the University of Verona, Berlin Institute of Health at Charité, and the University of Nottingham, etc. were contacted. Their opinion helps understand and validate current and emerging SLE therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in SLE.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“There is increasing enthusiasm for mechanism-based targeted therapies, including B-cell depletion strategies, BAFF inhibition, interferon pathway targeting, and emerging immune reprogramming approaches such as CAR-T therapies. These are viewed as promising avenues for patients with highly refractory SLE who fail conventional immunosuppressants and biologics.” |
|
Italy |
““From a clinical perspective, if a patient with Systemic Lupus Erythematosus is achieving stable disease control on a combination of Belimumab and Hydroxychloroquine, maintaining this regimen would be considered an appropriate approach to sustain disease stability and minimize the risk of long-term organ damage.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of SLE, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of SLE, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the SLE market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM SLE market.
Report Insights
- SLE Patient Population Forecast
- SLE Therapeutics Market Size
- SLE Pipeline Analysis
- SLE Market Size and Trends
- SLE Market Opportunity (Current and forecasted)
Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- SLE Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- SLE Treatment Addressable Market (TAM)
- SLE Competitive Landscape
- SLE Major Companies Insights
- SLE Price Trends and Analogue Assessment
- SLE Therapies Drug Adoption/Uptake
- SLE Therapies Peak Patient Share analysis
Report Assessment
- SLE Current Treatment Practices
- SLE Unmet Needs
- SLE Clinical Development Analysis
- SLE Emerging Drugs Product Profiles
- SLE Market Attractiveness
- SLE Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Market Insights
- What was the SLE market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of SLE?
- What are the disease risks, burdens, and unmet needs of SLE? What will be the growth opportunities across the 7MM concerning the patient population with SLE?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of SLE? What are the current guidelines for treating SLE in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the SLE market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.