Crohn’s Disease Market Size in the 7MM to Reach USD 16.8 Billion by 2034 at a CAGR of 4.40%
Get a Sneak Peek at the Latest crohns disease market forecast and insight Report
The Crohn’s Disease market size across the seven major markets (7MM) is projected to increase from approximately USD 11,367 million in 2025 to around USD 16,840 million by 2034. During this period, the market in key regions including the United States, EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan is expected to grow at a compound annual growth rate (CAGR) of 4.40% through 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Crohn’s Disease landscape.
In 2024, the United States accounted for the largest share of the Crohn’s disease market among the seven major markets (7MM), with a value of nearly USD 8,000 million, and it is expected to expand further at a CAGR of about 4.5%. Within Europe, Germany reported the highest Crohn’s disease market size at approximately USD 670 million, while Spain recorded the smallest market at around USD 207 million. Meanwhile, Japan’s Crohn’s disease market was valued at nearly USD 423 million in 2024, contributing roughly 4% to the total 7MM market.
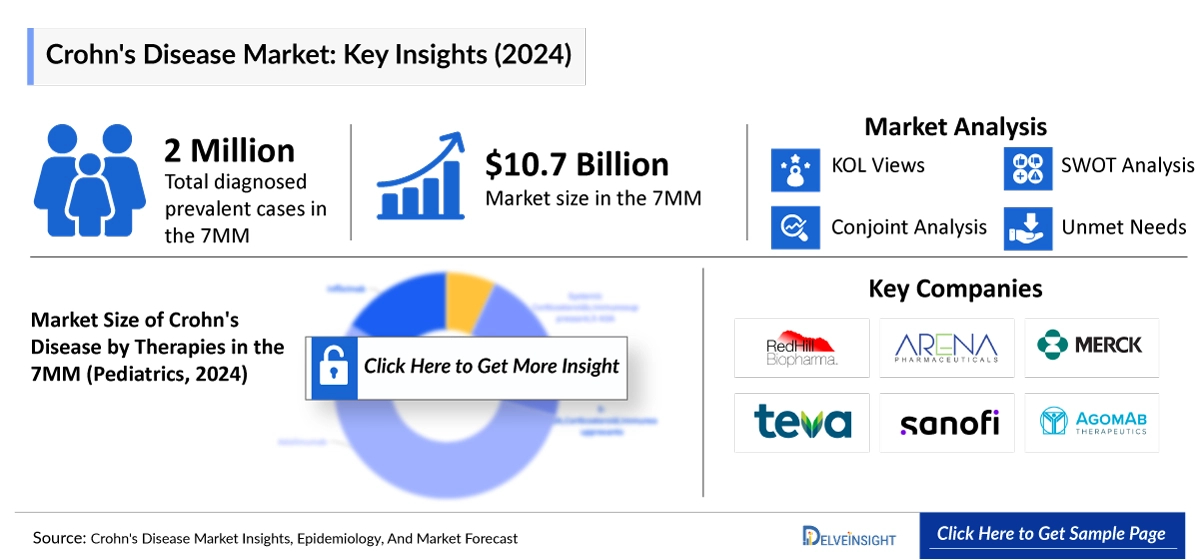
In 2024, the United States reported approximately 1.1 million diagnosed prevalent cases of Crohn’s disease, representing the largest share of the patient population among the seven major markets (7MM). The US accounted for nearly 54% of the total diagnosed prevalent cases in the 7MM, while the EU4 and the UK collectively represented around 42%, and Japan contributed about 4% in the same year.
Among the EU4 countries, Germany recorded the highest number of diagnosed Crohn’s disease cases at nearly 299,000, followed by the UK with approximately 234,000 cases. In contrast, Spain reported the lowest number of cases, with about 93,000 in 2024. Additionally, in the United States, around 461,000 cases were classified as mild severity, while nearly 755,000 cases were identified as moderate-to-severe in 2024.
According to DelveInsight’s estimates, in 2024, the age-specific distribution of diagnosed prevalent Crohn’s disease cases in the United States showed the highest number in the 18–44 years age group, with around 657,000 cases. This was followed by the 45–64 years group with approximately 298,000 cases, the 65–84 years group with about 104,000 cases, the 0–17 years group with nearly 81,000 cases, and individuals aged 85 years and above with roughly 11,000 cases.
DelveInsight’s report, “Crohn’s Disease Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Crohn’s Disease landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Crohn’s Disease market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Crohn’s Disease market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Crohn’s Disease Market Forecast
Some of the key facts of the Crohn’s Disease Market Report:
- Biosimilars have played a major role in shaping the market by lowering treatment costs and expanding patient access to biologic therapies. The first biosimilar to REMICADE received approval in 2016, and it was subsequently followed by additional biosimilars such as INFLECTRA, RENFLEXIS, and AVSOLA.
- Key Crohn’s Disease Companies: Merck, Eli Lilly and Company, RedHill Biopharma, Bristol Myers Squibb, Dr. Falk Pharma GmbH, Janssen-Cilag Ltd., AbbVie, EA Pharma Co., Ltd., Erasmus Medical Center, Takeda, Pfizer, Provention Bio, Inc., Mitsubishi Tanabe Pharma Corporation, and others
- Key Crohn’s Disease Therapies: OMVOH (mirikizumab/LY3074828), ENTYVIO (vedolizumab), Tulisokibart (MK-7240, PRA023), Mirikizumab, RHB-104, ZEPOSIA (ozanimod), budesonide, Guselkumab, Risankizumab SC, E6011, Infliximab, CDPATH™, Etrasimod, PRV-6527, MT-1303, and others
- The Crohn’s Disease market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Crohn’s Disease pipeline products will significantly revolutionize the Crohn’s Disease market dynamics.
Crohn’s Disease Overview
Crohn’s Disease is a chronic inflammatory condition of the digestive tract that can affect any part from the mouth to the anus. It causes symptoms like abdominal pain, diarrhea, weight loss, and fatigue. The exact cause is unknown, but it involves an abnormal immune response, genetics, and environmental factors. Crohn’s Disease is a type of inflammatory bowel disease (IBD) that can lead to complications such as strictures, fistulas, and malnutrition. Treatment focuses on reducing inflammation, managing symptoms, and maintaining remission.
Get a Free sample for the Crohn’s Disease Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/crohns-disease-cd-market
Key Trends in Crohn’s Disease Therapeutics Market:
- Growing adoption of biologic therapies, including anti-TNF agents, IL-12/23 inhibitors, and integrin inhibitors, improving disease management and patient outcomes.
- Increasing development of next-generation targeted therapies, such as JAK inhibitors and novel monoclonal antibodies, expanding treatment options.
- Rising availability of biosimilars, which are helping reduce treatment costs and improve patient access to biologic therapies.
- Strong pipeline of emerging drugs and clinical trials, focusing on precision medicine and personalized treatment approaches.
- Advancements in early diagnosis and disease monitoring, supported by improved biomarkers and imaging technologies.
Crohn’s Disease Epidemiology
The epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Crohn’s Disease Epidemiology Segmentation:
The Crohn’s Disease market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Diagnosed prevalent cases of Crohn’s disease
- Age-specific diagnosed prevalent cases of Crohn’s disease
- Severity-specific diagnosed prevalent cases of Crohn’s disease
- Treated cases of Crohn’s disease
Download the report to understand which factors are driving Crohn’s Disease epidemiology trends @ Crohn’s Disease Epidemiology Forecast
Recent Development In The Crohn’s Disease Treatment Landscape:
- In March 2026, AbbVie enters the current news cycle with its shares priced at around $234.26 and long-term returns that many investors may consider significant, including gains of about 14.5% over the past year and approximately 167.3% over the last five years. For a company of AbbVie’s scale, new late-stage clinical data in Crohn’s disease along with a recent FDA approval in leukemia could play an important role in shaping investor perspectives regarding the strength of its pipeline and the long-term sustainability of its major franchises.
- In March 2026, Redx Pharma Ltd, based in Alderley Park, UK, announced that the U.S. Food and Drug Administration has granted Fast Track Designation to RXC008 for the treatment of fibrostenotic Crohn’s disease, according to a company press release. RXC008 is a gastrointestinal-restricted pan-ROCK inhibitor currently under development as a potential therapy for this condition. The Fast Track designation is intended to accelerate the development and regulatory review of drugs that address serious diseases with significant unmet medical needs.
- In November 2025, Gomab Therapeutics is advancing its lead candidate, ontunisertib, into a Phase IIb trial after positive results in fibrostenosing Crohn’s disease (FSCD). In the Phase IIa STENOVA study (NCT05843578), the oral activin receptor-like kinase 5 (ALK5) inhibitor demonstrated safety and tolerability over 12 weeks in patients with symptomatic ileal narrowing. Adverse event incidence and severity were comparable between the treatment and placebo groups, and no treatment-emergent cardiac toxicity or inflammation was observed.
- In November 2025, Sandoz’s Tyruko (natalizumab-sztn) is now available for prescription in the U.S. Tyruko is the first FDA-approved biosimilar to Tysabri (natalizumab), indicated for adult patients with relapsing multiple sclerosis and for those with moderate to severe Crohn’s disease. The FDA granted approval in August 2023, with a planned 2024 launch contingent on a second FDA approval for a test detecting anti-JVC antibodies. Natalizumab therapies carry a risk of progressive multifocal leukoencephalopathy (PML), a rare brain infection caused by the John Cunningham virus, which can occur in immunocompromised patients.
- In April 2025, Sanofi decided to discontinue the development of its oral tumour necrosis factor (TNF) inhibitor, balinatunfib, as a standalone treatment after it failed to meet the primary endpoint in a Phase II trial. Preliminary findings from the SPECIFIC-PSO Phase II study (NCT06073119) in psoriasis patients indicated that balinatunfib produced clinically meaningful improvements in the primary endpoint, PASI-75 response, with efficacy similar to other oral psoriasis treatments. However, it did not achieve statistical significance, which Sanofi attributed to “the nature of this limited Phase II study.” PASI-75 is a standard measure assessing whether a patient has achieved a 75% reduction in symptoms from baseline.
- In March 2025, Agomab Therapeutics reported positive interim findings from 44 patients who completed treatment in the ongoing STENOVA1 Phase IIa trial of AGMB-129, an oral gastrointestinal (GI)-restricted small molecule ALK5 (TGF-β RI or ALK5) inhibitor being developed as a potential therapy for Fibrostenosing Crohn’s Disease (FSCD).
- In March 2025, Celltrion announced the U.S. launch of STEQEYMA® (ustekinumab-stba), a biosimilar to STELARA® (ustekinumab), after receiving FDA approval in December 2024. STEQEYMA is authorized for the same indications as STELARA, ensuring reliable treatment options for both patients and healthcare providers.
- In March 2025, Johnson & Johnson announced that the FDA has approved TREMFYA® (guselkumab), making it the first and only IL-23 inhibitor offering both subcutaneous (SC) and intravenous (IV) induction options for adults with moderately to severely active Crohn’s disease (CD), a chronic inflammatory condition affecting the gastrointestinal tract.
- In February 2025, Eli Lilly shared findings from the VIVID-2 open-label extension study at the Crohn's and Colitis Congress (CCC), revealing that most patients with moderately-to-severely active Crohn's disease treated continuously with OMVOH for two years achieved sustained clinical and endoscopic outcomes, including 43.8% of patients who had previously failed biologic therapies.
- In January 2025, The FDA approved OMVOH (mirikizumab) for Crohn’s disease, reinforcing the role of IL-23 inhibitors. Demonstrating robust long-term effectiveness, OMVOH is also under investigation for use in pediatric patients, potentially fulfilling a critical unmet need in this group.
Crohn’s Disease Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Crohn’s Disease market or expected to get launched during the study period. The analysis covers Crohn’s Disease market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Crohn’s Disease Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Crohn’s Disease Therapies and Key Companies
- OMVOH (mirikizumab/LY3074828): Eli Lilly
- ENTYVIO (vedolizumab): Takeda Pharmaceutical
- Tulisokibart (MK-7240, PRA023): Merck
- AGMB-129: Agomab Therapeutics
- TREMFYA: Johnson & Johnson
- SKYRIZI + ABBV-382: Abbvie
- Mirikizumab: Eli Lilly and Company
- RHB-104: RedHill Biopharma
- ZEPOSIA (ozanimod): Bristol Myers Squibb
- budesonide: Dr. Falk Pharma GmbH
- Guselkumab: Janssen-Cilag Ltd.
- Risankizumab SC: AbbVie
- E6011: EA Pharma Co., Ltd.
- Infliximab: Erasmus Medical Center
- CDPATH™: Takeda
- Etrasimod: Pfizer
- PRV-6527: Provention Bio, Inc.
- MT-1303: Mitsubishi Tanabe Pharma Corporation
- E6011: EA Pharma
Discover more about therapies set to grab major Crohn’s Disease market share @ Crohn’s Disease Treatment Landscape
Crohn’s Disease Market Drivers
- Rising prevalence of Crohn’s disease worldwide: Increasing incidence of inflammatory bowel diseases due to lifestyle changes, genetic predisposition, and environmental factors is significantly driving the demand for effective treatment options.
- Advancements in biologics and targeted therapies: Innovations such as anti-TNF agents, JAK inhibitors, and monoclonal antibodies have improved treatment outcomes, boosting the adoption of advanced therapeutics in the Crohn’s disease market.
- Growing awareness and improved diagnosis rates: Increased patient awareness, improved diagnostic technologies, and early screening initiatives are enabling earlier detection and treatment of Crohn’s disease, expanding the patient pool.
- Increasing investments in R&D and clinical trials: Pharmaceutical companies and research institutions are investing heavily in novel drug development, including microbiome therapies, stem-cell approaches, and personalized medicine.
- Expansion of healthcare infrastructure and reimbursement support: Improved healthcare spending, insurance coverage, and access to specialized care are facilitating the adoption of advanced therapies globally.
Crohn’s Disease Market Barriers
- High cost of biologics and advanced therapies: Expensive biologic drugs and long-term treatment regimens place a financial burden on patients and healthcare systems, limiting widespread access.
- Stringent regulatory requirements and long approval timelines: Regulatory agencies require extensive safety and efficacy data for new Crohn’s disease drugs, leading to lengthy and costly development processes.
- Complex and heterogeneous disease nature: Crohn’s disease varies significantly between patients, making it difficult to develop universally effective therapies and complicating clinical trials.
- Side effects and limited long-term efficacy of existing treatments: Conventional therapies may cause adverse effects or lose effectiveness over time, affecting patient adherence and treatment outcomes.
- Limited access to specialized healthcare in some regions: Shortage of trained gastroenterologists and inadequate healthcare infrastructure in developing regions can delay diagnosis and treatment.
Scope of the Crohn’s Disease Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Crohn’s Disease Companies: Merck, Eli Lilly and Company, RedHill Biopharma, Bristol Myers Squibb, Dr. Falk Pharma GmbH, Janssen-Cilag Ltd., AbbVie, EA Pharma Co., Ltd., Erasmus Medical Center, Takeda, Pfizer, Provention Bio, Inc., Mitsubishi Tanabe Pharma Corporation, and others
- Key Crohn’s Disease Therapies: OMVOH (mirikizumab/LY3074828), ENTYVIO (vedolizumab), Tulisokibart (MK-7240, PRA023), Mirikizumab, RHB-104, ZEPOSIA (ozanimod), budesonide, Guselkumab, Risankizumab SC, E6011, Infliximab, CDPATH™, Etrasimod, PRV-6527, MT-1303, and others
- Crohn’s Disease Therapeutic Assessment: Crohn’s Disease current marketed and Crohn’s Disease emerging therapies
- Crohn’s Disease Market Dynamics: Crohn’s Disease market drivers and Crohn’s Disease market barriers
- Crohn’s Disease Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Crohn’s Disease Unmet Needs, KOL’s views, Analyst’s views, Crohn’s Disease Market Access and Reimbursement
To know more about Crohn’s Disease companies working in the treatment market, visit @ Crohn’s Disease Clinical Trials and Therapeutic Assessment
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Crohn’s disease - Pipeline Insight, 2026
Crohn's Disease (CD) Pipeline Insights, 2026 report by DelveInsight outlays comprehensive insights of present clinical development scenario..
Crohn’s Disease – Epidemiology Forecast – 2034
DelveInsight's Crohn's Disease Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical and forecasted epidemiology of Crohn's Disease in United States