Alzheimer’s Disease Market Set to Surge from USD 5,038 Million in 2025 at 23.7% CAGR by 2034
Get a Sneak Peek at the Latest alzheimers disease market size and insights Report
The Alzheimer’s Disease market size was estimated at approximately USD 5,038 million in 2025 and is projected to expand at a robust CAGR of 23.7% over the forecast period from 2020 to 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Alzheimer’s Disease landscape.
In 2023, the United States represented the largest share of the Alzheimer’s Disease market, accounting for approximately 54% of the total 7MM market, compared to other major regions including the EU4 (Germany, France, Italy, and Spain), the UK, and Japan. Within the EU, Germany had the largest Alzheimer’s disease market at USD 267 million, while the UK reported the smallest at around USD 104 million. In Japan, the market was estimated at approximately USD 845 million in 2023.
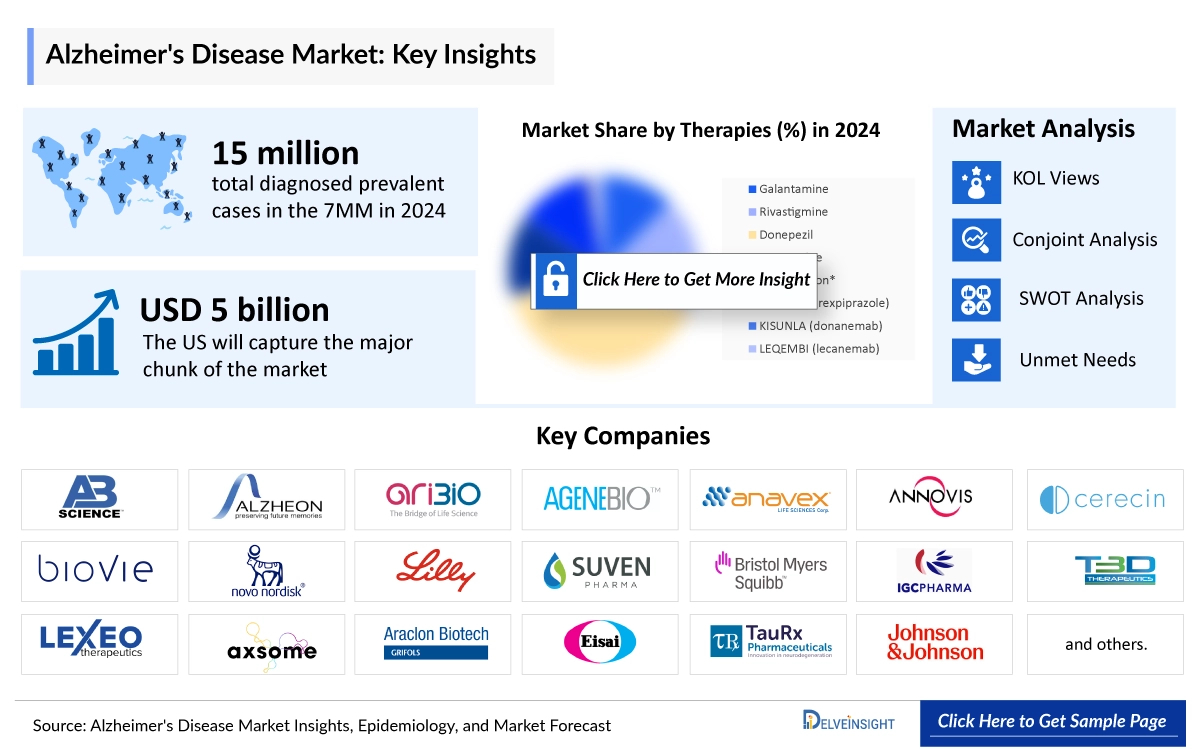
According to DelveInsight analysts, the 7MM had approximately 16 million diagnosed prevalent cases of Alzheimer’s Disease in 2024. The United States accounted for about 44% of these cases, totaling roughly 7 million. DelveInsight projects a further increase by 2034, underscoring the growing healthcare burden of Alzheimer’s disease in the US over the forecast period.
In the US, Alzheimer’s Disease cases are classified into four age groups: under 65, 65–74, 75–84, and 85 years and older. In 2024, the under-65 group accounted for approximately 180,000 cases, the 65–74 group for about 1.6 million cases, the 75–84 group for nearly 3 million cases, and the 85+ group for around 2.5 million cases.
Alzheimer’s Disease is also classified by severity into mild cognitive impairment (MCI), mild, moderate, and severe dementia. In 2024, the MCI stage was the most common in the EU4 and the UK, with around 2.6 million cases, while severe dementia had the lowest prevalence at approximately 650,000 cases. In Germany, diagnosed cases by genotype included roughly 180,000 homozygous, 790,000 heterozygous, and 560,000 non-carrier cases. In France, an estimated 5 million individuals were in the preclinical stage, showing early brain changes without noticeable symptoms.
In 2024, Spain had approximately 460,000 diagnosed cases of agitation related to Alzheimer’s Disease, with numbers projected to rise by 2034. This increasing prevalence emphasizes the need for targeted interventions to manage agitation and address the psychological and behavioral challenges of the disease. In Japan, there were around 1.4 million diagnosed cases in men and 2.5 million in women in 2024, highlighting a significant gender disparity and the importance of gender-specific treatment and care strategies.
DelveInsight’s report, “Alzheimer’s Disease Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Alzheimer’s Disease landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Alzheimer’s Disease market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Alzheimer’s Disease market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Alzheimer’s Disease Market Insights
Some of the key facts of the Alzheimer’s Disease Market Report:
- With the anticipated introduction of new Alzheimer’s Disease therapies including Masitinib, Valiltramiprosate/ALZ-801, Tricaprilin (CER0001), and Bezisterim (NE3107) the overall Alzheimer’s disease market is expected to experience notable growth in the coming years.
- Key Alzheimer’s Disease Companies: Biogen Inc./Eisai Co., Ltd., Eli Lilly and Company, AB Science, Alzheon, Inc., Cerecin, BioVie, Eisai Inc., Hoffmann-La Roche, AZ Therapies, Neurotrope bioscience, AZ Therapies, Cerecin, and others
- Key Alzheimer’s Disease Therapies: LEQEMBI (lecanemab), KISUNLA (Donanemab), Masitinib (AB1010), Valiltramiprosate (ALZ-801), Tricaprilin (CER-0001), Bezisterim (NE3107), BAN2401, Gantenerumab, ALZT-OP1, Bryostatin-1, ALZT-OP1, Tricaprilin
- The Alzheimer’s Disease epidemiology based on gender analyzed that more women than men have Alzheimer’s or other dementias
Alzheimer’s Disease Overview
Alzheimer’s Disease is a progressive neurodegenerative disorder and the most common cause of dementia, affecting memory, thinking, and behavior. It primarily occurs in older adults and gradually worsens over time, leading to loss of independence.
The disease is characterized by the accumulation of amyloid plaques and tau tangles in the brain, which damage nerve cells and disrupt communication between them. Early symptoms include mild memory loss and confusion, while advanced stages can result in severe cognitive decline, difficulty in communication, and impaired daily functioning.
Key Trends in Alzheimer’s Disease Therapeutics Market:
- Shift Toward Disease-Modifying Therapies (DMTs): Increasing focus on anti-amyloid and anti-tau therapies aimed at slowing disease progression rather than just managing symptoms.
- Emergence of Precision Medicine: Use of biomarkers, genetic profiling, and advanced imaging is enabling personalized treatment approaches.
- Strong R&D Pipeline: Ongoing late-stage clinical trials are driving innovation and expanding future treatment options.
- Growth in Combination Therapies: Development of multi-targeted approaches combining symptomatic treatments with DMTs for improved outcomes.
- Regulatory Support and Accelerated Approvals: Agencies are providing faster review pathways for breakthrough therapies, encouraging investment and innovation in Alzheimer’s disease therapeutics.
Get a Free sample for the Alzheimer’s Disease Market Report:
https://www.delveinsight.com/report-store/alzheimers-disease-ad-market
Alzheimer’s Disease Market
The dynamics of the Alzheimer’s Disease market are anticipated to change in the coming years owing to the expected launch of emerging therapies and others during the forecasted period 2020-2034.
“Of the emerging therapies, the most anticipated product to get launched is BAN2401. Apart from this, several other molecules are in early clinical stage or completed clinical studies such as ALZT-OP1, Gantenerumab, Brexpiprazole, and others.”
Alzheimer’s Disease Epidemiology
The epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Alzheimer’s Disease Epidemiology Segmentation:
The Alzheimer’s Disease market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalence of Alzheimer’s Disease
- Prevalent Cases of Alzheimer’s Disease by severity
- Gender-specific Prevalence of Alzheimer’s Disease
- Diagnosed Cases of Episodic and Chronic Alzheimer’s Disease
Download the report to understand which factors are driving Alzheimer’s Disease epidemiology trends @ Alzheimer’s Disease Epidemiological Insights
Recent Development In The Alzheimer’s Disease Treatment Landscape:
- In February 2026, Eisai Limited and Biogen Canada Inc. expressed disappointment and disagreement with the decision by the Canada's Drug Agency (CDA-AMC) not to recommend LEQEMBI® for public reimbursement in Canada. The companies emphasized that this draft decision comes despite clinical evidence demonstrating that LEQEMBI® can help slow disease progression and functional decline in patients with early Alzheimer’s Disease.
- In December 2025, Spinogenix’s lead asset, tazbentetol, demonstrated early, sustained cognitive improvements in individuals with mild-to-moderate Alzheimer’s disease in a mid-stage clinical study. In the Phase IIa trial (NCT06427668), the once-daily oral small molecule produced early cognitive gains, with patients on the 300 mg high dose showing an average improvement of more than 2.5 points in SMMSE scores compared with placebo within just four weeks. These benefits persisted in participants who continued into the 24-week open-label extension. Comparable positive effects were also observed across other primary endpoints, including CDR-SB and activities of daily living (ADL) assessments.
- In November 2025, Biogen Inc. (Nasdaq: BIIB) announced that it will present new scientific data at the 18th Clinical Trials on Alzheimer’s Disease (CTAD) Conference, scheduled for December 1–4 in San Diego. Presentations on LEQEMBI® (lecanemab-irmb) will cover results on subcutaneous initiation dosing, advantages of ongoing treatment, and projected long-term time savings over a decade based on Phase 3 data, along with real-world findings from a post-marketing study in Japan and the ALZ-NET registry. The company will also unveil updated information on BIIB080, an investigational antisense oligonucleotide, drawn from a healthy-volunteer biodistribution study, plus research enhancing understanding of disease progression.
- In September 2025, Eisai’s investigational anti-MTBR tau antibody, etalanetug (E2814), has been granted FDA Fast Track designation for treating Alzheimer’s disease.
- In September 2025, Actinogen Medical Limited (ASX: ACW) announced that it successfully completed its scheduled Type C meeting (via written response) with the U.S. FDA concerning Alzheimer’s disease (AD).
- In September 2025, Alzinova AB announced that the U.S. FDA has approved its Investigational New Drug (IND) application for a planned Phase II trial of ALZ-101, a vaccine candidate for Alzheimer’s disease.
- In August. 2025 Eisai Co., Ltd. (Headquarters: Tokyo, CEO: Haruo Naito, "Eisai") and Biogen Inc. (Nasdaq: BIIB, Headquarters: Cambridge, Massachusetts, CEO: Christopher A. Viehbacher, "Biogen") announced that the U.S. Food and Drug Administration (FDA) has approved the Biologics License Application (BLA) for once weekly lecanemab-irmb subcutaneous injection (U.S. brand name: LEQEMBI® IQLIK™, pronounced "I Click") for maintenance dosing. LEQEMBI IQLIK is a subcutaneous autoinjector (SC-AI) developed by Eisai, containing 360 mg/1.8 mL (200 mg/mL) that can be administered in approximately 15 seconds. LEQEMBI IQLIK autoinjector is indicated for maintenance dosing to treat Alzheimer's disease (AD) in patients with mild cognitive impairment (MCI) or mild dementia stage of disease (collectively referred to as early AD) in the U.S. After 18 months of LEQEMBI (lecanemab-irmb) intravenous (IV) treatment at 10 mg/kg every two weeks, patients may either continue IV infusions at 10 mg/kg once every four weeks or start the new weekly 360 mg subcutaneous injection using the LEQEMBI IQLIK autoinjector.
- In July 2025, Cognition Therapeutics’ investigational dementia treatment demonstrated up to a 129% reduction in the rate of cognitive decline among patients with mild Alzheimer’s disease in a Phase II trial. The Phase II SHINE study (NCT03507790), evaluating zervimesine in Alzheimer’s patients, revealed that those with less advanced brain pathology measured by lower levels of the blood biomarker p-tau217 experienced significant slowing of cognitive deterioration. Within this subgroup, approximately half of the participants had mild Alzheimer’s disease, while the other half were classified as having moderate Alzheimer’s disease.
- In July 2025, Roche’s Alzheimer’s disease therapy demonstrated sustained benefits in a long-term extension study, showing positive outcomes after 28 weeks of treatment. In the high-dose group of the ongoing Phase Ib/IIa Brainshuttle AD trial (NCT04639050), trontinemab successfully lowered amyloid levels below the 24-centiloid positivity threshold in 91% of patients, with 72% reaching deep clearance below 11 centiloids. Additionally, the study reported early and notable reductions in Alzheimer’s disease biomarkers, including total tau, phosphorylated tau (pTau)181, pTau217, and neurogranin, as measured in both CSF and plasma.
- In July 2025, INmune Bio’s stock dropped over 50% after its TNF inhibitor failed to show efficacy in Alzheimer’s patients in a Phase II trial. The MINDFuL study (NCT05318976) tested XPro (pegipanermin), a selective soluble TNF inhibitor, in early Alzheimer’s patients with inflammatory biomarkers. While no overall benefit was observed at the six-month mark in the intent-to-treat group, patients with two or more inflammation biomarkers showed a modest 0.27-point improvement on the Early Mild Alzheimer’s Cognitive Composite (EMACC) and a -0.20 change in pTau217 blood levels. Inmune Bio suggests this subgroup might benefit from XPro.
- In June 2025, The UAB Brain Aging and Memory Clinic, which opened in spring 2024, ushers in a new chapter of care for Alzheimer’s disease and memory disorders in Alabama and the surrounding region. This facility has played a key role in supporting clinical trials, strengthening UAB’s position as a leading site for testing the latest generation of anti-Alzheimer therapies.
- In May 2025, Nuravax Inc. received a USD 3 million grant from the National Institutes of Health (NIH) to advance Duvax, the first dual-target Alzheimer’s vaccine, into human clinical trials.
- In April 2025, Biogen Inc. (Nasdaq: BIIB) announced that the U.S. Food and Drug Administration (FDA) has awarded Fast Track designation to BIIB080, an experimental antisense oligonucleotide (ASO) therapy targeting tau, for treating Alzheimer’s disease. This designation aims to accelerate the development and review process of investigational drugs addressing serious diseases with unmet medical needs.
- In March 2025, Acumen Pharmaceuticals completed enrollment for its multi-center, double-blind Phase II ALTITUDE-AD trial evaluating the humanized monoclonal antibody sabirnetug (ACU193) in patients with early Alzheimer’s disease. Sabirnetug is noted as the first antibody to selectively target amyloid beta oligomers (AβOs), a toxic form of amyloid beta implicated in the early stages of Alzheimer’s. The ALTITUDE-AD trial, initiated last year, is a randomized, placebo-controlled study that has enrolled 542 participants across Canada, the EU, the US, and the UK.
Alzheimer’s Disease Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Alzheimer’s Disease market or expected to get launched during the study period. The analysis covers Alzheimer’s Disease market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Alzheimer’s Disease Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyses recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Alzheimer’s Disease Therapies and Key Companies
- LEQEMBI (lecanemab): Biogen Inc./Eisai Co., Ltd.
- KISUNLA (Donanemab): Eli Lilly and Company
- Masitinib (AB1010): AB Science
- Valiltramiprosate (ALZ-801): Alzheon, Inc.
- Tricaprilin (CER-0001): Cerecin
- Bezisterim (NE3107): BioVie
- BAN2401: Eisai Inc.
- Gantenerumab: Hoffmann-La Roche
- ALZT-OP1: AZ Therapies
- Bryostatin-1: Neurotrope bioscience
- ALZT-OP1: AZ Therapies
- Tricaprilin: Cerecin
To know more about Alzheimer’s Disease treatment, visit @ Alzheimer’s Disease Medications
Alzheimer’s Disease Market Drivers
- Growing Aging Population: The rising prevalence of Alzheimer’s Disease is closely linked to the expanding elderly population worldwide.
- Increasing Disease Awareness & Early Diagnosis: Improved awareness campaigns and advancements in biomarkers and imaging are enabling earlier detection.
- Advancements in Disease-Modifying Therapies: Emergence of anti-amyloid and anti-tau therapies is transforming the treatment landscape.
- Rising Healthcare Expenditure: Increased investments in neurological research and dementia care are supporting market growth.
- Strong Pipeline & Clinical Trials: Ongoing R&D and late-stage pipeline drugs are expanding future treatment possibilities.
Alzheimer’s Disease Market Barriers
- High Treatment Costs: Novel therapies and long-term care management are expensive, limiting accessibility.
- Limited Curative Options: Most available treatments only manage symptoms rather than cure the disease.
- Complex Disease Pathology: Multifactorial nature of Alzheimer’s makes drug development challenging with high failure rates.
- Side Effects & Safety Concerns: Some advanced therapies may pose risks such as brain swelling or bleeding.
- Diagnosis Challenges: Early-stage diagnosis remains difficult due to subtle symptoms and limited access to advanced diagnostic tools.
Scope of the Alzheimer’s Disease Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Alzheimer’s Disease Companies: Biogen Inc./Eisai Co., Ltd., Eli Lilly and Company, AB Science, Alzheon, Inc., Cerecin, BioVie, Eisai Inc., Hoffmann-La Roche, AZ Therapies, Neurotrope bioscience, AZ Therapies, Cerecin, and others
- Key Alzheimer’s Disease Therapies: LEQEMBI (lecanemab), KISUNLA (Donanemab), Masitinib (AB1010), Valiltramiprosate (ALZ-801), Tricaprilin (CER-0001), Bezisterim (NE3107), BAN2401, Gantenerumab, ALZT-OP1, Bryostatin-1, ALZT-OP1, Tricaprilin, and others
- Alzheimer’s Disease Therapeutic Assessment: Alzheimer’s Disease current marketed and Alzheimer’s Disease emerging therapies
- Alzheimer’s Disease Market Dynamics: Alzheimer’s Disease market drivers and Alzheimer’s Disease market barriers
- Alzheimer’s Disease Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Alzheimer’s Disease Unmet Needs, KOL’s views, Analyst’s views, Alzheimer’s Disease Market Access and Reimbursement
Discover more about therapies set to grab major Alzheimer’s Disease market share @ Alzheimer’s Disease Treatment Landscape
About DelveInsight
DelveInsight is a leading Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Alzheimer's disease (AD) - Epidemiology Forecast - 2034
Alzheimer's disease Epidemiology Forecast 2034 report delivers a detailed understanding of the disease, historical, and forecasted epidemiology in the 7MM.
Alzheimer's disease (AD) - Pipeline Insight, 2026
Alzheimer’s disease (AD) Pipeline Insights, 2026 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across the Alzheimer’s