Multiple System Atrophy Market Expected to Reach USD 1,753 million in 2034
Get a Sneak Peek at the Latest multiple system atrophy market size Report
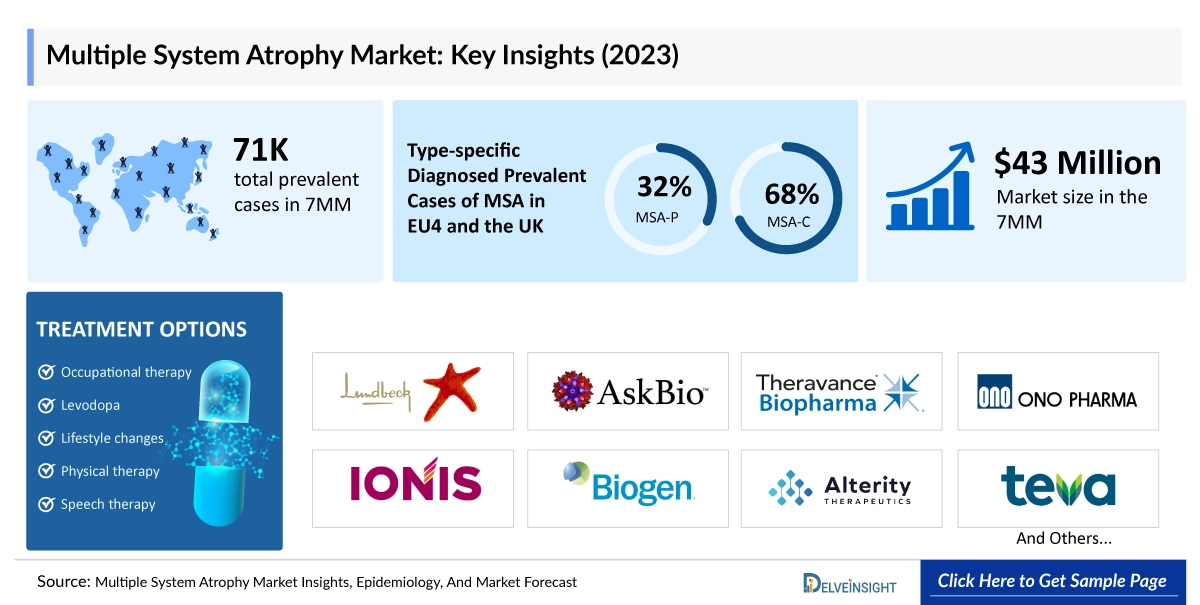
The Multiple System Atrophy market across the seven major markets (7MM) is undergoing significant expansion. Valued at approximately USD 44 million in 2023–2025, the market is forecasted to reach USD 1,753 million by 2034, demonstrating an impressive CAGR of 50.60%. The United States remains the largest contributor, representing around 60% of the total prevalent patient population in 2023. EU4 and the UK collectively account for 16%, while Japan contributes 24%.
Despite the growing prevalence of about 70,800 cases in 2023, there are currently no approved disease-modifying therapies for MSA. Existing treatment options mainly focus on symptom management through drugs such as levodopa, amantadine, droxidopa, adrenergic receptor agonists, and other off-label medications.
Future market growth is expected to be driven by the anticipated launch of innovative therapeutic candidates like Amlenetug (Lu AF82422), Ampreloxetine ATH434, TAK-341/MEDI1341, and Emrusolmin. However, challenges persist, including diagnostic complexity, rapid disease progression, high morbidity, and the significant burden placed on patients and caregivers. Overall, the Multiple System Atrophy market is positioned for strong, sustained growth through 2034, supported by advancements in clinical research and emerging treatment pipelines.
DelveInsight’s report, “Multiple System Atrophy Market Insights, Epidemiology, and Market Forecast-2034” provides a comprehensive analysis of the Multiple System Atrophy landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Multiple System Atrophy market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
Some of the key facts of the Multiple System Atrophy Market Report:
- The leading Multiple System Atrophy Companies developing therapies include H Lundbeck A/S, Brain Neurotherapy Bio, Inc., Asklepios BioPharmaceutical, Inc., Theravance Biopharma, Ono Pharmaceutical Co. Ltd, Ionis Pharmaceuticals, Inc., Biogen, Alterity Therapeutics, Teva Pharmaceutical, MODAG GmbH, and others.
- Potential future therapies for Multiple System Atrophy include Lu AF82422 (H. Lundbeck A/S/Genmab), Ampreloxetine (Theravance Biopharma), Emrusolmin (Teva Pharmaceutical/MODAG GmbH), and others.
- The future of Multiple System Atrophy treatment holds promise, driven by ongoing research and the development of innovative therapies. Current management strategies, such as symptomatic medications, physical therapy, and supportive care, remain essential.
Multiple System Atrophy Overview
Multiple System Atrophy is a rare, rapidly progressive neurodegenerative disorder that affects the autonomic nervous system and motor control. It is characterized by a combination of symptoms similar to Parkinson’s disease, cerebellar ataxia, and autonomic dysfunction. MSA is categorized based on the predominant clinical presentation:
- MSA-P (Parkinsonian type): Symptoms closely resemble Parkinson’s disease, including muscle rigidity, slowness of movement, and tremors.
- MSA-C (Cerebellar type): Dominated by coordination and balance impairments, including gait disturbances and difficulty with fine motor tasks.
Multiple System Atrophy Market Drivers
- Rising disease awareness and improved diagnosis rates
- High unmet medical need due to lack of disease-modifying therapies
- Emerging therapeutic pipeline (e.g., Amlenetug, Ampreloxetine, TAK-341/MEDI1341, Emrusolmin)
- Increasing prevalence of neurodegenerative disorders, especially in aging populations
- Supportive regulatory environment and orphan drug incentives
- Growing focus on symptom management and quality of life improvement
- Significant investment in clinical research and industry collaborations
Recent Developments in Multiple System Atrophy Clinical Trials
- In September 2025, Teva Pharmaceutical (NYSE:TEVA) announced that its investigational MSA treatment, emrusolmin, received Fast Track designation from the U.S. FDA. The drug is currently in Phase 2 development for Multiple System Atrophy.
- In August 2025, Tiziana Life Sciences received FDA clearance of its IND for a Phase 2a trial of intranasal foralumab in patients with Multiple System Atrophy (MSA), a rare neurodegenerative disease with no approved therapies. The six-month open-label study will evaluate microglial activation, clinical outcomes, and safety of the fully human anti-CD3 monoclonal antibody delivered via nasal spray.
- In July 2025, NKGen Biotech received FDA authorization for an Expanded Access Program (EAP) to use its NK cell therapy, troculeucel, in multiple neurodegenerative diseases—including Alzheimer’s, Parkinson’s, ALS, MSA, PSP, FTD, CBD, MS, and Lewy Body Dementia—beyond its current Phase 2a trial in moderate-stage Alzheimer’s disease.
- In May 2025, Alterity Therapeutics announced that the FDA has granted Fast Track designation for ATH434, its investigational treatment for Multiple System Atrophy (MSA). This designation aims to expedite the development and review of ATH434, recognizing its potential to address the significant unmet need in MSA, a condition currently without an approved therapy.
- In February 2025, Alterity Therapeutics’ ATH434 showed promising Phase 2 results in a randomized, double-blind trial for early-stage multiple system atrophy (Multiple System Atrophy). The treatment, particularly at a 50 mg dose, was well-tolerated and demonstrated both clinical and biomarker-based benefits.
Multiple System Atrophy Epidemiology
- United States: Highest prevalence with ~42,500 cases in 2023; projected to grow at a CAGR of 1.0% by 2034. Distribution by disease stage: Stage 0 (~400), Stage 1 (~800), Stage 2 (~2,100), Stage 3 (~15,000), Stage 4 (~13,000), Stage 5 (~10,000).
- Europe (EU4 + UK): Germany leads with ~2,700 cases, followed by the UK (~2,400 cases); Spain has the lowest (~1,500 cases).
- Japan: Total ~17,100 cases in 2023 (males ~5,100; females ~12,000), projected to rise by 2034. MSA-C is more common (~70%) than MSA-P (~30%). Symptomatic neurogenic orthostatic hypotension (nOH) cases: ~13,700, expected to reach ~14,100 by 2034.
- Age-specific prevalence: In Germany, the 70+ age group has the highest number (~1,400 cases) and is expected to increase to ~1,500 cases by 2034.
Multiple System Atrophy Epidemiology Segmentation
- Diagnosed Prevalent Cases of Multiple System Atrophy
- Gender-specific Diagnosed Prevalent Cases of Multiple System Atrophy
- Age-specific Diagnosed Prevalent Cases of Multiple System Atrophy
- Type-specific Diagnosed Prevalent Cases of Multiple System Atrophy
- Stage-specific Diagnosed Prevalent Cases of Multiple System Atrophy
- nOH Symptomatic Cases of Multiple System Atrophy
Multiple System Atrophy Therapies and Key Companies
- Amlenetug (Lu AF82422): H Lundbeck A/S/Genmab
- Ampreloxetine (TD-9855): Theravance Biopharma
- Emrusolmin (TEV-56286, Anle-138b): Teva Pharmaceutical/MODAG GmBH
- AAV2-GDNF Gene Therapy: Brain Neurotherapy Bio, Inc./ Asklepios BioPharmaceutical, Inc.
- ONO-2808: Ono Pharmaceutical Co. Ltd
- ION464: Ionis Pharmaceuticals, Inc. /Biogen
- ATH434: Alterity Therapeutics
- Emrusolmin: Teva Pharmaceutical/ MODAG GmbH
Multiple System Atrophy Market Barriers
- Lack of disease-modifying therapies: No approved treatments to slow neurodegeneration, limiting market potential.
- Diagnostic challenges: Complex symptoms and overlap with Parkinson’s disease hinder early and accurate diagnosis.
- Rapid disease progression: Fast worsening of symptoms leads to short treatment windows and poor patient outcomes.
- Small patient population: Being a rare disease, the limited number of patients reduces market size and commercial attractiveness.
- High clinical trial complexity: Recruiting patients and designing trials for MSA is challenging due to variability in symptoms and stages.
- High treatment costs: Symptom management and supportive care can be expensive, affecting accessibility.
- Limited awareness among caregivers and physicians: Insufficient knowledge can delay diagnosis and treatment initiation.
Scope of the Multiple System Atrophy Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Multiple System Atrophy Companies: AbbVie, AstraZeneca and Daiichi Sankyo, Shanghai Henlius Biotech, Moderna Therapeutics/ Merck, BeyondSpring, Cullinan Oncology/ Taiho Pharma, Genentech/Roche, Betta Pharma/ Xcovery, Pfizer, Takeda Pharmaceuticals, AstraZeneca, Janssen, Boehringer Ingelheim, Pfizer, Merck, Bristol-Myers Squibb/Ono Pharmaceutical, Genentech/ Hoffmann-La Roche, AstraZeneca, Bristol-Myers Squibb, AstraZeneca/ Daiichi Sankyo, Amgen, Blueprint Medicines/Rigel Pharmaceuticals, and others
- Key Multiple System Atrophy Therapies: KEYTRUDA (pembrolizumab), Telisotuzumab vedotin, Datopotamab deruxtecan (Dato-DXd), Serplulimab (HLX10), V940 (mRNA-4157) + Pembrolizumab, Plinabulin + Docetaxel, Zipalertinib, TECENTRIQ (atezolizumab), ENSACOVE (ensartinib), BRAFTOVI + MEKTOVI, ALUNBRIG, TAGRISSO (osimertinib), RYBREVANT (amivantamab), GILOTRIF/GIOTRIF (afatinib maleate), VIZIMPRO (dacomitinib), KEYTRUDA (pembrolizumab), OPDIVO (nivolumab), TECENTRIQ (atezolizumab), IMFINZI (durvalumab), AUGTYRO (repotrectinib), ENHERTU (trastuzumab deruxtecan), LUMAKRAS/ LUMYKRAS (sotorasib), GAVRETO (pralsetinib), and others
- Multiple System Atrophy Therapeutic Assessment: Multiple System Atrophy current marketed and Multiple System Atrophy emerging therapies
- Multiple System Atrophy Market Dynamics: Multiple System Atrophy market drivers and Multiple System Atrophy market barriers
- Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Multiple System Atrophy Unmet Needs, KOL’s views, Analyst’s views, Multiple System Atrophy Market Access and Reimbursement
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Multiple System Atrophy (MSA) - Pipeline Insight, 2025
Multiple System Atrophy Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across the market.
Multiple System Atrophy Market Insight, Epidemiology And Market Forecast - 2034
DelveInsight's Multiple System Atrophy (MSA) Market Insights, Epidemiology, and Market Forecast-2034" report delivers an in-depth understanding of the Multiple System Atrophy..
Multiple System Atrophy (MSA) - Epidemiology Forecast - 2034
DelveInsight's Multiple System Atrophy (MSA) - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted

-Market.png&w=256&q=75)

