Clostridioides Difficile Infection Market size is projected to grow at a CAGR of 13.5% by 2034
Get a Sneak Peek at the Latest clostridium difficile infections clostridium difficile associated disease market insights Report
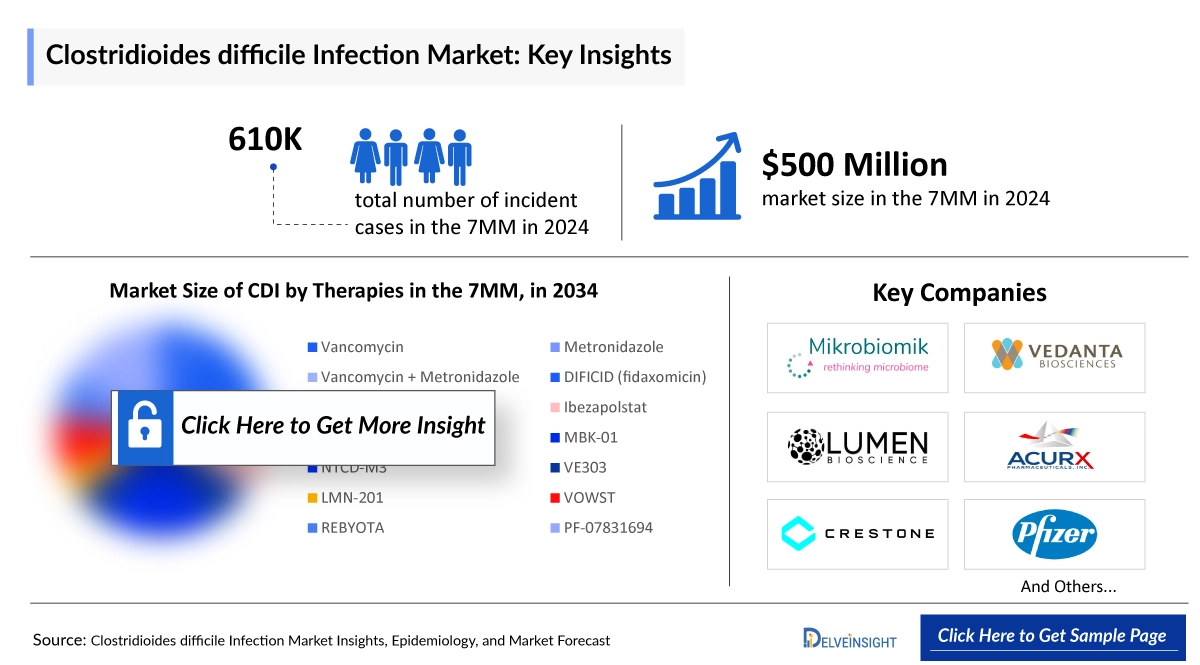
The Clostridioides difficile infection market size across the 7MM was valued at USD 551 million in 2025 and is expected to grow at a CAGR of 13.5%, reaching approximately USD 1,715 million by 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Clostridium Difficile Infections landscape.
DelveInsight’s estimates indicate that the US accounted for the largest market share of Clostridioides difficile infection in 2024. Among the EU4 and the UK, Germany held the highest market share, followed by France, while Spain recorded the lowest. In Japan, the market size was approximately USD 9 million in 2024 and is projected to increase over the forecast period (2025–2034).
In 2024, there were approximately 611,000 incident cases of Clostridioides difficile infection across the 7MM. The US reported the highest number, with around 380,000 cases, and this figure is expected to rise in the coming years. In 2024, Clostridioides difficile infection cases in the US were distributed as approximately 45% hospital-acquired (HA-CDI) and 55% community-acquired (CA-CDI). In Japan, significant gaps remain in epidemiological data, and the infection is often underdiagnosed despite its clinical importance.
In 2024, the incidence of Clostridioides difficile infection in the United States was higher among females than males. Across the 7MM, approximately 464,000 cases were identified as mild to moderate, while about 147,000 were classified as severe. In 2024, the UK reported an estimated 1,000 Clostridioides difficile infection cases in individuals under 18 years, around 7,500 cases in those aged 18–64 years, and approximately 17,000 cases among people aged 65 and older. This distribution underscores the substantially higher disease burden in the elderly population.
DelveInsight’s report “Clostridium Difficile Infections Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Clostridium Difficile Infections landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
The Clostridium Difficile Infections market report covers emerging drugs, current treatment practices, market share of the individual therapies, and current & forecasted market size from 2020 to 2034. It also evaluates the current treatment practice/algorithm, market drivers & barriers, and unmet medical needs to curate the best of the opportunities and assess the underlying potential of the market.
To Know in detail about the Clostridium Difficile Infections market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Clostridium Difficile Infections Market Forecast
Some of the key facts of the Clostridium Difficile Infections Market Report:
- Approved therapies for Clostridium difficile infection include Merck’s DIFICID (fidaxomicin) and ZINPLAVA (bezlotoxumab), which offer improved cure rates. However, their market availability is limited due to high costs.
- Companies focusing on Clostridium difficile infection are actively investing in R&D to develop new treatments. Prominent pipeline candidates include PF-06425090 (Pfizer’s vaccine), CP101 (Finch Therapeutics), VE303 (Vedanta Biosciences), and MGB-BP-3 (MGB Biopharma), with expected launches during the 2024–2034 forecast period.
- Key Clostridium Difficile Infections Companies: Merck & Co./Tillotts Pharma/Astellas Pharma, Seres Therapeutics/Nestlé Health Science, Acurx Pharmaceuticals, Mikrobiomik, Vedanta Biosciences, Adiso Therapeutics, Deinove, Acurx Pharmaceuticals, Inc, Lumen Bioscience, Inc, Summit Therapeutics, Finch Therapeutics Group, Seres Therapeutics, Vedanta Biosciences, MGB Biopharma, Replidyne, and others
- Key Clostridium Difficile Infections Therapies: DIFICID/DIFICLIR/DAFCLIR (fidaxomicin), VOWST, Ibezapolstat, MBK-01, VE303, ADS024, DNV3837, Ibezapolstat, LMN-201, Ridinilazole, CP101, SER 109, VE303, MGB-BP-3, CRS3123, and others
The Clostridium Difficile Infections epidemiology based on gender analyzed that more incident cases of Clostridium Difficile Infections were seen in females
Clostridium Difficile Infections Overview
Clostridium difficile infection (CDI) is a bacterial infection caused by Clostridium difficile, a toxin-producing bacterium that affects the colon. It often develops after the use of antibiotics, which disrupt the normal gut flora, allowing C. difficile to grow excessively.
The infection commonly leads to symptoms such as diarrhea, abdominal pain, fever, and, in severe cases, colitis or life-threatening complications. CDI is most prevalent among hospitalized or elderly patients and those with weakened immune systems. Early diagnosis, appropriate antibiotic therapy, and infection control measures are essential to prevent recurrence and spread.
Get a Free sample for the Clostridium Difficile Infections Market Report:
Clostridium Difficile Infections Epidemiology
The epidemiology section offers an in-depth look at the historical, current, and projected epidemiology trends across the seven major countries (7MM) from 2020 to 2034. By examining various studies and expert opinions, it identifies the factors driving present and future trends. This section also includes a comprehensive analysis of the diagnosed patient population and anticipated future patterns.
Clostridium Difficile Infections Epidemiology Segmentation:
The Clostridium Difficile Infections market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total incident cases of Clostridioides Difficile Infection
- Type-specific incident cases of Clostridioides Difficile Infection
- Gender-specific incident cases of Clostridioides Difficile Infection
- Severity-specific incident cases of Clostridioides Difficile Infection
- Age-specific incident cases of Clostridioides Difficile Infection
- Clostridioides Difficile Infection recurrence pool
- Clostridioides Difficile Infection mortality pool
Download the report to understand which factors are driving Clostridium Difficile Infections epidemiology trends @ Clostridium Difficile Infections Epidemiological Insights
Recent Developments In The Treatment Landscape:
- In April 2025, Lumen Bioscience shared top-line findings from the sentinel cohort of its RePreve Clinical Trial assessing LMN-201 for Clostridioides difficile infection (CDI). LMN-201 is an oral biologic combination therapy, provided in capsule form, designed to be administered alongside and after antibiotics to enhance clinical outcomes in CDI. The therapy is developed using Lumen’s proprietary spirulina-based GMP manufacturing platform.
- In February 2025, Microbiotica presented new insights into the mechanism of action of MB097 at the American Association for Cancer Research (AACR) Immuno-Oncology conference held in Los Angeles from February 23-26. MB097, a live biotherapeutic product (LBP), is being developed to complement immuno-oncology treatments, including MSD’s anti-PD-1 therapy, KEYTRUDA (pembrolizumab).
- In January 2025, Vedanta Biosciences, a late clinical-stage biotechnology company focused on developing defined bacterial consortia as oral therapies for gastrointestinal disorders, announced the publication of further findings from its Phase II CONSORTIUM trial of its lead candidate, VE303, aimed at preventing recurrent *Clostridioides difficile* infection (rCDI). The latest analyses were published this month in Nature Medicine and are accessible online
- In January 2025, MaaT Pharma revealed the topline results from ARES, a key European Phase 3 multicenter trial. This single-arm, open-label study assessed the efficacy and safety of MaaT013 in patients with acute gastrointestinal Graft-versus-Host Disease (GI-aGvHD). The trial focused on third-line treatment for patients who were steroid-resistant and either refractory or intolerant to ruxolitinib.
- In January 2025, PureTech Health plc announced that Vedanta Biosciences, a company it founded, has published new results from the Phase 2 CONSORTIUM study on VE303, its leading candidate under evaluation for preventing recurrent Clostridioides difficile infection (rCDI).
- In January 2025, Seres Therapeutics, Inc. revealed new translational biomarker findings from exploratory endpoints in its Phase 1b placebo-controlled SER-155 trial with patients receiving allogeneic stem cell transplantation (allo-HSCT).
Clostridium Difficile Infections Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Clostridium Difficile Infections market or expected to get launched during the study period. The analysis covers Clostridium Difficile Infections market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Clostridium Difficile Infections Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Clostridium Difficile Infections Therapies and Key Companies
- DIFICID/DIFICLIR/DAFCLIR (fidaxomicin): Merck & Co./Tillotts Pharma/Astellas Pharma
- VOWST: Seres Therapeutics/Nestlé Health Science
- Ibezapolstat: Acurx Pharmaceuticals
- MBK-01: Mikrobiomik
- VE303: Vedanta Biosciences
- ADS024: Adiso Therapeutics
- DNV3837: Deinove
- Ibezapolstat: Acurx Pharmaceuticals, Inc
- LMN-201:Lumen Bioscience, Inc
- Ridinilazole: Summit Therapeutics
- CP101: Finch Therapeutics Group
- SER 109: Seres Therapeutics
- VE303: Vedanta Biosciences
- MGB-BP-3: MGB Biopharma
- CRS3123: Replidyne
To know more about Clostridium Difficile Infections treatment, visit @ Clostridium Difficile Infections Medications
Clostridium Difficile Infections Market Drivers
- Rising incidence of hospital-acquired infections (HAIs) due to increased antibiotic usage.
- Growing elderly population with higher susceptibility to C. difficile infections.
- Improved diagnostic technologies enabling faster and more accurate detection.
- Introduction of novel antibiotics, vaccines, and microbiome-based therapies.
- Increasing awareness and surveillance programs for infection prevention and control.
- Strong R&D investments focused on developing safer and more effective treatment options.
Clostridium Difficile Infections Market Barriers
- High recurrence rates posing challenges for long-term disease management.
- Limited availability of highly effective and durable treatment options.
- Rising antimicrobial resistance (AMR) reducing the efficacy of existing antibiotics.
- High treatment costs and limited reimbursement in some healthcare systems.
- Delayed or inaccurate diagnosis in early disease stages.
- Lack of awareness among patients and healthcare providers in developing regions.
- Regulatory hurdles and lengthy approval timelines for emerging therapies.
Scope of the Clostridium Difficile Infections Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Clostridium Difficile Infections Companies: Merck & Co./Tillotts Pharma/Astellas Pharma, Seres Therapeutics/Nestlé Health Science, Acurx Pharmaceuticals, Mikrobiomik, Vedanta Biosciences, Adiso Therapeutics, Deinove, Acurx Pharmaceuticals, Inc, Lumen Bioscience, Inc, Summit Therapeutics, Finch Therapeutics Group, Seres Therapeutics, Vedanta Biosciences, MGB Biopharma, Replidyne, and others
- Key Clostridium Difficile Infections Therapies: DIFICID/DIFICLIR/DAFCLIR (fidaxomicin), VOWST, Ibezapolstat, MBK-01, VE303, ADS024, DNV3837, Ibezapolstat, LMN-201, Ridinilazole, CP101, SER 109, VE303, MGB-BP-3, CRS3123, and others
- Clostridium Difficile Infections Therapeutic Assessment: Clostridium Difficile Infections current marketed and Clostridium Difficile Infections emerging therapies
- Clostridium Difficile Infections Market Dynamics: Clostridium Difficile Infections market drivers and Clostridium Difficile Infections market barriers
- Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Clostridium Difficile Infections Unmet Needs, KOL’s views, Analyst’s views, Clostridium Difficile Infections Market Access and Reimbursement
Discover more about therapies set to grab major Clostridium Difficile Infections market share @ Clostridium Difficile Infections Treatment Landscape
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Clostridioides Difficile Infections - Epidemiology Forecast - 2034
Clostridium Difficile Infections Epidemiology Forecast report delivers an in-depth understanding of the historical, and forecasted epidemiology of Clostridium Difficile Infections in the 7MM