Chronic Spontaneous Urticaria market size is projected to grow at a CAGR of 15.9% by 2034
Get a Sneak Peek at the Latest chronic spontaneous urticaria market size and forecast Report
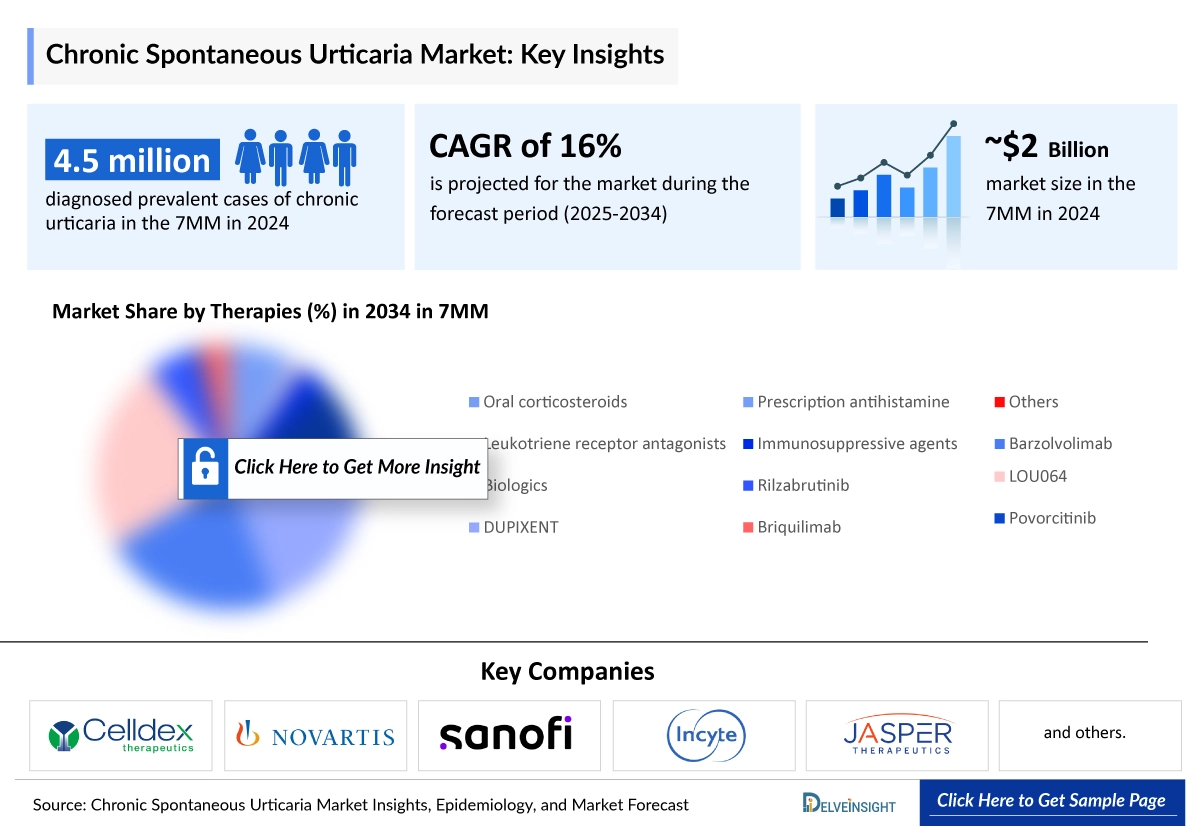
The Chronic Spontaneous Urticaria market size in the 7MM was estimated at around USD 2,000 million in 2025 and is expected to grow at a CAGR of 15.9% to reach USD 7,555 million by 2034, reflecting significant expansion over the forecast period. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Chronic Spontaneous Urticaria landscape.
In 2024, the Chronic Spontaneous Urticaria market in the United States was valued at around USD 1 billion and is projected to grow at a CAGR of 14% over the forecast period, driven by rising disease awareness and the introduction of emerging therapies. In 2024, the Chronic Spontaneous Urticaria market across the EU4 and the UK was estimated at around USD 620 million, accounting for nearly 30% of the total market revenue within the 7MM.
According to DelveInsight’s estimates, the total diagnosed prevalent cases of chronic urticaria in the 7MM were approximately 4.5 million in 2024, with these numbers anticipated to increase by 2034. Among the 7MM, the US accounted for nearly 19% of the total diagnosed prevalent cases of chronic urticaria, with around 885 thousand cases in 2024. These cases are expected to change during the forecast period (2025–2034). The EU4 and the UK accounted for approximately 39% of the total cases, while Japan accounted for around 42% in 2024.
DelveInsight’s estimates indicate that type-specific diagnosed prevalent cases of chronic urticaria, including Chronic Spontaneous Urticaria and Chronic Inducible Urticaria, were nearly 3.2 million and 1.5 million cases, respectively, in 2024 in the 7MM. The US accounted for approximately 600 thousand diagnosed prevalent cases of Chronic Spontaneous Urticaria in 2024, and these cases are projected to increase by 2034.
In 2024, among the EU4 and the UK, Germany reported the highest number of diagnosed prevalent cases of Chronic Spontaneous Urticaria with approximately 375 thousand cases, while Spain reported the lowest. Japan accounted for nearly 37% of the total diagnosed prevalent Chronic Spontaneous Urticaria cases across the 7MM in 2024.
DelveInsight’s severity-specific prevalence estimates for Japan in 2024 reported around 215 thousand, 455 thousand, and 515 thousand cases of mild, moderate, and severe Chronic Spontaneous Urticaria, respectively, with these numbers expected to change in the forecast period (2025–2034).
DelveInsight’s report “Chronic Spontaneous Urticaria Market Insights, Epidemiology, and Market Forecast-2034” provides a comprehensive analysis of the Chronic Spontaneous Urticaria landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
To Know in detail about the Chronic Spontaneous Urticaria market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Chronic Spontaneous Urticaria Market Forecast
Some of the key facts of the Chronic Spontaneous Urticaria Market Report:
- Key Chronic Spontaneous Urticaria Companies: Allakos, AstraZeneca, Biosana, Celldex Therapeutics, Celltrion, Eli Lilly and Company, Genentech, GI Innovation/Yuhan, Regeneron, GlaxoSmithKline, Taiho Pharma, Glenmark Pharmaceuticals, Gossamer Bio, Kiniksa Pharmaceuticals, Mycenax Biotech, Novartis, Regeneron, Synermore Biologics, UCB Biopharma, United BioPharma, and others
- Key Chronic Spontaneous Urticaria Therapies: Ligelizumab, Remibrutinib (LOU064), Tezepelumab, CDX-0159, GI-301, Dupilumab (DUPIXENT), UB-221, MTPS9579A, Lirentelimab (AK002), TAS5315, rilzabrutinib, GDC-0853, AZD1981, Desloratadine, and others
- The Chronic Spontaneous Urticaria epidemiology based on gender analyzed that Chronic Spontaneous Urticaria is more prominent in females in comparison to males
- The Chronic Spontaneous Urticaria market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Chronic Spontaneous Urticaria pipeline products will significantly revolutionize the Chronic Spontaneous Urticaria market dynamics.
Chronic Spontaneous Urticaria Overview
Chronic Spontaneous Urticaria (CSU) is a persistent skin disorder characterized by the spontaneous appearance of hives (wheals), swelling (angioedema), or both that last for six weeks or longer without any identifiable external trigger. The condition results from immune system dysregulation, leading to the release of histamine and other inflammatory mediators from mast cells. Symptoms include itching, redness, and swelling, which can significantly impact a patient’s quality of life. While antihistamines are commonly used, many patients require advanced biologic therapies such as omalizumab for effective symptom control.
Get a Free sample for the Chronic Spontaneous Urticaria Market Report: https://www.delveinsight.com/report-store/chronic-spontaneous-urticaria-market
Key Trends in Chronic Spontaneous Urticaria Therapeutics Market:
- Rising Focus on Biologic Therapies: Increasing adoption of biologics such as omalizumab and emerging monoclonal antibodies is transforming CSU management and improving patient outcomes.
- Pipeline Expansion and Innovation: Several novel agents targeting key immunological pathways, including anti-IgE and anti-IL-5, are in advanced stages of clinical development.
- Growing Disease Awareness: Enhanced understanding among healthcare professionals and patients is driving earlier diagnosis and better treatment adherence.
- Shift Toward Personalized Medicine: Research efforts are focusing on patient-specific treatment strategies to improve efficacy and minimize adverse effects.
- Market Growth Across Regions: Expanding healthcare access and increasing drug approvals in the US, EU, and Japan are fueling strong market growth projections through 2034.
Chronic Spontaneous Urticaria Epidemiology
The Chronic Spontaneous Urticaria epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Chronic Spontaneous Urticaria Epidemiology Segmentation:
The Chronic Spontaneous Urticaria market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Diagnosed Prevalent Cases of Chronic Urticaria
- Type-specific Cases of Chronic Urticaria
- Gender-specific Cases of Chronic Spontaneous Urticaria
- Age-specific Cases of Chronic Spontaneous Urticaria
- Severity-specific Cases of Chronic Spontaneous Urticaria
Download the report to understand which factors are driving Chronic Spontaneous Urticaria epidemiology trends @ Chronic Spontaneous Urticaria Epidemiology Forecast
Recent Developments In The Chronic Spontaneous Urticaria Treatment Landscape:
- In July 2025, Jasper Therapeutics, Inc. (Nasdaq: JSPR), a clinical-stage biotech developing briquilimab, a novel KIT (CD117)–targeting antibody therapy for mast cell–driven diseases such as chronic spontaneous urticaria (CSU) and chronic inducible urticaria (CIndU), has shared updated data from its BEACON Phase 1b/2a study of subcutaneous briquilimab in adults with CSU, along with a program update. Treatment with briquilimab in the 240mg and 360mg single-dose cohorts achieved rapid and deep disease control, with 8 of 9 participants (89%) attaining a complete response and 7 of 9 (78%) showing clinical response by week 2. Participants rolling over into the open-label extension at 180mg Q8W demonstrated strong efficacy, with 8 of 11 (73%) achieving a complete response by 12 weeks.
- In March 2025, Results from the Phase III REMIX-1 (NCT05030311) and REMIX-2 (NCT05032157) trials demonstrate that remibrutinib effectively alleviates symptoms of chronic spontaneous urticaria (CSU) in patients who do not respond to second-generation H1-antihistamines. The studies, published in The New England Journal of Medicine, showed that the drug has a rapid onset of action, with efficacy maintained for 24 weeks, and a favorable safety profile.
- In January 2025, Jasper Therapeutics, Inc. (Nasdaq: JSPR), a clinical-stage biotechnology company developing briquilimab, a novel c-Kit (CD117) targeting antibody for mast cell-driven diseases like chronic spontaneous urticaria (CSU), chronic inducible urticaria (CIndU), and asthma, has announced positive preliminary data from its ongoing BEACON Phase 1b/2a study of subcutaneous briquilimab in adults with CSU. The study reported significant reductions in UAS7 scores, with an average change of -26.6 at eight weeks in the 240mg single-dose cohort. Multiple dosing regimens at or above 120mg showed reductions of over -25 points. Clinical responses were observed within a week, and patients across all dose levels (80mg, 120mg, 180mg, and 240mg) achieved Complete Responses (UAS7 = 0), with all participants in the 240mg single-dose group maintaining their response through eight weeks. The treatment demonstrated dose-dependent durability, reductions in serum tryptase, and a favorable safety profile.
- In November 2024, The FDA accepted the resubmission of the supplemental biologics license application for Dupixent, developed by Regeneron, for the treatment of chronic spontaneous urticaria in both adults and children.
- In November 2024, The U.S. Food and Drug Administration (FDA) has accepted the resubmission of the supplemental biologics license application (sBLA) for Dupixent (dupilumab) to treat chronic spontaneous urticaria (CSU) in adults and pediatric patients aged 12 years and older, whose condition is not sufficiently managed with H1 antihistamine treatment
- In September 2024, Evommune announced the enrollment of the first patient in a Phase II trial of EVO756 in adults with CIndU.
Chronic Spontaneous Urticaria Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Chronic Spontaneous Urticaria market or expected to get launched during the study period. The analysis covers Chronic Spontaneous Urticaria market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Chronic Spontaneous Urticaria Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Chronic Spontaneous Urticaria Therapies and Key Companies
- Ligelizumab: Novartis
- Remibrutinib (LOU064): Novartis
- Tezepelumab: Amgen
- CDX-0159: Celldex Therapeutics
- GI-301: GI Innovation
- Dupilumab (DUPIXENT): Sanofi/ Regeneron
- UB-221: United BioPharma
- MTPS9579A: Genentech
- Lirentelimab (AK002): Allakos Inc.
- TAS5315: Taiho Pharma
- rilzabrutinib: Sanofi
- GDC-0853: Genentech, Inc.
- AZD1981: AstraZeneca
- Desloratadine: Organon and Co
Discover more about therapies set to grab major Chronic Spontaneous Urticaria market share @ Chronic Spontaneous Urticaria Treatment Market
Chronic Spontaneous Urticaria Market Drivers
- Rising Chronic Spontaneous Urticaria Prevalence: Increasing global incidence of chronic spontaneous urticaria, particularly among adults and women, is fueling demand for effective treatments.
- Advancements in Biologic Therapies: The introduction of targeted biologics like omalizumab and ligelizumab has transformed disease management and improved patient outcomes.
- Expanding Research and Development: Growing clinical research focused on identifying new molecular targets such as IgE, FcεRI, and IL-5 pathways is broadening the treatment landscape.
- Improved Diagnostic Awareness: Enhanced physician education and better understanding of disease mechanisms are leading to faster and more accurate diagnoses.
- Supportive Regulatory Environment: Fast-track approvals and orphan drug designations are encouraging innovation and accelerating therapy launches.
Chronic Spontaneous Urticaria Market Barriers
- High Treatment Costs for Chronic Spontaneous Urticaria: Biologic therapies are often expensive, limiting accessibility for patients, especially in developing regions.
- Limited Curative Options: Current treatments primarily manage symptoms rather than address the underlying cause, leaving unmet medical needs.
- Heterogeneous Disease Mechanisms: Variability in disease pathophysiology complicates treatment response and hinders personalized therapy approaches.
- Reimbursement Challenges: Stringent reimbursement policies and limited insurance coverage can restrict patient access to advanced biologics.
- Lack of Awareness in Mild Cases: Many patients with intermittent or mild symptoms remain undiagnosed or undertreated, reducing the overall treatment population.
Scope of the Chronic Spontaneous Urticaria Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Chronic Spontaneous Urticaria Companies: Allakos, AstraZeneca, Biosana, Celldex Therapeutics, Celltrion, Eli Lilly and Company, Genentech, GI Innovation/Yuhan, Regeneron, GlaxoSmithKline, Taiho Pharma, Glenmark Pharmaceuticals, Gossamer Bio, Kiniksa Pharmaceuticals, Mycenax Biotech, Novartis, Regeneron, Synermore Biologics, UCB Biopharma, United BioPharma, and others
- Key Chronic Spontaneous Urticaria Therapies: Ligelizumab, Remibrutinib (LOU064), Tezepelumab, CDX-0159, GI-301, Dupilumab (DUPIXENT), UB-221, MTPS9579A, Lirentelimab (AK002), TAS5315, rilzabrutinib, GDC-0853, AZD1981, Desloratadine, and others
- Chronic Spontaneous Urticaria Therapeutic Assessment: Chronic Spontaneous Urticaria current marketed and Chronic Spontaneous Urticaria emerging therapies
- Chronic Spontaneous Urticaria Market Dynamics: Chronic Spontaneous Urticaria market drivers and Chronic Spontaneous Urticaria market barriers Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Chronic Spontaneous Urticaria Unmet Needs, KOL’s views, Analyst’s views, Chronic Spontaneous Urticaria Market Access and Reimbursement
To know more about Chronic Spontaneous Urticaria companies working in the treatment market, visit @ Chronic Spontaneous Urticaria Clinical Trials and Therapeutic Assessment
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Chronic Spontaneous Urticaria (CSU) - Epidemiology Forecast - 2034
DelveInsight's Chronic spontaneous urticaria- Market Insights, Epidemiology and Market Forecast - 2034 report provides an in depth understanding of historical..
Chronic Spontaneous Urticaria- Pipeline Insight, 2025
"Chronic Spontaneous Urticaria Pipeline Insights, 2025" report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across..