Crohn's Disease Market is Expected to Grow at a CAGR of 4.40% Through 2034, Driven by Revolutionary Therapies and Expanding Treatment Arsenal | DelveInsight
Get a Sneak Peek at the Latest crohns disease treatment market Report
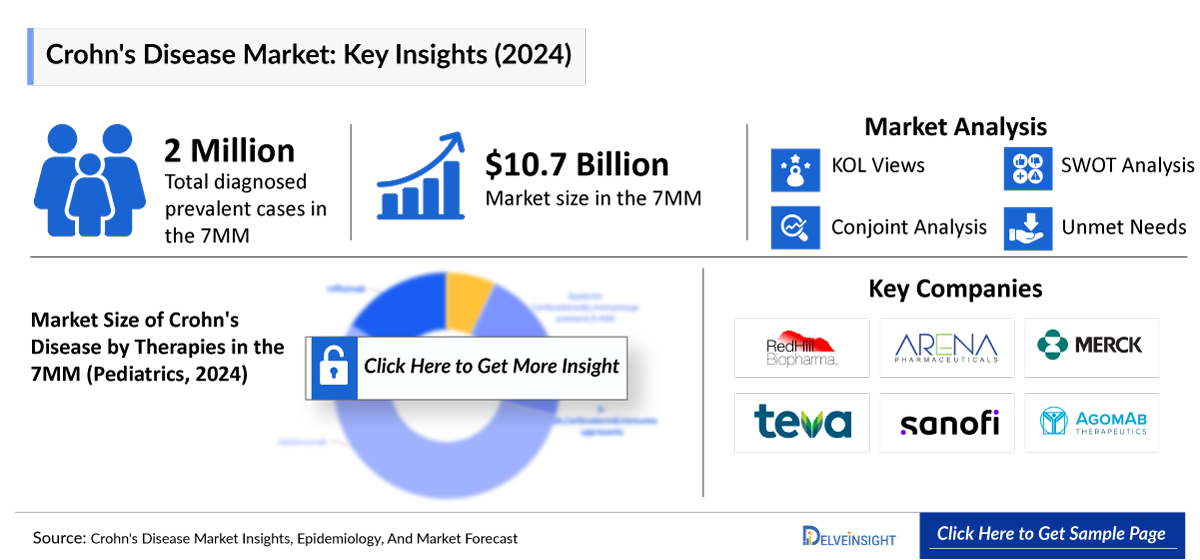
DelveInsight Business Research’s comprehensive "Crohn's Disease Market Insights, Epidemiology and Market Forecast – 2034" report reveals significant market expansion opportunities for pharmaceutical executives. The report demonstrates that the Crohn's Disease therapeutics market is projected to grow with a robust compound annual growth rate (CAGR) of 4.40% across the seven major markets (7MM), thanks to key Crohn’s disease companies including Takeda Pharmaceutical, Janssen Pharmaceuticals, UCB, Biogen, AbbVie, AstraZeneca, NImmune Biopharma, Tillotts Pharma (Zeria Pharmaceutical), Gilead Sciences, Galapagos, Boehringer Ingelheim, Bristol Myers Squibb, Eli Lilly and Company, RedHill Biopharma, Arena Pharmaceuticals, Mesoblast, Merck, Pfizer, Agomab Therapeutics, Teva Pharmaceuticals, Sanofi, and others, all actively engaged in developing, marketing, or advancing novel therapies for Crohn's Disease.
Crohn's Disease Market Dynamics Signal Unprecedented Growth Opportunity
The Crohn's Disease treatment landscape is experiencing a transformative period, with the United States commanding the largest market share at approximately USD 8.0 billion in 2024, representing 74% of the total 7MM. This dominance reflects both the higher prevalence of diagnosed cases and the premium pricing environment for advanced biologics in the American healthcare system. Furthermore, according to Crohn’s disease epidemiology analysis, the total diagnosed prevalent cases across the 7MM currently stand at approximately 2 million patients, with the United States accounting for 1.1 million cases that are expected to reach 1.5 million by 2034.
DelveInsight’s Crohn’s Disease epidemiology segmentation reveals that pediatric patients contribute roughly 10% of overall revenue generation, creating specialized opportunities for pharmaceutical companies developing age-appropriate formulations. The severity-specific case distribution shows moderate-to-severe cases representing 62% of the US patient population, while mild cases account for 38%, highlighting the significant unmet medical need for effective therapies targeting more severe disease manifestations.
Recent Regulatory Approvals Reshape Crohn's Disease Treatment Landscape
The year 2025 has marked a watershed moment for Crohn's Disease therapeutics, with several groundbreaking approvals transforming the treatment landscape. In January 2025, the FDA approved Eli Lilly's OMVOH (mirikizumab-mrkz) for the treatment of moderately to severely active Crohn's disease in adults, marking the first biologic in over 15 years to provide two-year Phase 3 efficacy data at the time of approval. This IL-23p19 inhibitor demonstrated significant clinical remission and endoscopic response rates in the pivotal VIVID-1 study, offering patients a novel mechanism of action targeting intestinal inflammation.
Additionally, March 2025 brought another significant milestone with Johnson & Johnson's TREMFYA (guselkumab) receiving FDA approval as the first and only IL-23 inhibitor offering both subcutaneous and intravenous induction options for adults with moderately to severely active Crohn's disease. This approval represents a paradigm shift in treatment convenience, allowing patients greater flexibility in therapy administration while maintaining therapeutic efficacy.
The biosimilar landscape has also experienced dramatic expansion in 2025, with multiple STELARA (ustekinumab) biosimilars launching to increase patient access and reduce treatment costs. Notably, Celltrion's STEQEYMA (ustekinumab-stba) launched in March 2025 with an impressive 85% discount to the reference product's wholesale acquisition cost, potentially democratizing access to IL-12/23 inhibitor therapy. Additionally, Teva Pharmaceuticals and Alvotech's SELARSDI (ustekinumab-aekn) received FDA approval as an interchangeable biosimilar, further intensifying market competition.
Learn more about the recent advancements in Crohn’s disease treatment landscape @ Crohn’s Disease Recent Developments
Robust Clinical Pipeline Promises Continued Innovation
The Crohn's Disease clinical pipeline demonstrates remarkable depth and diversity, with leading pharmaceutical companies investing heavily in novel therapeutic targets and mechanisms of action. Merck's tulisokibart (MK-7240), targeting the novel TL1A pathway, has shown exceptional promise in Phase 2 trials, with recent data demonstrating that nearly 50% of patients achieved clinical remission compared to 16% in historical studies. This investigational anti-TL1A monoclonal antibody is currently advancing through Phase 3 development and represents a potentially paradigm-shifting approach to treating both inflammation and fibrosis in Crohn's disease.
RedHill Biopharma's RHB-204 program has received positive FDA feedback for a groundbreaking Phase 2 study design that will test the first-ever clinical trial in a specifically defined Mycobacterium avium subspecies paratuberculosis-positive (MAP+) Crohn's disease patient population. This innovative approach targets MAP as a potential root cause of Crohn's disease, offering the possibility of treating both the suspected underlying infection and disease symptoms simultaneously.
The S1P receptor modulator class is also gaining momentum, with Pfizer's VELSIPITY (etrasimod) demonstrating strong efficacy in ulcerative colitis and showing potential for Crohn's disease applications. This oral, once-daily therapy offers patients a convenient alternative to injectable biologics while maintaining robust anti-inflammatory activity.
Current Treatment Landscape Dominated by Biologics
The existing Crohn's Disease treatment option is dominated by biologic therapies, with adalimumab (HUMIRA and biosimilars) commanding the largest market share at approximately USD 4.0 billion in 2024 across the 7MM. The current therapeutic arsenal includes well-established TNF inhibitors such as infliximab (REMICADE and biosimilars), integrin receptor antagonists like ENTYVIO (vedolizumab), and IL-12/23 inhibitors including STELARA (ustekinumab).
The recent addition of JAK inhibitors to the Crohn's Disease treatment options, exemplified by RINVOQ (upadacitinib), has provided physicians with oral small-molecule alternatives for patients who do not respond adequately to biologic therapies. This diversification of therapeutic mechanisms reflects the complex and heterogeneous nature of Crohn's disease pathophysiology, requiring personalized treatment approaches based on individual patient characteristics and disease manifestations.
Emerging Therapeutic Modalities Drive Future Growth
The Crohn's Disease pipeline analysis reveals several innovative therapeutic approaches that are expected to drive market expansion during the forecast period. IL-23 selective inhibitors, including SKYRIZI (risankizumab) and the recently approved OMVOH and TREMFYA, represent a refined approach to cytokine inhibition with potentially improved safety profiles compared to broader IL-12/23 inhibitors.
JAK inhibitor development continues with Pfizer's LITFULO (ritlecitinib) targeting JAK3 specifically, offering potential advantages in selectivity and safety compared to pan-JAK inhibitors. Additionally, novel targets such as TGFβR1 (ALK5) are being explored by Agomab Therapeutics with AGMB-129, specifically designed for fibrostenosing Crohn's disease, addressing a significant unmet medical need in patients with stricturing complications.
Download the Crohn’s Disease Market report to access in-depth epidemiological data, pipeline analysis, market forecasts, and expert perspectives that will inform your strategic decision-making in this lucrative and growing market opportunity.
Crohn's Disease Competitive Landscape Intensifies with New Market Entrants
The competitive landscape encompasses a diverse array of established pharmaceutical leaders and emerging biotechnology companies. Major Crohn's Disease companies include Takeda Pharmaceutical, Janssen Pharmaceuticals, UCB, Biogen, AbbVie, AstraZeneca, and Eli Lilly, each contributing unique therapeutic approaches and mechanisms of action. Emerging companies such as RedHill Biopharma, Arena Pharmaceuticals, Mesoblast, and Agomab Therapeutics are advancing innovative programs targeting novel pathways and patient populations.
The Crohn's Disease market dynamics are further complicated by the ongoing biosimilar wave, which is simultaneously reducing treatment costs while intensifying price competition among originator products. This environment creates opportunities for companies with differentiated products offering superior efficacy, safety, or convenience profiles compared to existing standards of care.
The report also identifies several critical success factors for pharmaceutical companies operating in the Crohn's disease treatment market, including the development of oral formulations, targeting of treatment-refractory populations, and integration of personalized medicine approaches.
Crohn's Disease Market Access and Regulatory Considerations
DelveInsight’s Crohn's Disease market report covers multiple regulatory jurisdictions with varying approval pathways and reimbursement considerations. The European Medicines Agency (EMA) has generally aligned with FDA decisions regarding major new approvals, with TREMFYA receiving approval for Crohn's disease and ulcerative colitis from the MHRA in May 2025. This regulatory harmonization facilitates global development strategies and reduces time-to-market for innovative therapies.
Pediatric Crohn's Disease patient pool represents a significant opportunity, with Johnson & Johnson seeking FDA approval for STELARA in children two years and older with moderately to severely active Crohn's disease. The pediatric market currently relies heavily on HUMIRA and REMICADE, creating substantial opportunities for companies developing age-appropriate formulations and dosing regimens.
Strategic Implications for Pharmaceutical Executives
The evolving Crohn's Disease therapeutic market presents significant strategic opportunities for pharmaceutical companies across the development spectrum. The market's projected growth to USD 16.840 billion by 2034 reflects both increasing disease prevalence and the premium pricing sustainability for innovative therapies that demonstrate superior clinical outcomes.
Companies should consider the competitive advantages of novel mechanisms of action, improved dosing convenience, and enhanced safety profiles when developing market entry strategies. The success of recent approvals demonstrates that regulatory agencies and payers are willing to embrace innovation, which can improve patient outcomes and clear patient benefit propositions.
The comprehensive DelveInsight Crohn's Disease Market Report provides pharmaceutical executives with the detailed market intelligence, competitive analysis, and strategic insights necessary to navigate this complex and rapidly evolving therapeutic landscape.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Crohn's Disease Market Insight, Epidemiology And Market Forecast - 2034
DelveInsight's Crohn's Disease (CD) Market Insights, Epidemiology, and Market Forecast-2034" report delivers an in-depth understanding of the Crohn's Disease (CD), historical and forecasted..

