Prurigo Nodularis market size is projected to grow at a CAGR of 12.9% by 2034
Get a Sneak Peek at the Latest prurigo nodularis market size and forecast Report
In 2025, the Prurigo Nodularis market size across the 7MM was valued at around USD 1,488 million and is projected to expand at a CAGR of 12.9%, reaching approximately USD 4,433 million by 2034 over the forecast period. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Prurigo Nodularis landscape.
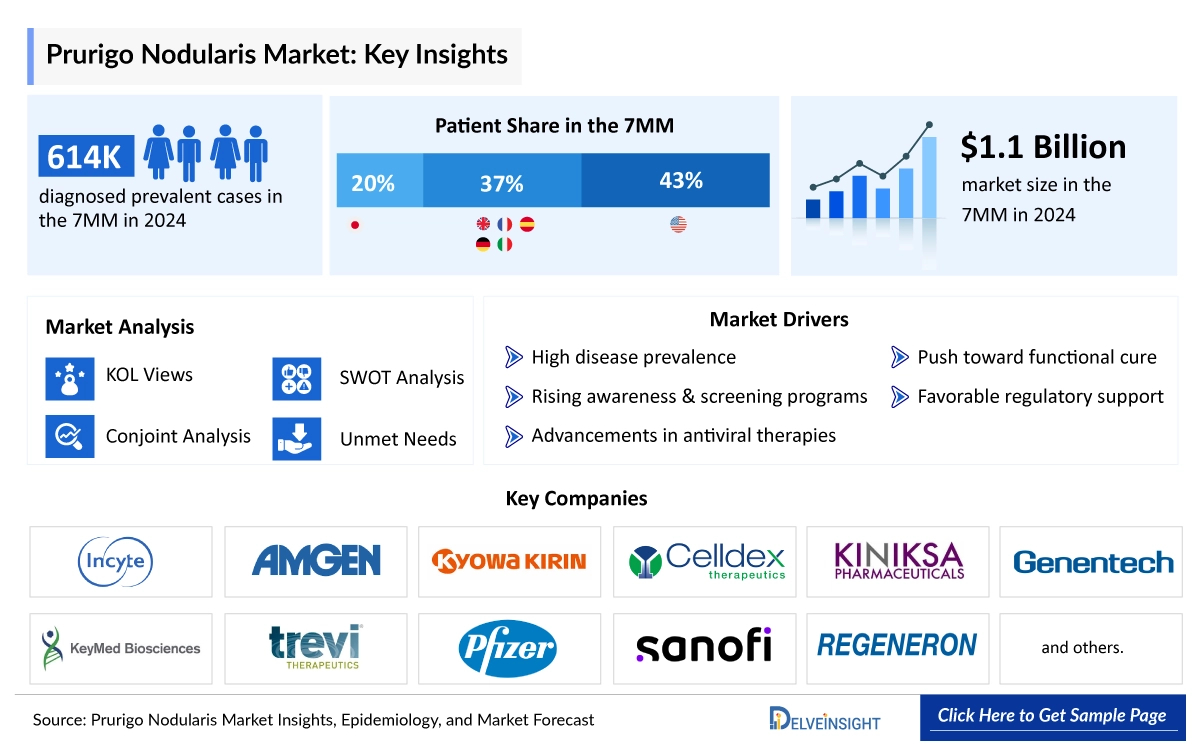
In 2024, the United States represented the largest share of the Prurigo Nodularis market among the 7MM, contributing nearly 75% of the total market value. Within the 7MM, the United States held the largest Prurigo Nodularis market share, valued at approximately USD 810 million in 2024.
The total diagnosed prevalent cases of prurigo nodularis in the 7MM were ~614,000 in 2024, out of which the highest prevalent cases of this disease were in the United States. The total diagnosed prevalent cases of prurigo nodularis in the US were around 261,900 cases in 2024. The US contributed to the largest diagnosed prevalent population of prurigo nodularis, acquiring ~43% of the 7MM in 2024. Whereas EU4 and the UK, and Japan accounted for around 37% and 20% of the total population share, respectively, in 2024.
According to DelveInsight estimates, in 2024, among the age-specific diagnosed prevalent cases of prurigo nodularis in the US, the highest number of cases were found in the =65 years age group (~124,400), followed by 25–44 year age group while the lowest number of cases was observed in <15 years age group. The diagnosed prevalent cases of prurigo nodularis were distributed on the basis of severity; mild, moderate, and severe. In 2024, severe cases (~137,850) were most prevalent followed by moderate and mild cases in the US.
In Japan, the diagnosed prevalent cases of prurigo nodularis were more in male; ~70,100 then in females in 2024.
DelveInsight’s report “Prurigo Nodularis Market Insights, Epidemiology, and Market Forecast-2034” provides a comprehensive analysis of the Prurigo Nodularis landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
To Know in detail about the Prurigo Nodularis market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Prurigo Nodularis Market Forecast
Some of the key facts of the Prurigo Nodularis Market Report:
- Key Prurigo Nodularis Companies: Trevi Therapeutics, Galderma, Genentech, Incyte Corporation, Vyne Therapeutics Inc., Celgene Corporation, Novartis, Pfizer, Regeneron Pharma, and others
- Key Prurigo Nodularis Therapies: Nalbuphine ER, Nemolizumab, Vixarelimab(KPL-716), Povorcitinib(INCB-54707), Ruxolitinib Cream, INCB054707, serlopitant, CC-10004, Pimecrolimus, Abrocitinib, Dupilumab SAR231893, and others
- The Prurigo Nodularis epidemiology based on gender analyzed that females are affected more as compared to males, in the case of Prurigo Nodularis
- The Prurigo Nodularis market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Prurigo Nodularis pipeline products will significantly revolutionize the Prurigo Nodularis market dynamics.
Prurigo Nodularis Overview
Prurigo Nodularis is a chronic inflammatory skin disorder characterized by the formation of intensely itchy, firm nodules on the skin. The condition often results from persistent scratching or rubbing, which worsens the lesions and creates a cycle of itch and irritation. Although the exact cause is not fully understood, PN is associated with immune system dysregulation, nerve hypersensitivity, and underlying conditions such as atopic dermatitis or liver and kidney diseases. The chronic itch severely impacts patients’ quality of life, sleep, and mental health. Treatment typically includes corticosteroids, antihistamines, and emerging biologic therapies targeting key inflammatory pathways.
Get a Free sample for the Prurigo Nodularis Market Report: https://www.delveinsight.com/report-store/prurigo-nodularis-market
Key Trends in Prurigo Nodularis Therapeutics Market:
- Emergence of Targeted Biologic Therapies: The approval and development of biologics like dupilumab and nemolizumab are revolutionizing treatment by addressing the underlying inflammatory pathways.
- Strong R&D Pipeline: Increasing investment in clinical research focusing on novel immunomodulators and neurokinin receptor antagonists is expanding future treatment options.
- Rising Disease Awareness and Diagnosis Rates: Improved understanding of PN among dermatologists and patients is leading to earlier diagnosis and increased treatment demand.
- Unmet Need Driving Innovation: Limited availability of effective therapies continues to encourage pharmaceutical companies to explore new mechanisms of action.
Market Expansion and Regional Growth: The US and Europe are witnessing rapid market growth driven by recent approvals, reimbursement support, and growing clinical adoption of advanced therapies.
Prurigo Nodularis Epidemiology
The Prurigo Nodularis epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Prurigo Nodularis Epidemiology Segmentation:
The Prurigo Nodularis market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalent Cases of Prurigo Nodularis in the 7MM
- Total Diagnosed Prevalent Cases of Prurigo nodularis in the 7MM
- Gender-specific Diagnosed Prevalent Cases of Prurigo nodularis in the 7MM
- Age-specific Diagnosed Prevalence Cases of Prurigo nodularis in the 7MM
- Severity-specific Diagnosed Prevalent Cases of Prurigo nodularis in the 7MM
Download the report to understand which factors are driving Prurigo Nodularis epidemiology trends @ Prurigo Nodularis Epidemiology Forecast
Recent Developments In The Prurigo Nodularis Treatment Landscape:
- In June 2025, Galderma has released new interim data from the OLYMPIA long-term extension study evaluating its monoclonal antibody, Nemluvio, for treating moderate-to-severe prurigo nodularis a chronic skin disorder characterized by thick nodules and severe itching. The findings show that Nemluvio is well-tolerated and delivers sustained relief from symptoms such as skin lesions and itch for up to two years. The OLYMPIA trial, which includes 508 participants from the earlier Phase II and Phase III OLYMPIA 1 and 2 studies, is designed to assess the long-term efficacy and safety of Nemluvio.
- In March 2025, Incyte (Nasdaq: INCY) announced findings from its pivotal Phase 3 TRuE-PN clinical trial program, including the TRuE-PN1 and TRuE-PN2 studies, assessing the safety and efficacy of twice-daily ruxolitinib cream 1.5% (Opzelura®), a topical JAK1/2 inhibitor, in adult patients (18 years and older) with prurigo nodularis (PN).
- In March 2025, Incyte (Nasdaq: INCY) announced positive topline results from its pivotal Phase 3 STOP-HS trial assessing the safety and efficacy of povorcitinib (INCB054707), an oral JAK1 inhibitor, in adults (≥18 years) with moderate to severe hidradenitis suppurativa (HS). Povorcitinib, a selective JAK1 inhibitor, is also being investigated in Phase 3 trials for vitiligo and prurigo nodularis (PN) and in Phase 2 trials for asthma and chronic spontaneous urticaria (CSU).
- In February 2025, Galderma (SWX:GALD) announced that the European Commission has granted approval for Nemluvio in the EU to treat both moderate-to-severe atopic dermatitis and prurigo nodularis. Nemluvio is now authorized for subcutaneous use in patients aged 12 and older with moderate-to-severe atopic dermatitis requiring systemic therapy, as well as in adults with moderate-to-severe prurigo nodularis eligible for systemic treatment.
Prurigo Nodularis Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Prurigo Nodularis market or expected to get launched during the study period. The analysis covers Prurigo Nodularis market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Prurigo Nodularis Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Prurigo Nodularis Therapies and Key Companies
- Nalbuphine ER: Trevi Therapeutics
- Nemolizumab: Galderma
- Vixarelimab(KPL-716): Genentech
- Povorcitinib(INCB-54707): Incyte Corporation
- Ruxolitinib Cream: Incyte Corporation
- INCB054707: Incyte Corporation
- serlopitant: Vyne Therapeutics Inc.
- CC-10004: Celgene Corporation
- Pimecrolimus: Novartis
- Abrocitinib: Pfizer
- Dupilumab SAR231893: Regeneron Pharma
Discover more about therapies set to grab major Prurigo Nodularis market share @ Prurigo Nodularis Treatment Market
Prurigo Nodularis Market Drivers
- Rising Prurigo Nodularis awareness among healthcare providers and patients leading to earlier diagnosis and treatment.
- Advancements in biologic therapies targeting specific inflammatory pathways, improving treatment outcomes.
- Increasing prevalence of chronic skin conditions, including atopic dermatitis, contributing to higher Prurigo Nodularis incidence.
- Strong R&D pipeline with multiple promising candidates under clinical development by major pharmaceutical companies.
- Supportive regulatory environment encouraging innovation and faster drug approvals for rare dermatological disorders.
Prurigo Nodularis Market Barriers
- Limited understanding of disease pathophysiology, hindering the development of highly targeted therapies.
- High treatment costs and restricted access to advanced biologics in low- and middle-income regions.
- Delayed diagnosis and underreporting, especially in patients with overlapping dermatological conditions.
- Adverse effects and limited efficacy of existing treatment options reducing patient adherence.
- Lack of long-term safety data for novel therapies, leading to cautious physician adoption.
Scope of the Prurigo Nodularis Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Prurigo Nodularis Companies: Trevi Therapeutics, Galderma, Genentech, Incyte Corporation, Vyne Therapeutics Inc., Celgene Corporation, Novartis, Pfizer, Regeneron Pharma, and others
- Key Prurigo Nodularis Therapies: Nalbuphine ER, Nemolizumab, Vixarelimab(KPL-716), Povorcitinib(INCB-54707), Ruxolitinib Cream, INCB054707, serlopitant, CC-10004, Pimecrolimus, Abrocitinib, Dupilumab SAR231893, and others
- Prurigo Nodularis Therapeutic Assessment: Prurigo Nodularis current marketed and Prurigo Nodularis emerging therapies
- Prurigo Nodularis Market Dynamics: Prurigo Nodularis market drivers and Prurigo Nodularis market barriers
- Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Prurigo Nodularis Unmet Needs, KOL’s views, Analyst’s views, Prurigo Nodularis Market Access and Reimbursement
To know more about Prurigo Nodularis companies working in the treatment market, visit @ Prurigo Nodularis Clinical Trials and Therapeutic Assessment
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Prurigo Nodularis - Pipeline Insight, 2025
Prurigo Nodularis Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across..
Prurigo Nodularis - Epidemiology Forecast - 2034
DelveInsight's Prurigo Nodularis - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of Prurigo Nodularis