Uncomplicated Gonorrhea Market
Uncomplicated Gonorrhea Insights and Trends
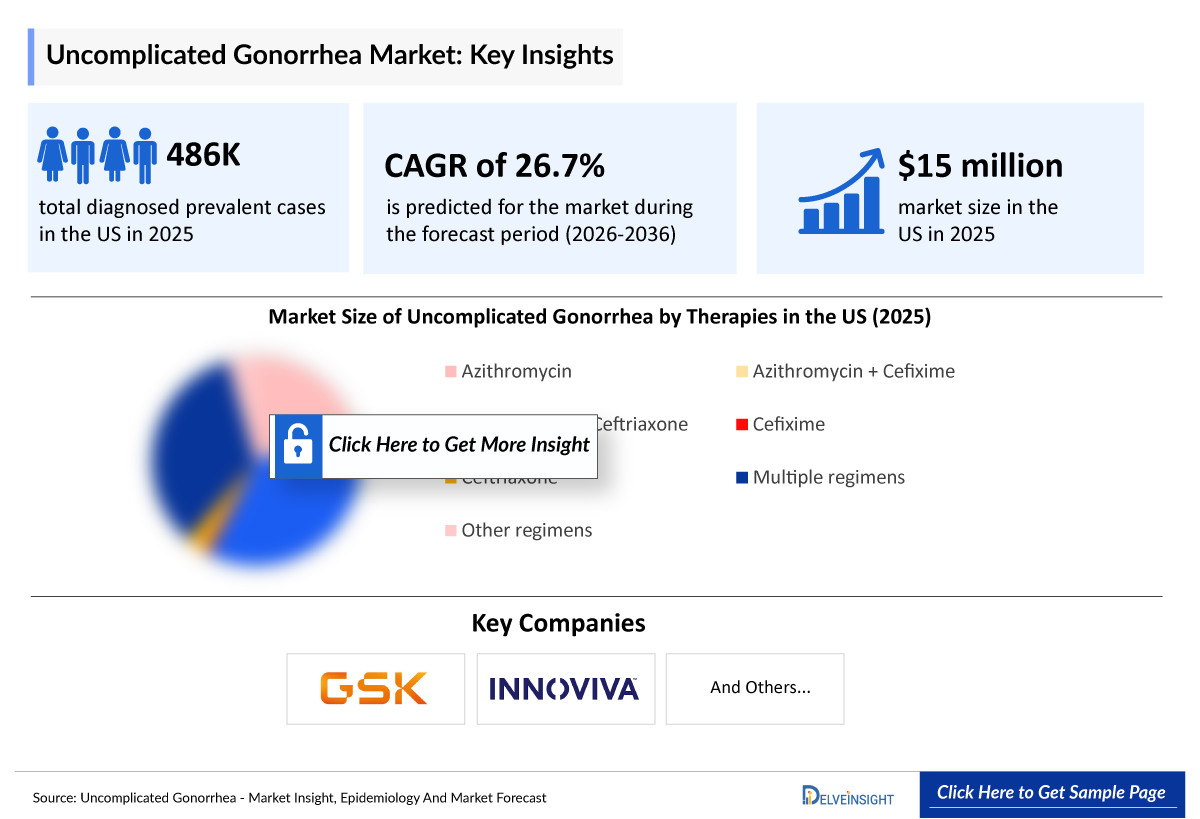
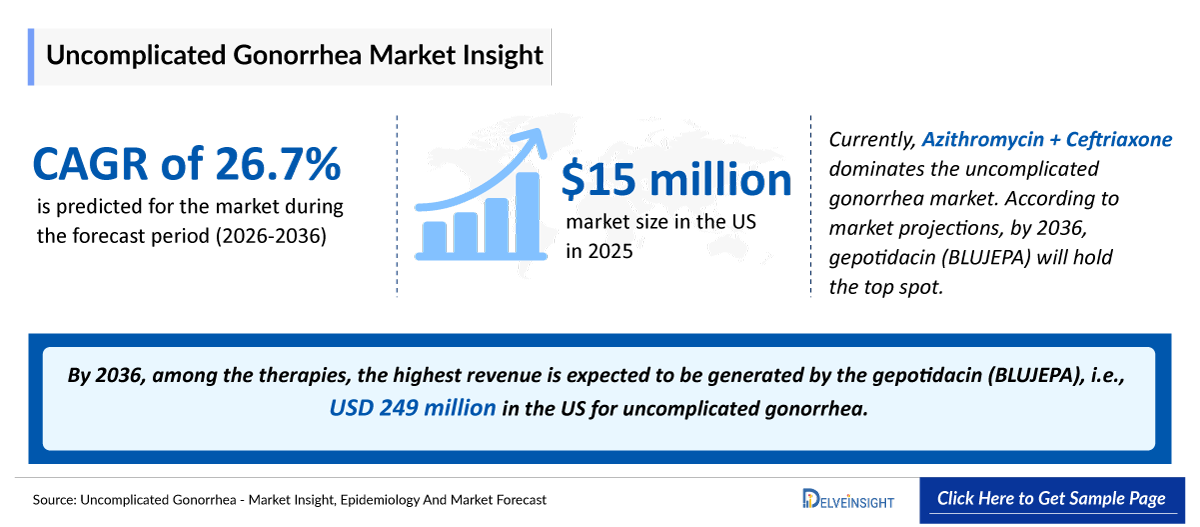
- According to DelveInsight’s analysis, the Uncomplicated Gonorrhea market in the United States was estimated at approximately USD 15 million in 2025.
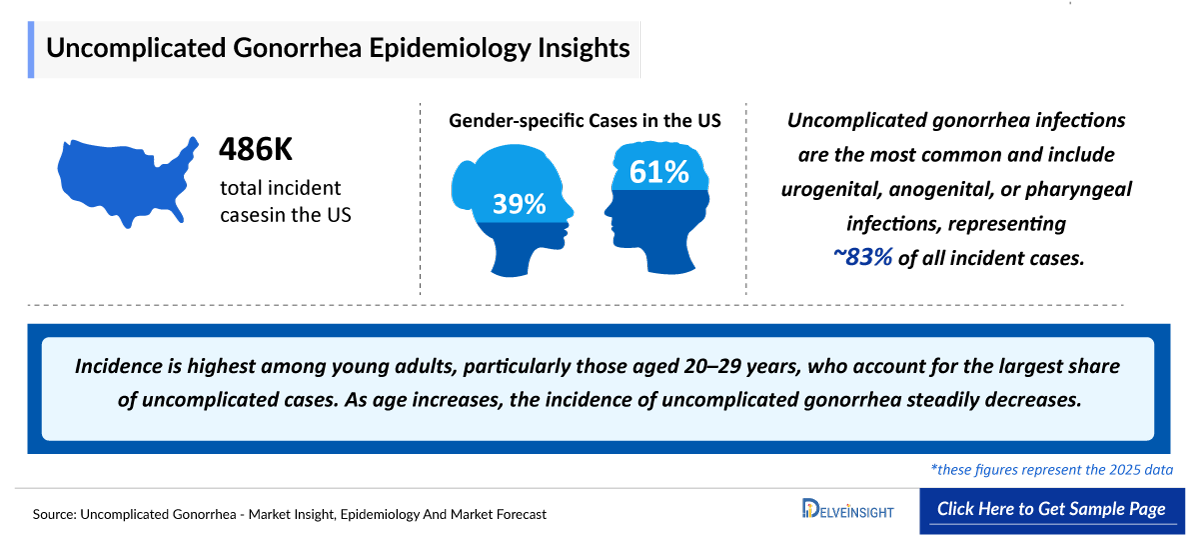
- Gonorrhea incidence has fluctuated significantly over the past decades, declining sharply with the introduction of penicillin in the 1950s, rising again in the 1960s–70s due to demographic and behavioral changes, and falling in the late 1980s–1990s alongside national screening efforts and the HIV/AIDS epidemic. After reaching a historic low in 2009, US cases have risen steadily since 2010, although WHO data indicate a decline globally since 2021. The highest burden remains among individuals aged 20–24 years.
- Gonorrhea treatment has evolved due to rising antimicrobial resistance. Older agents, including penicillin, tetracyclines, fluoroquinolones, macrolides, and ciprofloxacin, are no longer recommended, and ceftriaxone remains the last highly effective single-dose therapy. Reflecting azithromycin resistance, 2020 CDC guidelines now recommend 500 mg intramuscular ceftriaxone monotherapy for uncomplicated urogenital, rectal, and pharyngeal infections in adults and adolescents.
- The uncomplicated gonorrhea treatment market is undergoing a major shift following the Food and Drug Administration (FDA) approvals of two oral therapies in December 2025: NUZOLVENCE (Innoviva) and BLUJEPA (GSK). Previously, treatment relied largely on intramuscular ceftriaxone, often combined with azithromycin, a regimen increasingly challenged by antimicrobial resistance. The introduction of these first-in-class oral antibiotics addresses a key unmet need for effective non-injectable options and is expected to drive revenue growth for both manufacturers beginning in 2026.
- BLUJEPA was FDA-approved for uncomplicated urogenital gonorrhea, following its March 2025 clearance for uncomplicated UTIs (uUTI). It is the first new antibiotic class in over 30 years, triazaacenaphthylene topoisomerase inhibitors, approved for gonorrhea. While both BLUJEPA and NUZOLVENCE target DNA gyrase, they act differently, potentially lowering cross-resistance risk. BLUJEPA’s broader uUTI label contrasts with NUZOLVENCE’s gonorrhea-only indication, designed to preserve its effectiveness.
- Several investigational agents have failed in late-stage development, including solithromycin and delafloxacin, highlighting the difficulty of gonorrhea drug development. Currently, the late-stage pipeline remains scarce.
Uncomplicated Gonorrhea Market size and forecast in the United States
- 2025 Uncomplicated Gonorrhea Market Size: ~USD 15 million
- 2036 Projected Uncomplicated Gonorrhea Market Size: USD 466 million
- Uncomplicated Gonorrhea Growth Rate (2026–2036): 26.7% CAGR
DelveInsight's ‘Uncomplicated Gonorrhea – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Uncomplicated Gonorrhea, historical and forecasted epidemiology, as well as the Uncomplicated Gonorrhea market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Uncomplicated Gonorrhea market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Uncomplicated Gonorrhea patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Uncomplicated Gonorrhea and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Uncomplicated Gonorrhea Market CAGR (Study period/Forecast period) |
26.7% (2022-2036) |
|
Uncomplicated Gonorrhea Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Uncomplicated Gonorrhea Companies |
|
|
Uncomplicated Gonorrhea Therapies |
|
|
Uncomplicated Gonorrhea Market |
Segmented by
|
|
Analysis |
|
Key Factors Driving the Uncomplicated Gonorrhea Market
Rising Disease Burden, Antimicrobial Resistance & Advancing Treatment Landscape
The uncomplicated gonorrhea market is primarily driven by the rising global prevalence of infections and the large proportion of cases that are diagnosed at an uncomplicated stage, ensuring consistent demand for treatment. Growth is further supported by increasing antimicrobial resistance, which is creating a need for new and more effective therapies, alongside continued reliance on guideline-recommended first-line treatments. Additionally, advancements in drug development, expansion of screening and diagnostic programs, supportive public health initiatives, and the availability of convenient outpatient treatment options collectively contribute to sustained market growth.
Uncomplicated Gonorrhea Understanding and Treatment Algorithm
Uncomplicated Gonorrhea Overview and Diagnosis
Uncomplicated Gonorrhea is a common sexually transmitted infection (STI) caused by the bacterium Neisseria gonorrhoeae, characterized by localized infection of mucosal surfaces such as the urethra, cervix, rectum, oropharynx, and conjunctiva. The disease typically presents with symptoms including urethral or vaginal discharge, dysuria, and genital irritation, although many infections particularly in women may be asymptomatic. Uncomplicated gonorrhea refers to infection confined to these mucosal sites without systemic dissemination or complications such as pelvic inflammatory disease or disseminated gonococcal infection. If untreated, the infection can lead to reproductive tract complications, infertility, and increased risk of HIV transmission, while growing antimicrobial resistance has become a major challenge in disease management worldwide.
Uncomplicated Gonorrhea Diagnosis
The approach to diagnostic testing for N. gonorrhoeae has evolved from traditional cultivation to the widespread use of Nucleic Acid Amplification Tests (NAATs). Gram stain, another non-culture test, can be used if available for the diagnosis of urethral gonorrhea in symptomatic males. Culture is still recommended if antimicrobial resistance is a concern, especially in cases of suspected treatment failure. Routine screening for gonococcal infection in persons at increased risk is recommended in order to decrease morbidity and reduce the burden of disease in the community. Testing samples should be obtained from the anatomic sites of sexual exposure.
Further details are provided in the report.
Uncomplicated Gonorrhea Treatment
Treatment of Uncomplicated Gonorrhea primarily relies on dual antibiotic therapy to ensure high cure rates and limit the emergence of resistance in Neisseria gonorrhoeae. Current clinical practice commonly recommends a third-generation cephalosporin, most often Ceftriaxone administered intramuscularly, combined with a second agent such as Azithromycin or Doxycycline, which also addresses possible coinfection with Chlamydia trachomatis. Dual therapy is preferred because N. gonorrhoeae has developed resistance to multiple historical treatments including sulfonamides, penicillins, tetracyclines, and fluoroquinolones while reduced susceptibility to cephalosporins has also been reported. Consequently, treatment strategies emphasize combination regimens, ongoing antimicrobial-resistance surveillance, and follow-up testing to detect coinfections such as Syphilis and ensure effective disease control.
Further details related to country-based variations are provided in the report.
Uncomplicated Gonorrhea Unmet Needs
The section “unmet needs of Uncomplicated Gonorrhea” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of active Emerging Therapies
- Challenges in Gonococcal Vaccine Development
- Diagnostic and Screening Gaps in Uncomplicated Gonorrhea
- Urgent Need for New Antibiotics amid Increasing Resistance, and others..
Comprehensive unmet needs insights in Uncomplicated Gonorrhea and their strategic implications are provided in the full report.
Uncomplicated Gonorrhea Epidemiology
Key Findings from Uncomplicated Gonorrhea Epidemiological Analysis and Forecast
- According to DelveInsight’s estimates, the total incident cases of Gonorrhea in the US were approximately 401,240 in 2025.
- In 2025, uncomplicated gonorrhea represents the majority of cases in the US, compared to complicated gonorrhea.
- The total number of Male Uncomplicated Gonorrhea patients was found to be more ~242,840 as compared to Female patients ~160,400 in 2025 in the US.
Uncomplicated Gonorrhea Drug Chapters & Competitive Analysis
The Uncomplicated Gonorrhea drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Uncomplicated Gonorrhea treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Uncomplicated Gonorrhea therapeutics market.
Approved Therapies for Uncomplicated Gonorrhea
Gepotidacin (BLUJEPA): GlaxoSmithKline
BLUJEPA is a bactericidal, first-in-class triazaacenaphthylene antibiotic that inhibits bacterial DNA replication by a distinct binding site, a novel mechanism of action, and, for most pathogens, provides well-balanced inhibition of two different Type II topoisomerase enzymes. This provides activity against N. gonorrhoeae and most target uropathogens (such as E. coli and Staphylococcus saprophyticus), including isolates resistant to current antibiotics.
In December 2025, GSK announced that the US FDA had approved a supplemental New Drug Application (sNDA) for gepotidacin as an oral option for adult and paediatric patients from 12 years of age weighing at least 45 kg who have limited or no alternative options for the treatment of uncomplicated urogenital gonorrhoea caused by susceptible strains of N. gonorrhoeae (e.g., where standard of care is contraindicated, or where patients are intolerant or unwilling to use first line treatment.
|
Uncomplicated Gonorrhea Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Gepotidacin (BLUJEPA) |
GlaxoSmithKline |
Uncomplicated gonorrhea |
Small molecule |
Oral |
Triazaacenaphthylene bacterial type II topoisomerase inhibitor |
US: 2025 |
|
Zoliflodacin (NUZOLVENCE) |
Innoviva Specialty Therapeutics |
Uncomplicated gonorrhea |
Small molecule |
Oral |
Spiropyrimidinetrione bacterial type II topoisomerase inhibitor |
US: 2025 |
Uncomplicated Gonorrhea Key Players, Market Leaders and Emerging Companies
- GlaxoSmithKline
- Innoviva Specialty Therapeutics, and others
Uncomplicated Gonorrhea Drug Updates
- In August 2025, According to GSK’s Q3 2025 presentation, BLUJEPA has been granted FTD and QIDP designation from the US FDA for the treatment of uncomplicated urogenital gonorrhea. It announced that the US FDA had accepted for priority review an sNDA for BLUJEPA as an oral option for the treatment of uncomplicated urogenital gonorrhea in patients 12 years of age and older (weighing =45 kg).
- In December 2025, Innoviva Specialty Therapeutics announced that the US FDA has approved NUZOLVENCE for the treatment of uncomplicated urogenital gonorrhea in adults and pediatric patients 12 years and older weighing at least 35 kg. The Company plans to commercialize NUZOLVENCE in the second half of 2026, either in collaboration with a commercialization partner or independently.
Drug Class Insights
Uncomplicated Gonorrhea Market Outlook
The Uncomplicated Gonorrhea market is undergoing a notable transition after decades of reliance on conventional antibiotics, as the remarkable ability of Neisseria gonorrhoeae to develop antimicrobial resistance has progressively eroded the effectiveness of historical therapies such as Penicillin, Tetracycline, and Ciprofloxacin. Currently, Ceftriaxone remains the cornerstone of treatment, although increasing resistance concerns and limited therapeutic alternatives have intensified the need for novel agents. The recent approvals of first-in-class antibiotics such as Gepotidacin (BLUJEPA) and Zoliflodacin (NUZOLVENCE) mark a significant shift toward mechanism-driven antimicrobial innovation, providing new oral treatment options and addressing critical gaps in the management of drug-resistant infections.
With the emergence of these novel therapies, the treatment landscape for uncomplicated gonorrhea is expected to evolve similarly to other infectious diseases facing antimicrobial resistance, where innovative agents stimulate renewed clinical and commercial activity. The United States is anticipated to remain the largest market, supported by higher diagnosis rates, strong surveillance programs such as those led by the Centers for Disease Control and Prevention, and faster adoption of new therapies compared with markets in Europe and Japan.
Overall, the introduction of first-in-class antibiotics, ongoing antimicrobial-resistance surveillance, and increasing global awareness of sexually transmitted infections are expected to drive steady market evolution for uncomplicated gonorrhea over the coming decade, with meaningful opportunities for both newly launched therapies and emerging pipeline candidates.
- According to the estimates, the largest market size of Uncomplicated Gonorrhea was captured by the United States, i.e., ~USD 466 million by 2036.
- By 2036, among the therapies, the highest revenue is expected to be generated by the gepotidacin (BLUJEPA), i.e., USD 249 million in the US for uncomplicated gonorrhea.
- Azithromycin is no longer recommended as monotherapy because of rising resistance; however, it may still be used in combination with ceftriaxone as part of dual therapy, in line with CDC recommendations.
- The intramuscular injection is considered cumbersome and painful, requiring provider administration and limiting treatment to clinical settings, which constrains telehealth and expedited partner therapy. A non-inferior oral alternative could greatly improve convenience, adherence, and patient uptake.
- A single injection (standard of care) ensures full compliance in-clinic but requires a visit. Oral therapies like BLUJEPA and NUZOLVENCE offer greater convenience and are expected to improve adherence among patients less likely to return for an injection, even with a second at-home dose.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Uncomplicated Gonorrhea (2022–2036 Forecast)
The Uncomplicated Gonorrhea treatment landscape comprises cephalosporin antibiotics, novel bacterial topoisomerase inhibitors, and next-generation antibacterial agents, alongside alternative combination regimens, each targeting key bacterial replication or survival pathways in Neisseria gonorrhoeae.
Third-generation cephalosporins: Ceftriaxone remains the cornerstone first-line therapy for uncomplicated gonorrhea, acting by inhibiting bacterial cell-wall synthesis through binding to penicillin-binding proteins. Administered as a single intramuscular dose, it achieves high microbiological cure rates and currently defines the global standard of care, although rising antimicrobial resistance has increased the urgency for additional therapeutic options.
Novel bacterial topoisomerase inhibitors:
Gepotidacin (BLUJEPA) represents a first-in-class triazaacenaphthylene antibiotic that inhibits bacterial DNA replication through balanced inhibition of DNA gyrase and topoisomerase IV. Approved in 2025, it offers a convenient oral treatment option and validates the development of next-generation antibacterials designed to overcome resistance to existing antibiotic classes.
Zoliflodacin (NUZOLVENCE) is another first-in-class oral antibiotic that selectively inhibits bacterial DNA gyrase via a mechanism distinct from fluoroquinolones. Its clinical development demonstrated strong activity against drug-resistant N. gonorrhoeae, highlighting its role as a promising alternative therapy in the evolving gonorrhea treatment paradigm. Together, cephalosporins and emerging next-generation topoisomerase inhibitors define the core innovation landscape in uncomplicated gonorrhea, with ceftriaxone maintaining current clinical dominance while novel oral agents such as gepotidacin and zoliflodacin are expected to drive the next phase of therapeutic expansion between 2022 and 2036.
Further details will be provided in the report….
Uncomplicated Gonorrhea Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Uncomplicated Gonorrhea market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in Uncomplicated Gonorrhea is expected to vary across established cephalosporin antibiotics and newly introduced next-generation antibacterial agents. The long-standing standard of care Ceftriaxone is expected to maintain dominant uptake due to its proven efficacy, guideline support, and widespread clinical familiarity. Recently approved novel oral antibiotics such as Gepotidacin (BLUJEPA) and Zoliflodacin (NUZOLVENCE) are anticipated to demonstrate gradual but increasing uptake, supported by their novel mechanisms of action, activity against resistant Neisseria gonorrhoeae strains, and the convenience of oral administration. Their adoption is likely to be driven by growing antimicrobial resistance, the need for alternative options in patients unable to receive injectable therapy, and increasing clinical experience as these agents are integrated into future treatment guidelines.
Further detailed analysis of emerging therapies' drug uptake in the report…
Market Access and Reimbursement of Uncomplicated Gonorrhea
The United States
Gepotidacin (BLUJEPA)
BLUJEPA Patient Savings
Patients who qualify may pay as little as USD 30 for BLUJEPA. This offer is valid for one use, and each covered prescription counts as one use.
Eligibility
- Patients may be eligible for this offer if they have commercial insurance and insurance does not cover the full cost of the prescription.
- Patients are not eligible for this program if they are enrolled in Medicare Part B, Medicare Part D, Medicaid, Medigap, Veterans Affairs (VA), Department of Defense (DoD) programs, or TRICARE.
- Patients are also ineligible for this program if they are Medicare eligible and enrolled in an employer-sponsored group waiver health plan or government-subsidized prescription drug benefit program for retirees.
- Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report….
Uncomplicated Gonorrhea Therapies Price Scenario & Trends
Pricing and analogue assessment of Uncomplicated Gonorrhea therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Uncomplicated Gonorrhea Approved Drugs
GSK’s BLUJEPA received FDA approval in the US in 2025 for two indications: first, as a first-in-class, twice-daily oral therapy for uncomplicated urinary tract infections on March 25, 2025, and later for uncomplicated urogenital gonorrhea in December 2025. The estimated therapy cost is USD 1,864.
Further details are provided in the final report….
Industry Experts and Physician Views for Uncomplicated Gonorrhea
To keep up with Uncomplicated Gonorrhea market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the Uncomplicated Gonorrhea emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Uncomplicated Gonorrhea, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the University of North Carolina at Chapel Hill, Berlin Institute of Health at Charité, and the University of Nottingham, etc. were contacted. Their opinion helps understand and validate current and emerging Uncomplicated Gonorrhea therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Uncomplicated Gonorrhea.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“The approval of new treatments for gonorrhoea is an important and timely development in the context of rising global incidence, increasing antimicrobial resistance and the very limited therapeutic options currently available.” |
|
Europe |
“The emergence of strains with decreased susceptibility to extended-spectrum cephalosporins represents a major threat to effective gonorrhoea control.” |
Qualitative Analysis: SWOT and Conjoint Analysis
- We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
- In the SWOT analysis of Uncomplicated Gonorrhea, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
- Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
- The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Uncomplicated Gonorrhea, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Uncomplicated Gonorrhea market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Uncomplicated Gonorrhea market.
Report Insights
- Uncomplicated Gonorrhea Patient Population Forecast
- Uncomplicated Gonorrhea Therapeutics Market Size
- Uncomplicated Gonorrhea Pipeline Analysis
- Uncomplicated Gonorrhea Market Size and Trends
- Uncomplicated Gonorrhea Market Opportunity (Current and Forecasted)
Report Key Strengths
- Epidemiology-based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year Forecast
- Uncomplicated Gonorrhea Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Uncomplicated Gonorrhea Treatment Addressable Market (TAM)
- Uncomplicated Gonorrhea Competitive Landscape
- Uncomplicated Gonorrhea Major Companies Insights
- Uncomplicated Gonorrhea Price Trends and Analogue Assessment
- Uncomplicated Gonorrhea Therapies Drug Adoption/Uptake
- Uncomplicated Gonorrhea Therapies Peak Patient Share Analysis
Report Assessment
- Uncomplicated Gonorrhea Current Treatment Practices
- Uncomplicated Gonorrhea Unmet Needs
- Uncomplicated Gonorrhea Clinical Development Analysis
- Uncomplicated Gonorrhea Emerging Drugs Product Profiles
- Uncomplicated Gonorrhea Market Attractiveness
- Uncomplicated Gonorrhea Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Market Insights
- What was the Uncomplicated Gonorrhea market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Uncomplicated Gonorrhea?
- What are the disease risks, burdens, and unmet needs of Uncomplicated Gonorrhea? What will be the growth opportunities across the 7MM concerning the patient population with Uncomplicated Gonorrhea?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Uncomplicated Gonorrhea? What are the current guidelines for treating Uncomplicated Gonorrhea in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Uncomplicated Gonorrhea market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.