Hemophilia A market is projected to grow at a CAGR of 2.9% to reach USD 16,756 million by 2034
Get a Sneak Peek at the Latest hemophilia a market size and forecast Report
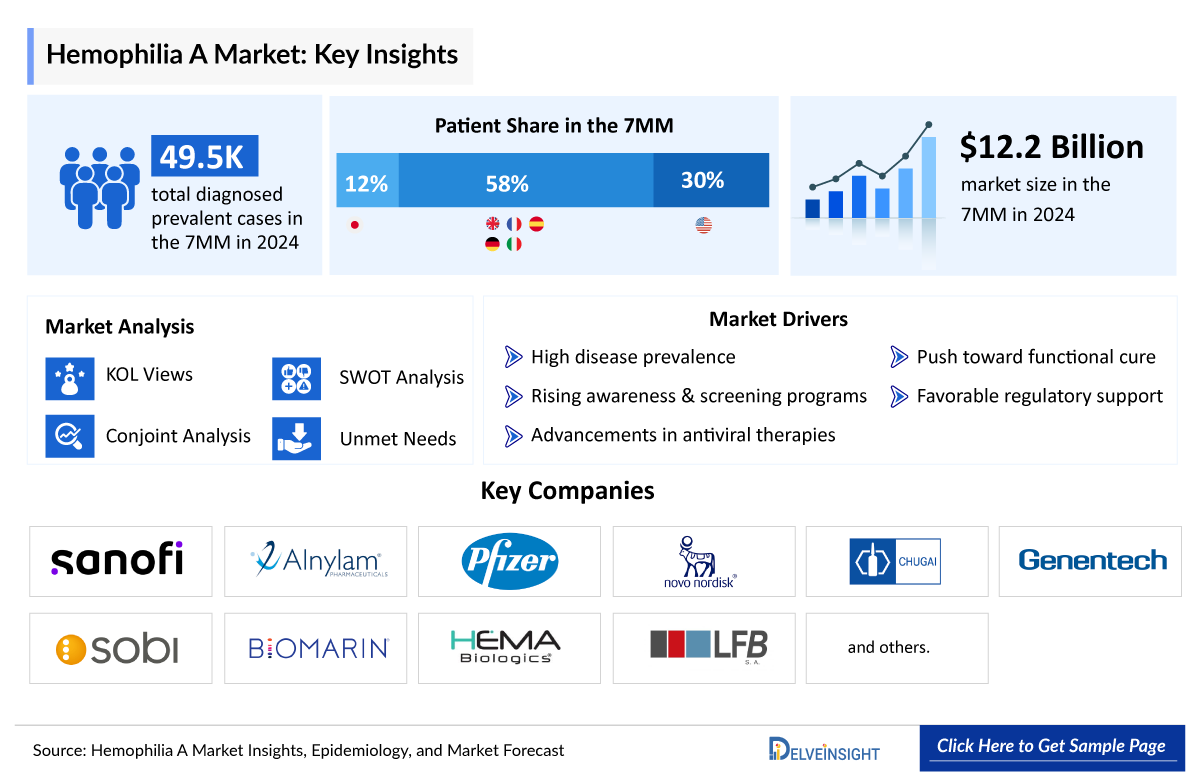
In 2025, the Hemophilia A market across the seven major markets (7MM) was valued at USD 12,918 million and is expected to expand at a CAGR of 2.9%, reaching approximately USD 16,756 million by 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Hemophilia A landscape.
Several pharmaceutical companies are actively developing innovative therapies for Hemophilia A, including Novo Nordisk (Mim8, ALHEMO), Sangamo Therapeutics (Giroctocogene fitelparvovec), Roche/Chugai (NXT007), ASC Therapeutics (ASC-618), Ultragenyx (DTX201), and others.
In 2024, the total diagnosed prevalent population of Hemophilia A across the seven major markets (7MM) was approximately 49,500 cases, with projections indicating further growth over the forecast period. The United States accounted for around 14,900 of these cases. Among the U.S. population, about 35% were classified as mild, 15% as moderate, and 50% as severe Hemophilia A. Additionally, around 2,000 cases involved inhibitors, while approximately 12,700 were identified as non-inhibitor cases in 2024.
In 2024, the EU4 and the UK collectively represented the largest share of the Hemophilia A prevalent population, accounting for approximately 58% of the total cases across the seven major markets (7MM). Within these regions, France recorded the highest number of diagnosed cases, followed by the UK, while Spain reported the lowest prevalence of Hemophilia A.
DelveInsight’s report “Hemophilia A Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Hemophilia A landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
The latest healthcare forecast report provides an in-depth analysis of Hemophilia A, offering comprehensive insights into the Hemophilia A revenue trends, prevalence, and treatment landscape. The report delves into key Hemophilia A statistics, highlighting the current and projected market size, while examining the efficacy and development of emerging Hemophilia A therapies. Additionally, we cover the landscape of Hemophilia A clinical trials, providing an overview of ongoing and upcoming studies that are poised to shape the future of Hemophilia A treatment. This report is an essential resource for understanding the market dynamics and the evolving therapeutic options within the Hemophilia A space.
To Know in detail about the Hemophilia A market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Hemophilia A Market Forecast
Some of the key facts of the Hemophilia A Market Report:
- Key Hemophilia A Companies: Takeda, Novo Nordisk, Ultragenyx Pharmaceutical, Bayer, CSL Behring, Pfizer, and others
- Key Hemophilia A Therapies: ROCTAVIAN (valoctocogene roxaparvovec)
- OBIZUR (susoctocog alfa), Mim8, Peboctocogene camaparvovec (DTX201 or BAY2599023), NNC0365-3769 (Mim8) PPX, Kogenate FS, rVIII-SingleChain, Adynovate, PF-07055480, and others
- The epidemiology segment also provides the Hemophilia A epidemiology data and findings across the United States, EU4 (Germany, France, Italy, and Spain) and the United Kingdom, and Japan.
Hemophilia A Overview
Hemophilia A is a rare inherited bleeding disorder caused by a deficiency or absence of clotting factor VIII, which is essential for normal blood clotting. This condition leads to prolonged bleeding, frequent nosebleeds, easy bruising, and internal bleeding, especially in joints and muscles. It primarily affects males and is passed through an X-linked recessive inheritance pattern. Treatment focuses on factor VIII replacement therapy, gene therapy, or non-factor therapies to prevent or control bleeding episodes. Early diagnosis and proper management are vital to reducing complications and improving the quality of life for individuals with Hemophilia A.
Get a Free sample for the Hemophilia A Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/hemophilia-a2030-market
Key Trends in Hemophilia A Therapeutics Market:
- Shift Toward Gene Therapy & Curative Approaches: The market is moving beyond traditional factor replacement to long-lasting gene therapies that aim to provide sustained or potentially lifelong correction of Factor VIII deficiency.
- Growing Adoption of Non-Factor Therapies: Drugs like Hemlibra (emicizumab) are gaining traction due to less frequent dosing, subcutaneous administration, and effectiveness in patients with and without inhibitors, reducing reliance on intravenous factor infusions.
- Advancements in Extended Half-Life (EHL) Products: Next-generation recombinant Factor VIII therapies with prolonged activity are extending dosing intervals, improving patient quality of life, and offering better bleed control.
- Rising Focus on Personalized & Prophylactic Treatment: There is an increasing shift from on-demand treatment to preventive care, supported by tailored therapy based on bleed risk, genetic profile, and patient lifestyle.
- Strong R&D Pipeline & Strategic Collaborations: Major biopharma companies are investing in novel modalities including CRISPR-based therapies, siRNA drugs, and improved gene delivery systems, with multiple partnerships accelerating clinical development and regulatory filings.
Hemophilia A Epidemiology
The Hemophilia A epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2019 to 2032. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Hemophilia A Epidemiology Segmentation:
The Hemophilia A market report proffers epidemiological analysis for the study period 2019–2032 in the 7MM segmented into:
- Total Prevalence of Hemophilia A
- Prevalent Cases of Hemophilia A by severity
- Gender-specific Prevalence of Hemophilia A
- Diagnosed Cases of Episodic and Chronic Hemophilia A
Download the report to understand which factors are driving Hemophilia A epidemiology trends @ Hemophilia A Epidemiology Forecast
Recent Developments in the Hemophilia A Treatment Landscape:
- In May 2025, the FDA approved Jivi (antihemophilic factor [recombinant], PEGylated-aucl; Bayer) for treating pediatric patients aged 7 and older with hemophilia A, as announced by Bayer.
- In May 2025, the FDA approved Antihemophilic factor (recombinant), PEGylated-aucl (Jivi; Bayer) for treating pediatric patients aged 7 and older with hemophilia A. The approval was supported by results from the Alfa-PROTECT and PROTECT Kids trials.
- In April 2025, the FDA approved Qfitlia (fitusiran) for routine prophylaxis to prevent or reduce bleeding episodes in patients aged 12 and older with hemophilia A or B, with or without factor VIII or IX inhibitors.
- In March 2025, Alnylam Pharmaceuticals announced the FDA's approval of Qfitlia™ (fitusiran), the sixth Alnylam-discovered RNAi therapeutic approved in the U.S. It is the first and only treatment to lower antithrombin (AT), aiming to promote thrombin generation, rebalance hemostasis, and prevent bleeds.
Hemophilia A Drugs Uptake and Pipeline Development Activities
The Hemophilia A drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Hemophilia A market or expected to get launched during the study period. The analysis covers Hemophilia A market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Hemophilia A Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Hemophilia A Therapies and Key Companies
- ROCTAVIAN (valoctocogene roxaparvovec)
- OBIZUR (susoctocog alfa): Takeda
- Mim8: Novo Nordisk
- Peboctocogene camaparvovec (DTX201 or BAY2599023): Ultragenyx Pharmaceutical
- NNC0365-3769 (Mim8) PPX: Novo Nordisk
- Kogenate FS: Bayer
- rVIII-SingleChain: CSL Behring
- Adynovate: Takeda
- PF-07055480: Pfizer
Discover more about therapies set to grab major Hemophilia A market share @ Hemophilia A Treatment Landscape
Hemophilia A Market Drivers
- Growing adoption of gene therapy and novel non-factor replacement treatments improving long-term disease management.
- Increased awareness and early diagnosis leading to better treatment access and patient outcomes.
- Rising R&D investments and clinical trials focused on curative and extended half-life therapies.
- Expanding healthcare infrastructure and reimbursement support in developed regions.
- Ongoing technological advancements in drug delivery systems and personalized medicine.
Hemophilia A Market Barriers
- High treatment costs associated with gene and factor replacement therapies.
- Limited access to advanced treatments in low- and middle-income countries.
- Developmental challenges in achieving durable gene therapy outcomes.
- Regulatory hurdles and lengthy approval timelines for novel therapies.
- Risk of immune response or inhibitor development reducing treatment effectiveness.
Scope of the Hemophilia A Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Hemophilia A Companies: Takeda, Novo Nordisk, Ultragenyx Pharmaceutical, Bayer, CSL Behring, Pfizer, and others
- Key Hemophilia A Therapies: ROCTAVIAN (valoctocogene roxaparvovec)
OBIZUR (susoctocog alfa), Mim8, Peboctocogene camaparvovec (DTX201 or BAY2599023), NNC0365-3769 (Mim8) PPX, Kogenate FS, rVIII-SingleChain, Adynovate, PF-07055480, and others - Hemophilia A Therapeutic Assessment: Hemophilia A current marketed and Hemophilia A emerging therapies
- Hemophilia A Market Dynamics: Hemophilia A market drivers and Hemophilia A market barriers
- Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Hemophilia A Unmet Needs, KOL’s views, Analyst’s views, Hemophilia A Market Access and Reimbursement
To know more about Hemophilia A companies working in the treatment market, visit @ Hemophilia A Clinical Trials and Therapeutic Assessment
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Hemophilia A - Pipeline Insight, 2025
Hemophilia A Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across the Hemophilia A market.
Hemophilia A - Epidemiology Forecast - 2034
DelveInsight's Hemophilia A - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of Hemophilia A in...