Chronic Venous Insufficiency market size is projected to grow at a CAGR of 9.5% to reach USD 5,951 million by 2034
Get a Sneak Peek at the Latest chronic venous insufficiency market size and forecast Report
The Chronic Venous Insufficiency market size across the 7MM was valued at USD 2,652 million in 2025 and is expected to expand at a CAGR of 9.5%, reaching approximately USD 5,951 million by 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Chronic Venous Insufficiency landscape.
The US Chronic Venous Insufficiency market was valued at roughly USD 1,200 million in 2023 and is projected to expand throughout the 2025–2034 forecast period. By 2034, Laser Ablation/Radiofrequency Ablation is expected to generate the highest revenue at about USD 1,787.9 million, while TR987 is forecasted to bring in the lowest revenue at approximately USD 20.3 million across the 7MM.
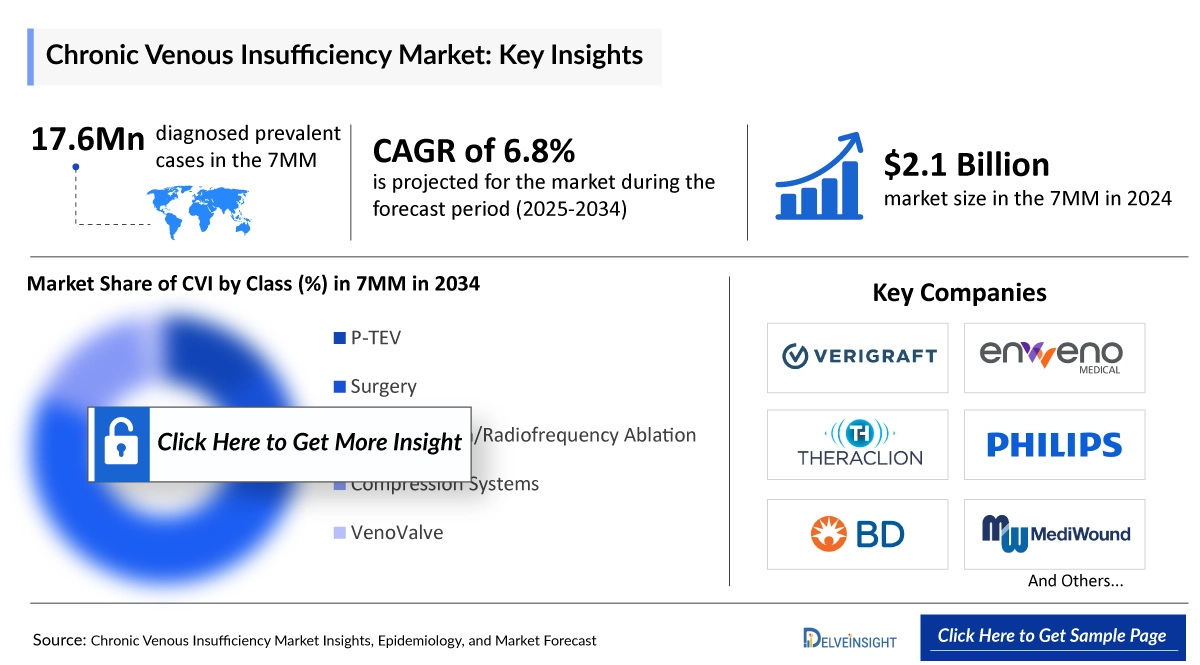
Across the 7MM, there were roughly 17.64 million diagnosed cases of chronic venous insufficiency (CVI) in 2020, and this number is projected to grow at a CAGR of 1.4% through 2034. In the US, the prevalence was notably higher in women, with about 4,900 cases compared to 1,600 in men, and these figures are expected to rise over the 2025–2034 forecast period. Additionally, the US recorded around 52.1 million diagnosed cases of chronic venous disease (CVD) in 2020, a number anticipated to reach nearly 64.93 million by 2034, increasing at a CAGR of 1.6%. In 2023, women continued to represent the majority of CVI cases in the US.
DelveInsight’s report “Chronic Venous Insufficiency Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Chronic Venous Insufficiency landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Chronic Venous Insufficiency market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Chronic Venous Insufficiency market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Chronic Venous Insufficiency Market Forecast
Some of the key facts of the Chronic Venous Insufficiency Market Report:
- Key Chronic Venous Insufficiency Companies: enVVeno Medical Corporation, Verigraft, MediWound, TissueTech/Amniox Medical, Alfasigma S.p.A, Boehringer Ingelheim, Takeda, Abbott, Servier, Ten Sun Pharma Company, Pfizer, AstraZeneca, JMI Laboratories, and others
- Key Chronic Venous Insufficiency Therapies: VenoValve, P-TEV, EscharEx, TTAX01, Vessel, Antistax®, Coumarin/troxerutin, Venocur Triplex®, Detralex, Esarin Gel, Sirolimus, ticagrelor, Neutrolin, and others
- In the United States, Chronic Venous Insufficiency displays a significant gender imbalance, with women representing 77.99% of cases, while men make up only 22.01%.
- The Chronic Venous Insufficiency market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Chronic Venous Insufficiency pipeline products will significantly revolutionize the Chronic Venous Insufficiency market dynamics.
Chronic Venous Insufficiency Overview
Chronic Venous Insufficiency (CVI) is a long-term condition in which the veins in the legs fail to efficiently return blood to the heart. This occurs due to damaged vein valves or weakened vein walls, leading to blood pooling in the lower limbs. Common symptoms include leg swelling, pain, heaviness, varicose veins, skin discoloration, and ulcers. Risk factors include aging, obesity, pregnancy, prolonged standing, and a history of deep vein thrombosis (DVT). Treatment options range from compression therapy and lifestyle changes to minimally invasive procedures or surgery to improve blood flow and prevent complications.
Get a Free sample for the Chronic Venous Insufficiency Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/chronic-venous-insufficiency-market
Key Trends in Chronic Venous Insufficiency Therapeutics Market:
- Shift Toward Minimally Invasive Treatment Options: Therapies such as endovenous laser ablation (EVLA), radiofrequency ablation (RFA), and sclerotherapy are increasingly preferred over conventional surgical stripping due to faster recovery and fewer complications.
- Growing Adoption of Compression Therapy & Advanced Medical Devices: Demand for smart compression garments, intermittent pneumatic compression (IPC) devices, and gradient stockings is rising, supported by technological enhancements and patient compliance tools.
- Rising Use of Pharmacological Agents Targeting Underlying Pathophysiology: Venoactive drugs (e.g., flavonoids, micronized purified flavonoid fraction), anticoagulants, and anti-inflammatory agents are gaining attention as adjunctive or standalone treatments for CVI management.
- Increased Investment in Regenerative & Biologic Therapies: Research into stem-cell therapy, growth factor modulation, and tissue engineering solutions is expanding as companies seek long-term healing approaches beyond symptom management.
- Growing Awareness & Diagnosis Due to Aging Population & Sedentary Lifestyle: Higher prevalence of obesity, desk jobs, and elderly patients is driving earlier diagnosis, preventive care, and demand for long-term CVI therapeutics.
- Rise in Combination Treatment Approaches: Healthcare providers are adopting integrated treatment plans involving minimally invasive procedures + pharmacotherapy + lifestyle management for enhanced patient outcomes.
- Expansion of Reimbursement Coverage & Outpatient Treatment Models: Increasing insurance support and shift from hospital-based to clinic/outpatient settings are improving treatment accessibility and driving market growth.
Chronic Venous Insufficiency Epidemiology
The Chronic Venous Insufficiency epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Chronic Venous Insufficiency Epidemiology Segmentation:
The Chronic Venous Insufficiency market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalent Cases of CVD
- Total Diagnosed Prevalent Cases of CVD
- Stage-Specific Diagnosed Prevalent cases of Chronic Venous Insufficiency
- Diagnosed Prevalent Cases of Chronic Venous Insufficiency
- Gender-Specific Cases of Chronic Venous Insufficiency
Download the report to understand which factors are driving Chronic Venous Insufficiency epidemiology trends @ Chronic Venous Insufficiency Epidemiology Forecast
Recent Developments in the Chronic Venous Insufficiency Treatment Landscape:
- In September 2025, enVVeno Medical Corporation (Nasdaq: NVNO) stated that it intends to submit a supervisory appeal following the not-approvable letter issued by the U.S. FDA’s Center for Devices and Radiological Health (CDRH). The letter, dated August 19, 2025, was issued in response to the company’s Premarket Approval (PMA) application for VenoValve®, a surgical venous valve implant developed to address severe deep chronic venous insufficiency (CVI).
- In August 2025, enVVeno Medical Corporation reported that the FDA has issued a not-approvable letter for its Premarket Approval (PMA) application for VenoValve®, a surgically implanted replacement venous valve intended for patients with severe deep chronic venous insufficiency (CVI).
- In July 2025, TR Therapeutics shared findings from a Phase 3 randomized, double-blind, parallel-group study assessing the efficacy, tolerability, and safety of TR987® 0.1% Gel compared to the standard of care for treating leg ulcers caused by chronic venous insufficiency.
- In October 2024, InterVene, Inc., a clinical-stage medical device company focused on developing interventional treatments for chronic venous insufficiency (CVI), announced the successful completion of its $13 million Series A funding round. The investment was co-led by new investor Treo Ventures and existing investor RiverVest Venture Partners®. As part of the financing, Brad Vale, founding general partner at Treo Ventures, has joined InterVene’s Board of Directors.
- In January 2024, VERIGRAFT achieved significant progress in its TECVI-1 first-in-man trial, having treated half of the participants. The trial reached the final regulatory safety milestone in early January and is showing sustained graft functionality 12 months after treatment.
- In March 2024, enVVeno Medical revealed positive topline efficacy data from the SAVVE US pivotal trial for the VenoValve, demonstrating notable clinical improvements. This data was presented at the 2024 American Venous Forum (AVF) Annual Meeting.
Chronic Venous Insufficiency Drugs Uptake and Pipeline Development Activities
The Chronic Venous Insufficiency drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Chronic Venous Insufficiency market or expected to get launched during the study period. The analysis covers Chronic Venous Insufficiency market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Chronic Venous Insufficiency Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Chronic Venous Insufficiency Therapies and Key Companies
- VenoValve: enVVeno Medical Corporation
- P-TEV: Verigraft
- EscharEx: MediWound
- TTAX01: TissueTech/Amniox Medical
- Vessel: Alfasigma S.p.A
- Antistax®: Boehringer Ingelheim
- Coumarin/troxerutin: Takeda
- Venocur Triplex®: Abbott
- Detralex: Servier
- Esarin Gel: Ten Sun Pharma Company\
- Sirolimus: Pfizer
- ticagrelor: AstraZeneca
- Neutrolin: JMI Laboratories
Discover more about therapies set to grab major Chronic Venous Insufficiency market share @ Chronic Venous Insufficiency Treatment Landscape
Chronic Venous Insufficiency Market Drivers
- Rising Prevalence Due to Aging Population & Sedentary Lifestyles: Increased cases linked to obesity, prolonged sitting/standing, and growing elderly population are expanding the patient base.
- Growing Adoption of Minimally Invasive Treatment Options: Rising preference for procedures like endovenous laser therapy, radiofrequency ablation, and sclerotherapy is boosting market growth.
- Technological Advancements in Diagnostic and Therapeutic Devices: Improved vascular imaging tools, compression systems, and catheter-based technologies are enhancing clinical outcomes and driving demand.
- Increasing Awareness and Early Diagnosis Efforts: More screening programs, public health campaigns, and physician education are accelerating treatment rates.
- Rising Healthcare Expenditure and Access to Vascular Care: Better reimbursement frameworks and expanding specialty clinics support overall market expansion.
Chronic Venous Insufficiency Market Barriers
- High Treatment Costs and Limited Reimbursement in Some Regions: Advanced vein procedures and long-term care products can be expensive, restricting patient access in low-income markets.
- Shortage of Skilled Vascular Specialists & Uneven Healthcare Infrastructure: Limited access to trained professionals, especially in rural and developing regions, slows adoption of advanced therapies.
- Underdiagnosis and Lack of Disease Awareness Among Patients: Many early-stage CVI cases go untreated due to misdiagnosis or perception that symptoms are cosmetic rather than medical.
- Risk of Complications and Recurrence After Treatment: Post-procedural complications such as thrombosis, nerve injury, or need for repeat interventions affect patient and physician confidence.
- Competition from Low-Cost Conservative Management: Widespread use of compression stockings and lifestyle-based management reduces demand for higher-priced interventional therapies.
Scope of the Chronic Venous Insufficiency Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Chronic Venous Insufficiency Companies: enVVeno Medical Corporation, Verigraft, MediWound, TissueTech/Amniox Medical, Alfasigma S.p.A, Boehringer Ingelheim, Takeda, Abbott, Servier, Ten Sun Pharma Company, Pfizer, AstraZeneca, JMI Laboratories, and others
- Key Chronic Venous Insufficiency Therapies: VenoValve, P-TEV, EscharEx, TTAX01, Vessel, Antistax®, Coumarin/troxerutin, Venocur Triplex®, Detralex, Esarin Gel, Sirolimus, ticagrelor, Neutrolin, and others
- Chronic Venous Insufficiency Therapeutic Assessment: Chronic Venous Insufficiency current marketed and Chronic Venous Insufficiency emerging therapies
- Chronic Venous Insufficiency Market Dynamics: Chronic Venous Insufficiency market drivers and Chronic Venous Insufficiency market barriers
- Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Chronic Venous Insufficiency Unmet Needs, KOL’s views, Analyst’s views, Chronic Venous Insufficiency Market Access and Reimbursement
To know more about Chronic Venous Insufficiency companies working in the treatment market, visit @ Chronic Venous Insufficiency Clinical Trials and Therapeutic Assessment
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Chronic Venous Insufficiency - Epidemiology Forecast - 2034
DelveInsight's Chronic Venous Insufficiency - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology..
Chronic Venous Insufficiency - Pipeline Insight, 2025
"Chronic Venous Insufficiency Pipeline Insights, 2025" report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across..