Tenosynovial Giant Cell Tumors Market Summary
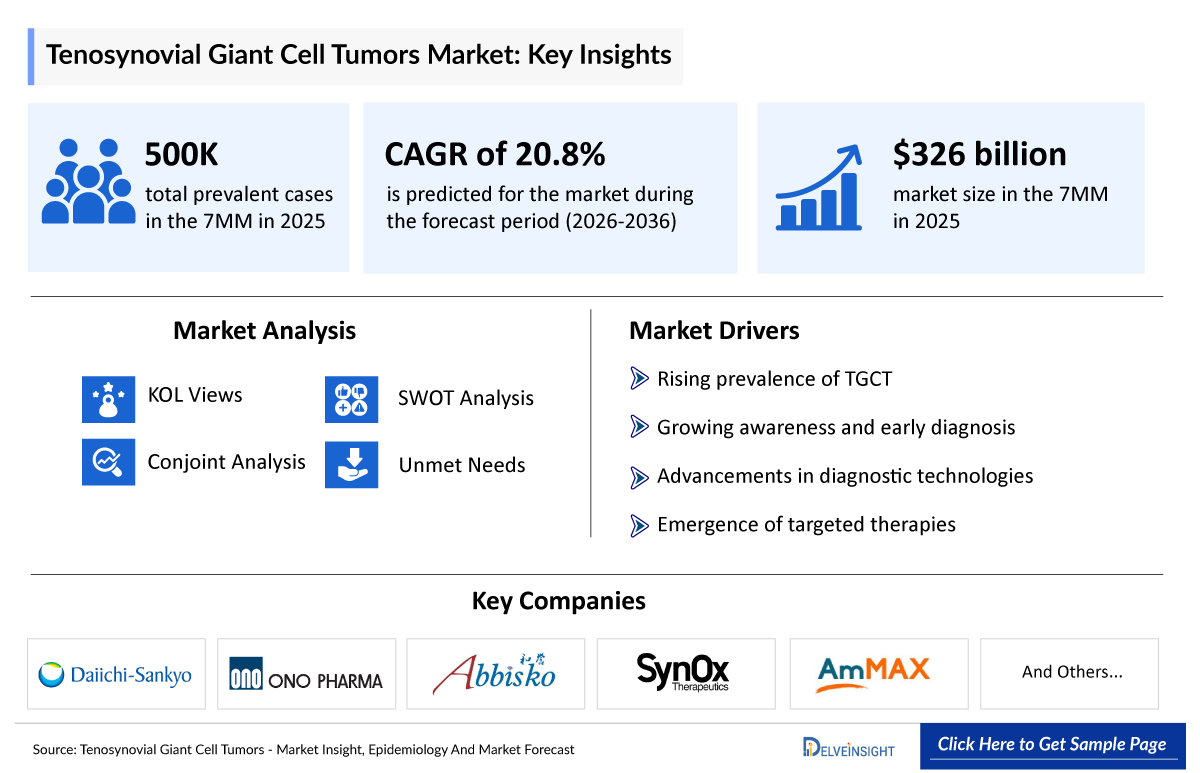
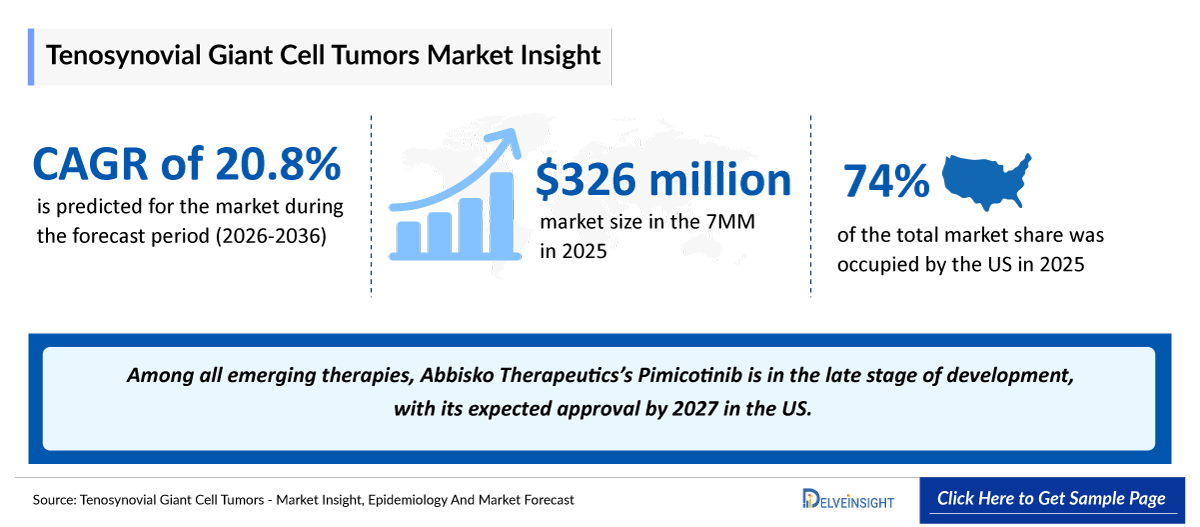
- According to DelveInsight’s analysis, the Tenosynovial Giant Cell Tumors Treatment Market Size was found to be ~USD 326 Million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- The leading Tenosynovial Giant Cell Tumors Companies such as Daiichi Sankyo, Ono Pharmaceutical, Abbisko Therapeutics, SynOx Therapeutics Limited, AmMax Bio and others.
Tenosynovial Giant Cell Tumor Market Insights and Trends
- TGCT are caused by a translocation of certain parts of chromosomes 1 and 2 causing the cells to overproduce a protein called colony-stimulating factor-1 or CSF-1. There are no environmental, genetic, occupational, lifestyle, demographic or regional risk factors that have been conclusively shown to be involved with the development of these tumors.
- TGCT have a wide clinical spectrum that affect patients of all ages. The signs and symptoms of Tenosynovial Giant Cell Tumor can vary depending upon the exact location involved and the subtype present.
- TGCT occurs in localized and diffuse forms and typically presents as a well-defined, rubbery, multinodular mass surrounded by a fibrous capsule. The cut surface shows variable coloration based on the relative amounts of fibrous tissue, hemosiderin, and lipid-laden macrophages. Clinically, patients often present with a slow-growing, painless swelling that may later become painful or edematous, commonly causing intermittent knee swelling.
- Localized TGCT grows slowly and usually affects small joints, particularly the fingers and tendon sheaths, and is often referred to as giant cell tumor of the tendon sheath (GCTTS). Diffuse TGCT grows more aggressively and typically involves large joints such as the knee, hip, ankle, shoulder, or elbow, and is also known as pigmented villonodular synovitis (PVNS). TGCT is rarely seen in the head and neck region.
- Historically, TGCT has been treated primarily with surgical synovectomy, using arthroscopic or open techniques, sometimes combined with adjuvant radiotherapy. Localized disease is generally curable with surgery, while diffuse TGCT is associated with high recurrence rates. Open surgical approaches in diffuse disease can lead to increased morbidity, including postoperative stiffness and pain.
- The diagnosis of Tenosynovial Giant Cell Tumors is based on a description of the symptoms and physical examinations. In addition, Magnetic Resonance Imaging is usually necessary in order to see the lesions and determine the best surgical intervention. Other tests that may help with the diagnosis include X-rays, synovial fluid sample from around the joints, and a biopsy of tissues from the joint.
- Surgical resection continues to be the mainstay of treatment for patients with localized and diffused TGCT. The significant rates of recurrence and risks associated with surgery point to the need for novel systemic treatments for patients with symptomatic advanced TGCT. The recent approval of a systemic therapy for selected adults with symptomatic TGCT underscores the need for improved and more coordinated multidisciplinary care.
Tenosynovial Giant Cell Tumor Market Size and Forecast
- 2025 TGCT Market Size: ~USD 326 Million
- 2036 Projected TGCT Market Size: ~USD 3,347 Million
- TGCT Growth Rate (2026–2036): 20.8% CAGR
Request for Unlocking the Sample Page of the "Tenosynovial Giant Cell Tumors Treatment Market"
Key Factors Driving the Tenosynovial Giant Cell Tumor Market Growth by DelveInsight
-
Increasing Prevalence and Diagnosis of TGCT
Rising awareness among healthcare professionals and advancements in diagnostic imaging are improving the identification of localized and diffuse TGCT cases.
-
Growing Demand for Targeted Therapies
The transition from traditional surgical approaches to targeted systemic therapies, particularly CSF1R inhibitors, is significantly driving market growth.
-
High Recurrence Rates Creating Unmet Medical Need
Diffuse TGCT often recurs after surgery, increasing the need for more effective and long-term treatment options.
-
Emergence of Novel CSF1R Inhibitors
Newly approved and emerging therapies targeting the CSF1/CSF1R pathway are expected to transform the treatment landscape and improve patient outcomes.
-
Strong Pipeline and Clinical Trial Activity
Pharmaceutical and biotechnology companies are actively developing innovative therapies with enhanced safety and efficacy profiles.
-
Advancements in Diagnostic Technologies
Increased use of MRI, biopsy techniques, and advanced imaging modalities is supporting early diagnosis and better treatment planning.
-
Growing Focus on Rare Disease Therapeutics
Increasing investments in orphan drug development and rare oncology disorders are accelerating research and commercialization efforts.
DelveInsight's ‘Tenosynovial Giant Cell Tumor Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the TGCT, historical and forecasted epidemiology, as well as the TGCT market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Tenosynovial Giant Cell Tumor Treatment Market Report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates Tenosynovial Giant Cell Tumors patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth Tenosynovial Giant Cell Tumors Market Size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key Tenosynovial Giant Cell Tumors Unmet Medical Needs and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Scope of the Tenosynovial Giant Cell Tumors Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Tenosynovial Giant Cell Tumors Market CAGR | |
|
Tenosynovial Giant Cell Tumors Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Tenosynovial Giant Cell Tumors Companies |
|
|
Tenosynovial Giant Cell Tumors Therapies |
|
|
Tenosynovial Giant Cell Tumors Market |
Segmented By
|
|
Tenosynovial Giant Cell Tumors Analysis |
|
Tenosynovial Giant Cell Tumor Understanding and Treatment Algorithm
Tenosynovial Giant Cell Tumor Overview and Diagnosis
EGFR is a protein in cells that helps them grow. A mutation in the gene for EGFR can make it grow too much, which can cause cancer. There are different types of EGFR mutations, including deletions or insertions and point mutations. In test results, individuals may be identified as having an EGFR 19 deletion or an EGFR L858R point mutation, which are the most common types of EGFR mutations. These mutations are typically treated the same way. Amongst the EGFR mutations that are tested for in lung cancer, a few rare types are treated differently than the more common EGFR mutations. The major example of this in lung cancer is EGFR exon 20 insertions. This is a type of EGFR mutation that does not respond to the typical treatment for EGFR-positive lung cancer, which are called Tyrosine Kinase Inhibitors.
In general, there are two ways to detect EGFR mutations. The best way is through comprehensive next-generation sequencing. This type of testing places tissue from a patient’s tumor (gathered from a biopsy) in a machine that looks for a large number of possible biomarkers at one time. There may be some situations where a patient cannot undergo the biopsy needed to perform NGS, so liquid biopsy is recommended. A liquid biopsy can look for certain biomarkers in a patient’s blood.
Current Tenosynovial Giant Cell Tumor Treatment Landscape
Treatment of Tenosynovial Giant Cell Tumor primarily involves surgery and targeted systemic therapies, depending on disease type and severity. Surgical excision or synovectomy remains the mainstay, particularly for localized TGCT, while diffuse or unresectable cases often require systemic treatment. Targeted therapies inhibiting the CSF1/CSF1R pathway, such as pexidartinib and vimseltinib, are the only approved options and have significantly improved outcomes by reducing tumor burden and alleviating symptoms. In select cases, off-label tyrosine kinase inhibitors like imatinib and sunitinib may also be used. Additionally, radiation therapy and supportive care can be considered based on disease progression, recurrence risk, and functional impairment.
Further details related to country-based variations are provided in the report.
Tenosynovial Giant Cell Tumor Unmet Needs
The section “unmet needs of Tenosynovial Giant Cell Tumor” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Need for Effective Treatment Options
- Limited Understanding of Pathogenesis
- Limited Access and Affordability
- High Recurrence Rates
- Significant Research Gaps in Tenosynovial Giant Cell Tumor
Tenosynovial Giant Cell Tumor Epidemiology
Key Findings from TGCT Epidemiological Analysis and Forecast
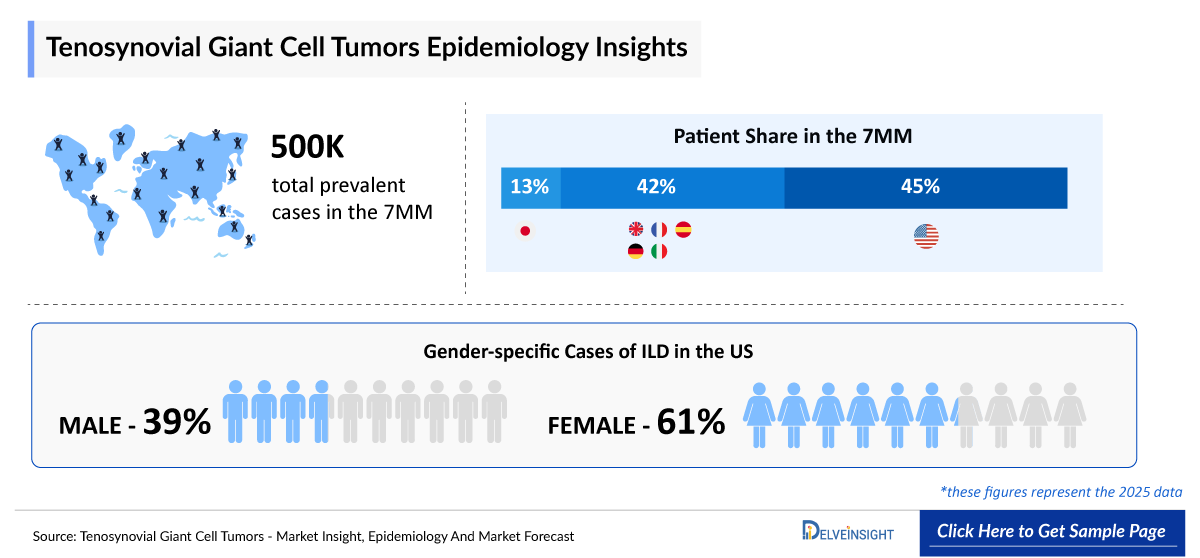
- According to DelveInsight’s estimates, the total number of Tenosynovial Giant Cell Tumors Incident Cases in the 7MM was nearly 580,000 cases in 2025 and is projected to increase during the forecasted period.
- The total number of Tenosynovial Giant Cell Tumors Prevalent Cases in the United States was nearly 250,290 in 2025.
- The Tenosynovial Giant Cell Tumors gender-specific prevalent population is segregated based on the tumor’s growth pattern, i.e., localized and diffused form. In the United States, ~77,920 males and ~120,782 females had localized TGCT in 2025. On the other hand, in diffused form there were ~24,170 and ~27,400 cases of males and females, respectively, in 2025.
- The giant cell tumors are detected in the knee, ankle, hip, and other depending upon localized or diffused. DelveInsight has estimated tumor localization of diffused TGCT to be maximum in the knee, with nearly 33,010 cases in 2025 in the United States, which might reach nearly 58,110 cases by 2036.
- TGCT most commonly occurs in large joints, with the knee being the most frequently affected site, followed by the hip, ankle, and shoulder. Localized TGCT often involves smaller joints such as the fingers, hands, and wrist, typically arising from the tendon sheath. In contrast, diffuse TGCT more commonly affects larger, weight-bearing joints and is associated with extensive synovial involvement. The distribution of disease is largely driven by synovial tissue presence rather than external risk factors, with no strong geographic variation reported.
Tenosynovial Giant Cell Tumor Drug Analysis & Competitive Landscape
The TGCT drug chapter provides a detailed, market-focused review of approved therapies and the emerging Tenosynovial Giant Cell Tumors Pipeline across Phase II–III Tenosynovial Giant Cell Tumors Clinical Trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the TGCT treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the TGCT therapeutics market.
Tenosynovial Giant Cell Tumors Approved Therapies
-
TURALIO (Pexidartinib): Daiichi Sankyo
TURALIO (Pexidartinib, formerly PLX3397) is an orally administered small molecule tyrosine kinase inhibitor that targets Colony-stimulating Factor 1 Receptor (CSF1R), KIT proto-oncogene receptor tyrosine kinase (KIT), and FMS-like Tyrosine Kinase 3 (FLT3) harboring an Internal Tandem Duplication (ITD) mutation. Overexpression of the CSF1R ligand promotes cell proliferation and accumulation in the synovium. In vitro, pexidartinib inhibited the proliferation of cell lines, which is dependent on CSF1R and ligand-induced auto-phosphorylation of CSF1R. Pexidartinib also inhibited the proliferation of a CSF1R-dependent cell line in vivo. It is a kinase inhibitor indicated for the treatment of adult patients with symptomatic TGCT associated with severe morbidity or functional limitations and not amenable to improvement with surgery. TURALIO was discovered by Plexxikon Inc., the small molecule structure-guided R&D center of Daiichi Sankyo.
TGCT Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region (First approval) |
|
TURALIO (pexidartinib) |
Daiichi Sankyo |
Tenosynovial Giant Cell Tumor |
Small molecule |
Fms-like tyrosine kinase 3 inhibitors; CSF1R Inhibitor; KIT proto-oncogene receptor tyrosine kinase Inhibitor |
Oral |
US: 2019 |
|
ROMVIMZA (vimseltinib) |
Ono Pharmaceutical Co. Ltd |
Symptomatic Tenosynovial Giant Cell Tumor |
Small molecule |
CSF1R inhibitor |
Oral |
US: 2025 EU: 2025 |
Tenosynovial Giant Cell Tumor Pipeline Analysis
-
Emactuzumab: SynOx Therapeutics (Celleron Therapeutics)
Emactuzumab is a potent, specific inhibitor of CSF1R, and data generated to date show its potential as a therapeutic platform targeting serious macrophage-driven inflammatory, fibrotic, and neovascular diseases. The CSF1 receptor, via its binding to two regulatory cytokines, CSF1 and IL-34, is critically involved in regulating macrophages and related cells in multiple biological processes across many organ systems, making it an attractive target with broad therapeutic applications. It was originally discovered and developed by Roche and has been tested in several phase 1a/b studies as monotherapy and in combination with other agents, including chemotherapeutics and immunotherapies in patients with a variety of solid tumors.
Currently, SynOx Therapeutics is conducting a Phase III TANGENT (NCT05417789) trial to study the drug for treating patients with localized or diffuse TGCT where surgical removal of the tumor is not viewed as an option. Emactuzumab has been investigated in the Phase III stage of clinical trial for the treatment of TCGT.
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Emactuzumab |
SynOx Therapeutics Limited |
III |
TGCT |
Intravenous |
CSF1R inhibitor |
2027 |
|
AMB 051 |
AmMax Bio |
II |
TGCT |
Intra-articular |
Macrophage CSF1R antagonists |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Tenosynovial Giant Cell Tumor Companies, Market Leaders and Emerging Companies
- Daiichi Sankyo
- Ono Pharmaceutical
- Abbisko Therapeutics
- SynOx Therapeutics Limited
- AmMax Bio, and others
Tenosynovial Giant Cell Tumor Drug Updates
- According to SynOx Therapeutics, the top-line results from study evaluating emactuzumab for Tenosynovial Giant Cell Tumours are expected in First Quarter of 2026.
- In November 2025, Abbisko Therapeutics Co. Ltd. announced the poster presentation of longer-term efficacy, safety and patient-reported outcomes from the global Phase III MANEUVER study of pimicotinib (ABSK021) in patients with tenosynovial giant cell tumour (TGCT) at the Connective Tissue Oncology Society (CTOS) 2025 Annual Meeting.
- In November 2025, Ono Pharmaceutical Co., Ltd. announced that data from multiple pipeline programs, including long-term and safety results from its MOTION Phase III study of vimseltinib in patients with TGCT will be presented during the CTOS Annual Meeting 2025, taking place November 12-15 in Boca Raton, Florida.
- In October 2025, Ono Pharmaceutical Co., Ltd. presented the two-year efficacy and safety results from its MOTION Phase III study of vimseltinib in patients with TGCT in cases where surgical removal of the tumor is not an option as a poster during the 2025 European Society for Medical Oncology Congress (ESMO), taking place October 17-21 in Berlin, Germany.
Tenosynovial Giant Cell Tumor Market Outlook
TGCT remains complex due to its heterogeneous nature, with localized disease typically benign and diffuse TGCT (PVNS) more aggressive and prone to recurrence. Surgery is the mainstay of treatment, including excision or synovectomy, but recurrence—especially in diffuse disease—remains a key challenge. Radiation therapy may be used as an adjunct, while symptomatic treatments such as NSAIDs do not address disease progression.
The introduction of targeted therapies against CSF1R has improved management in unresectable or recurrent TGCT. Off-label agents such as imatinib and sunitinib have shown benefit, while pexidartinib and vimseltinib are the only approved systemic therapies, establishing the standard of care in advanced settings. However, safety concerns and limited long-term data continue to impact treatment optimization.
The competitive landscape is evolving with emerging therapies such as pimicotinib, emactuzumab, and AMB-05X, which aim to improve efficacy and tolerability. While surgery remains dominant in early management, systemic therapies are increasingly used in diffuse or refractory cases. Overall, the TGCT market is becoming more competitive, though significant unmet needs remain.
Overall, the launch of first-in-class therapies, improved genetic testing, and rising disease awareness are expected to drive steady growth in the 7MM TGCT market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- Among the 7MM, the US accounted for the largest Tenosynovial Giant Cell Tumors Treatment Market Size. i.e., USD ~240 million in 2025.
- In 2036, among all the therapies for TGCT, the highest revenue is estimated to be generated by Vimseltinib followed by Emactuzumab, in the US.
- The entry of late-stage candidates such as Pimicotinib, and other is expected to intensify competition in the TGCT treatment landscape during the latter forecast period.
Tenosynovial Giant Cell Tumors Drug Class/Insights into Leading Emerging and Marketed Therapies (2022–2036 Forecast)
The TGCT market comprises small molecules and monoclonal antibodies, each targeting the CSF1/CSF1R pathway and related disease mechanisms. Small molecules: Key small-molecule therapies include approved agents such as pexidartinib (TURALIO) and vimseltinib (ROMVIMZA), along with emerging candidates like pimicotinib (ABSK021) by Abbisko Therapeutics. These therapies dominate the landscape due to oral administration, targeted CSF1R inhibition, and strong clinical efficacy, making them central to both current treatment and pipeline development.
Monoclonal antibodies: Notable antibody-based therapies in development include emactuzumab (SynOx Therapeutics) and AMB-05X (AmMax Bio), which target CSF1R through alternative mechanisms. These agents are designed to improve selectivity and safety, with some utilizing localized delivery approaches to enhance efficacy while minimizing systemic exposure.
Small molecules define the core of the TGCT treatment landscape, supported by approved therapies and a robust late-stage pipeline, while monoclonal antibodies represent an emerging class with the potential to expand treatment options. Overall, innovation in TGCT remains highly focused on CSF1R-targeted approaches, with future growth driven by improved safety, tolerability, and durable response outcomes
Tenosynovial Giant Cell Tumor Drug Uptake
This section focuses on the uptake rate of potential Tenosynovial Giant Cell Tumors drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the TGCT drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The uptake of therapies in TGCT is expected to vary based on clinical positioning, safety profile, and differentiation from currently approved CSF1R inhibitors. Approved agents such as pexidartinib (TURALIO) and vimseltinib (ROMVIMZA) will continue to shape the treatment paradigm, with vimseltinib expected to see stronger adoption due to its improved selectivity and tolerability profile. However, overall uptake of systemic therapies will remain moderate, as surgery continues to dominate first-line management, particularly in localized disease.
Among emerging therapies, pimicotinib (ABSK021) is expected to achieve relatively faster uptake, supported by strong Phase III data, regulatory momentum, and potential positioning as a best-in-class CSF1R inhibitor. In contrast, monoclonal antibodies such as emactuzumab and AMB-05X are likely to experience gradual uptake due to earlier-stage development, limited long-term data, and the need to demonstrate clear differentiation in efficacy and safety versus existing therapies.
Overall, adoption of new therapies in TGCT will be driven by their ability to address key unmet needs, including improved safety, durable responses, and use in patients with unresectable or recurrent disease. While the market is expected to expand with new entrants, uptake will remain selective, with physicians favoring therapies that demonstrate meaningful clinical benefit over existing CSF1R-targeted options.
Tenosynovial Giant Cell Tumor Market Access and Reimbursement of Approved Therapies
The Tenosynovial Giant Cell Tumors Treatment Market Report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc. Currently, there are two approved therapies, i.e., TURALIO (pexidartinib) by Daiichi sankyo that is approved in the US and ROMVIMZA (Vimseltinib) by Ono Pharmaceutical, approved in the US and EU.
The United States
US Reimbursement for Tenosynovial Giant Cell Tumour Therapies | |
|
Drug |
Access Program |
|
TURALIO |
|
|
ROMVIMZA |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Tenosynovial Giant Cell Tumor Therapies Price Scenario & Trends
Pricing and analogue assessment of TGCT therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of TGCT-Approved Drugs
In TGCT, annual treatment costs vary significantly, with conventional systemic therapies such as imatinib and sunitinib estimated at USD 13,925 per year, while Turalio (pexidartinib) reaches approximately USD 280,242 annually. Emerging therapies further increase the annual cost burden, with agents like DCC-3014 (vimseltinib) estimated at around USD 339,664 per year, reflecting the premium associated with novel targeted treatments.
Tenosynovial Giant Cell Tumor Industry Experts and Physician Views
To keep up with TGCT Treatment Market Trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the TGCT emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in TGCT, including MD, Ph.D, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 15+ KOLs to gather insights at country level. Centers such as the Oxford University Hospitals, Memorial Sloan Kettering Cancer Center, and National Cancer Center Hospital, etc. were contacted. Their opinion helps understand and validate current and emerging TGCT therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in TGCT.
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Tenosynovial Giant Cell Tumor is a rare, benign tumor that affects the synovium, bursae, and tendon sheaths, causing joint swelling, pain, and restricted movement. Although benign, TGCT can be locally aggressive and challenging to treat. The FDA had approved pexidartinib, a CSF-1 receptor inhibitor, for patients with symptomatic TGCT who are not candidates for surgery. Additionally, observational studies like the TGCT Observational Platform Project (TOPP) collect patient-reported outcomes to better understand the disease's impact on quality of life.” |
|
Germany |
“The European approval of TURALIO was rejected as the EMA believed that the benefits of treatment with TURALIO could not outweigh the risks associated with its use. There were serious concerns regarding its potentially-life threatening hepatotoxic effects while being used for the treatment of a non-fatal condition.” |
Tenosynovial Giant Cell Tumors Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and Tenosynovial Giant Cell Tumors Treatment Market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis. In the SWOT analysis of TGCT, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Tenosynovial Giant Cell Tumors Market Report
- The Tenosynovial Giant Cell Tumors Therapeutics Market Report covers a segment of key events, an executive summary, a descriptive overview, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the Tenosynovial Giant Cell Tumors epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Tenosynovial Giant Cell Tumor Treatment Market, historical and forecasted Tenosynovial Giant Cell Tumors Market Size, Tenosynovial Giant Cell Tumors Market Share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Tenosynovial Giant Cell Tumors Therapeutics Market Report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Tenosynovial Giant Cell Tumor Drugs Market.
Tenosynovial Giant Cell Tumors Market Report Insights
- TGCT Patient Population Forecast
- TGCT Therapeutics Market Size
- TGCT Pipeline Analysis
- TGCT Market Size and Trends
- TGCT Market Opportunity (Current and forecasted)
Tenosynovial Giant Cell Tumors Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year forecast
- TGCT Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- TGCT Treatment Addressable Market (TAM)
- TGCT Competitive Landscape
- TGCT Major Companies Insights
- TGCT Price Trends and Analogue Assessment
- TGCT Therapies Drug Adoption/Uptake
- TGCT Therapies Peak Patient Share Analysis
Tenosynovial Giant Cell Tumors Market Report Assessment
- TGCT Current Treatment Practices
- TGCT Unmet Needs
- TGCT Clinical Development Analysis
- TGCT Emerging Drugs Product Profiles
- TGCT Market Attractiveness
- TGCT Qualitative Analysis (SWOT and conjoint analysis)
Key Questions Answered in the Tenosynovial Giant Cell Tumors Market Report
Tenosynovial Giant Cell Tumors Treatment Market Insights
- What was the TGCT Market Size, the Tenosynovial Giant Cell Tumors Treatment Market Size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of TGCT?
- What impact will patent expiry have on the EGFR therapy market?
- What are the disease risks, burdens, and unmet needs of TGCT? What will be the growth opportunities across the 7MM concerning the patient population with TGCT?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of TGCT? What are the current guidelines for treating TGCT in the US, Europe, and Japan?
Reasons to Buy the Tenosynovial Giant Cell Tumors Market Report
- The Tenosynovial Giant Cell Tumors Therapeutics Market Report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the TGCT Market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease Tenosynovial Giant Cell Tumors Incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the Tenosynovial Giant Cell Tumors Drugs Market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Tenosynovial Giant Cell Tumors Drugs Market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.
Stay updated with us for Recent Articles
-market.png&w=256&q=75)
-pipeline.png&w=256&q=75)




