IgA Nephropathy Market Size was Valued at USD 730 Million in 2024, Set for Strong Growth Through 2034
Get a Sneak Peek at the Latest iga nephropathy market size and forecast Report
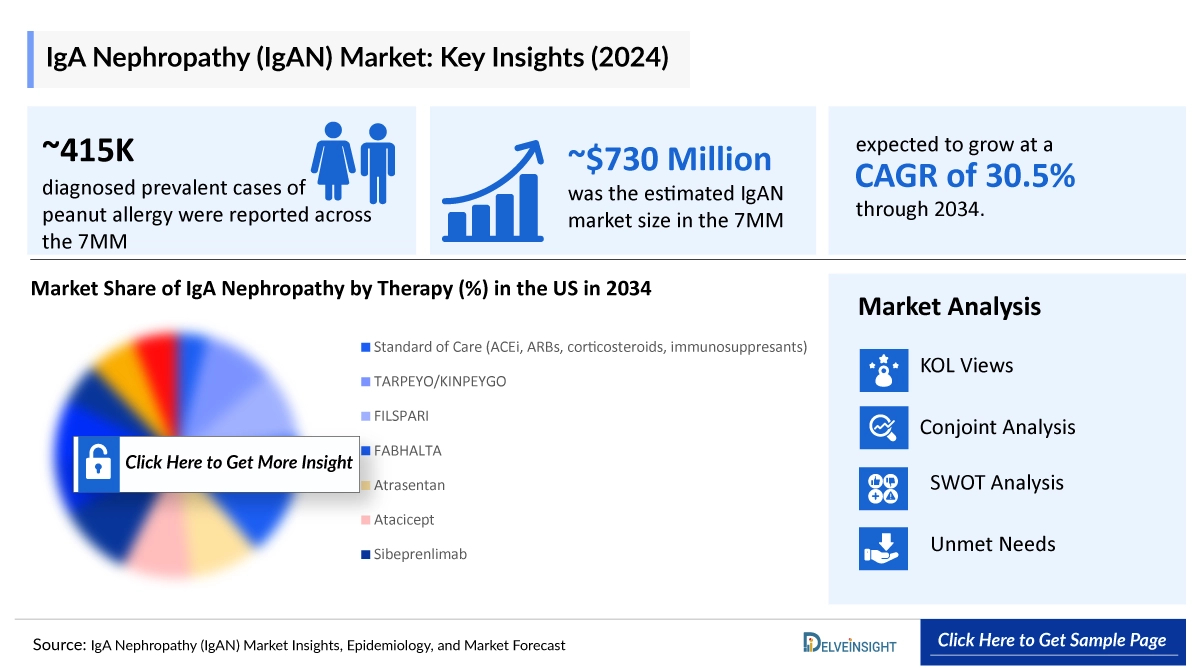
The IgA Nephropathy market size was valued approximately USD 730 million in 2024 and is anticipated to grow with a significant CAGR during the study period (2020-2034). DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the IgA Nephropathy landscape.
In 2024, the US IgA Nephropathy (IgAN) market was valued at approximately USD 455 million and is expected to expand with the introduction of new therapies. The combined market for the EU4 and the UK was estimated at around USD 150 million, accounting for nearly 21% of the total 7MM market revenue, with growth projected through 2034. Within this region, Germany led the market with roughly USD 45 million in revenue, followed by France at about USD 35 million and Italy at nearly USD 30 million. In Japan, the IgAN market was estimated at approximately USD 125 million in 2024, with further growth anticipated over the forecast period from 2025 to 2034.
DelveInsight estimates that approximately 415,000 prevalent cases of IgA Nephropathy (IgAN) were reported across the 7MM in 2024, with diagnosed cases projected to grow at a CAGR of 0.6% by 2034. In 2024, the United States accounted for around 133,000 diagnosed prevalent IgAN cases, a number expected to rise by 2034. Within the EU4 and the UK, Germany reported the highest diagnosed prevalence at approximately 30,000 cases, while Spain had the lowest at about 5,000 cases. Japan recorded the largest number of diagnosed IgAN cases among the 7MM in 2024, with nearly 175,000 cases, which is projected to increase over the forecast period. In France, diagnosed cases in 2024 included roughly 16,000 males and 8,000 females, with both figures expected to grow by 2034.
DelveInsight’s report “IgA Nephropathy Market Insights, Epidemiology, and Market Forecast-2034”, provides a comprehensive analysis of the IgA Nephropathy landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines IgA Nephropathy market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the IgA Nephropathy market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; IgA Nephropathy Market Forecast
Some of the key facts of the IgA Nephropathy Market Report:
- Key IgA Nephropathy Companies: Travere Therapeutics, Asahi Kasei (Calliditas Therapeutics), Vertex Pharmaceuticals, Vera Therapeutics, Visterra, Chinook Therapeutics, Novartis, IgA proteases Selecta Biosciences, Shanghai Alebund Pharmaceuticals, Rohto Pharmaceutical, Wuhan Createrna Science and Technology, Guangdong Hengrui Pharmaceutical, LRx Ionis Pharmaceuticals, Jiangsu HengRui Medicine Co., Ltd., Travere Therapeutics, and others
- Key IgA Nephropathy Therapies: VANRAFIA (atrasentan), FILSPARI (sparsentan), TARPEYO/KINPEYGO (budesonide), Povetacicept (ALPN-303), Atacicept, Zigakibart (FUB523), Sibeprenlimab, Atrasentan, LNP023, Research programme, AP 305, ADR-001, MY 008, SHR-2010, IONIS FB, HR19042, Sparsentan, HR19042, and others
- The IgA Nephropathy epidemiology based on gender analyzed that IgAN is more prominent in males in comparison to females. Prevalence of IgAN occurs in men around two times more than females worldwide.
- The IgA Nephropathy market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage IgA Nephropathy pipeline products will significantly revolutionize the IgA Nephropathy market dynamics.
IgA Nephropathy Overview
IgA Nephropathy (IgAN), also known as Berger’s disease, is a kidney disorder caused by the buildup of immunoglobulin A (IgA) antibodies in the glomeruli, the filtering units of the kidneys. This accumulation leads to inflammation, which can impair kidney function over time. Common symptoms include blood in the urine (hematuria), protein in the urine (proteinuria), swelling (edema), and high blood pressure. IgA nephropathy can progress slowly and, in some cases, may lead to chronic kidney disease or kidney failure.
Get a Free sample for the IgA Nephropathy Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/iga-nephropathy-igan-market
Key Trends in IgA Nephropathy Therapeutics Market:
- Emergence of Targeted Therapies: Development of novel treatments, including complement inhibitors, immune‑modulating agents, and receptor‑targeted drugs, is expanding therapeutic options beyond traditional immunosuppressants.
- Shift Toward Precision Medicine: Increased use of biomarkers and genetic profiling is enabling more personalized treatment strategies tailored to disease severity and patient response.
- Greater Focus on Slowing Disease Progression: Clinical efforts are emphasizing therapies that reduce proteinuria and preserve kidney function to delay or prevent progression to end‑stage renal disease (ESRD).
- Improved Diagnostic and Monitoring Tools: Advances in non‑invasive diagnostic methods, like novel biomarkers and imaging techniques, support earlier detection and better disease monitoring.
- Increased Clinical Trial Activity: A growing pipeline of Phase II and III studies reflects heightened R&D investment, along with regulatory incentives aimed at accelerating approval of promising IgAN therapies.
IgA Nephropathy Epidemiology
The IgA Nephropathy epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
IgA Nephropathy Epidemiology Segmentation:
The IgA Nephropathy market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalence of IgA Nephropathy
- Prevalent Cases of IgA Nephropathy by severity
- Gender-specific Prevalence of IgA Nephropathy
- Diagnosed Cases of Episodic and Chronic IgA Nephropathy
Download the report to understand which factors are driving IgA Nephropathy epidemiology trends @ IgA Nephropathy Epidemiology Forecast
Recent Development In The IgA Nephropathy Treatment Landscape:
- In November 2025, The FDA has granted approval to sibeprenlimab-szsi (Voyxact; Otsuka) for lowering proteinuria in adults with immunoglobulin A nephropathy (IgAN), supported by interim results from the phase 3 VISIONARY trial (NCT05248646). The therapy is administered as a subcutaneous self-injection using a prefilled syringe every four weeks at home, with a recommended dose of 400 mg per administration.
- In November 2025, Voyxact (sibeprenlimab-szsi) is a newly FDA-approved treatment designed to reduce proteinuria in adults with primary immunoglobulin A nephropathy (IgAN) who are at risk of disease progression. IgAN is a chronic kidney disorder caused by the accumulation of IgA antibodies in the kidneys, triggering inflammation and tissue damage. This process results in elevated protein levels in the urine and a progressive loss of kidney function. Without appropriate treatment, IgAN can ultimately progress to kidney failure.
- In September 2025, Vertex Pharmaceuticals Incorporated (Nasdaq: VRTX) announced notable progress across its programs for immunoglobulin A Nephropathy (IgAN), APOL1-mediated kidney disease (AMKD), and autosomal dominant polycystic kidney disease (ADPKD). These developments mark important strides toward the company’s mission of delivering first-in-class or best-in-class therapies that address the root causes of these serious kidney conditions.
- In September 2025, CSL Vifor and Travere Therapeutics, Inc. (NASDAQ: TVTX) have expressed support for the newly published KDIGO 2025 clinical practice guideline for managing IgA Nephropathy (IgAN) and Immunoglobulin A vasculitis. The updated guidelines outline diagnostic criteria, treatment objectives, and management strategies, aiming to advance care for patients with IgAN and provide guidance for the clinicians treating them.
- In May 2025, Otsuka Pharmaceuticals recently announced that the FDA has accepted the Biologics License Application (BLA) for sibeprenlimab, an APRIL (a proliferation-inducing ligand) inhibitor being developed for the treatment of immunoglobulin A nephropathy (IgAN). This follows the FDA’s 2024 Breakthrough Therapy Designation granted to sibeprenlimab for the same condition. The BLA submission is backed by data from the Phase III VISIONARY trial. According to GlobalData, the priority review underscores sibeprenlimab’s potential advantages over current therapies and reinforces its promise as a treatment for the complex nature of IgAN. If approved, it would become the first disease-modifying option addressing both clinical and practical aspects of managing IgAN.
- In March 2025, Arrowhead Pharmaceuticals, Inc. (NASDAQ: ARWR) has announced topline findings from Part 2 of its Phase 1/2 clinical trial evaluating ARO-C3, an investigational RNA interference (RNAi) therapy aimed at lowering liver production of complement component 3 (C3) to potentially treat multiple complement-mediated disorders.
- In November 2024, Otsuka Pharmaceutical Development & Commercialization, Inc. (OPDC) and Otsuka Pharmaceutical Co., Ltd. have announced plans to submit a Biologics License Application (BLA) in the U.S. for sibeprenlimab, an investigational therapy for adult patients with immunoglobulin A nephropathy (IgA nephropathy), in the first half of 2025. This decision follows a recent meeting with the U.S. FDA to review the positive interim findings from the Phase 3 VISIONARY trial (NCT05248646).
- In January 2024, Vera Therapeutics, Inc. (Nasdaq: VERA), a biotechnology company in the advanced stages of clinical development focused on innovative treatments for individuals with severe immunologic conditions, announced today the addition of two seasoned industry professionals to spearhead the advancement of the company's drug initiatives. This includes their late-stage candidate, atacicept, presently undergoing a Phase 3 clinical trial for IgA nephropathy (IgAN). Effective immediately, Robert M. Brenner, M.D., assumes the role of Chief Medical Officer, succeeding Dr. Celia Lin, M.D., while William D. Turner takes on the position of Chief Development Officer.
IgA Nephropathy Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the IgA Nephropathy market or expected to get launched during the study period. The analysis covers IgA Nephropathy market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the IgA Nephropathy Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
IgA Nephropathy Therapies and Key Companies
- VANRAFIA (atrasentan): Novartis
- FILSPARI (sparsentan): Travere Therapeutics
- TARPEYO/KINPEYGO (budesonide): Asahi Kasei (Calliditas Therapeutics)
- Povetacicept (ALPN-303): Vertex Pharmaceuticals
- Atacicept: Vera Therapeutics
- Zigakibart (FUB523): Novartis
- Sibeprenlimab: Visterra
- Atrasentan: Chinook Therapeutics
- LNP023: Novartis Pharmaceuticals
- Research programme: IgA proteases Selecta Biosciences
- AP 305: Shanghai Alebund Pharmaceuticals
- ADR-001: Rohto Pharmaceutical
- MY 008: Wuhan Createrna Science and Technology
- SHR-2010: Guangdong Hengrui Pharmaceutical
- IONIS FB: LRx Ionis Pharmaceuticals
- HR19042: Jiangsu HengRui Medicine Co., Ltd.
- Sparsentan: Travere Therapeutics
- HR19042: Jiangsu HengRui Medicine Co., Ltd.
Discover more about therapies set to grab major IgA Nephropathy market share @ IgA Nephropathy Treatment Landscape
IgA Nephropathy Market Drivers
- Rising Prevalence of IgAN: Increasing cases of kidney disorders and chronic kidney disease are driving market demand.
- Advancements in Therapeutics: Development of novel targeted therapies, including immunosuppressants and complement inhibitors, is expanding treatment options.
- Improved Diagnostic Tools: Enhanced kidney biopsy techniques and biomarker-based diagnostics enable earlier and more accurate diagnosis.
- Growing Awareness and Screening: Increased physician and patient awareness supports timely diagnosis and management of IgAN.
- Supportive R&D and Regulatory Environment: Investment in clinical research and expedited approval pathways encourage development of new therapies.
IgA Nephropathy Market Barriers
- Limited Approved Therapies: Few FDA-approved treatments are available, creating unmet clinical needs.
- High Treatment Costs: Specialized therapies and long-term management may restrict patient access, particularly in emerging markets.
- Chronic and Progressive Nature: Slow progression and asymptomatic early stages complicate timely diagnosis and treatment.
- Safety and Side Effect Concerns: Long-term use of immunosuppressants can lead to adverse effects, affecting compliance.
- Market Fragmentation: Variability in healthcare infrastructure and treatment availability across regions limits uniform market growth.
Scope of the IgA Nephropathy Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key IgA Nephropathy Companies: Travere Therapeutics, Asahi Kasei (Calliditas Therapeutics), Vertex Pharmaceuticals, Vera Therapeutics, Visterra, Chinook Therapeutics, Novartis, IgA proteases Selecta Biosciences, Shanghai Alebund Pharmaceuticals, Rohto Pharmaceutical, Wuhan Createrna Science and Technology, Guangdong Hengrui Pharmaceutical, LRx Ionis Pharmaceuticals, Jiangsu HengRui Medicine Co., Ltd., Travere Therapeutics, and others
- Key IgA Nephropathy Therapies: VANRAFIA (atrasentan), FILSPARI (sparsentan), TARPEYO/KINPEYGO (budesonide), Povetacicept (ALPN-303), Atacicept, Zigakibart (FUB523), Sibeprenlimab, Atrasentan, LNP023, Research programme, AP 305, ADR-001, MY 008, SHR-2010, IONIS FB, HR19042, Sparsentan, HR19042, and others
- IgA Nephropathy Therapeutic Assessment: IgA Nephropathy current marketed and IgA Nephropathy emerging therapies
- IgA Nephropathy Market Dynamics: IgA Nephropathy market drivers and IgA Nephropathy market barriers
- IgA Nephropathy Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- IgA Nephropathy Unmet Needs, KOL’s views, Analyst’s views, IgA Nephropathy Market Access and Reimbursement
- To know more about IgA Nephropathy companies working in the treatment market, visit @ IgA Nephropathy Clinical Trials and Therapeutic Assessment
To know more about IgA Nephropathy companies working in the treatment market, visit @ IgA Nephropathy Clinical Trials and Therapeutic Assessment
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
IgA Nephropathy (IgAN) - Epidemiology Forecast to 2034
DelveInsight's IgA Nephropathy (IgAN) - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology..
IgA Nephropathy (IgAN) - Pipeline Insight, 2025
IgA Nephropathy (IgAN) Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across the IgA Nephropat..