Pulmonary Arterial Hypertension Market Estimated at USD 6,971M in 2025, Projected CAGR 6.3% (2024–2034)
Get a Sneak Peek at the Latest pulmonary arterial hypertension market size and forecast Report
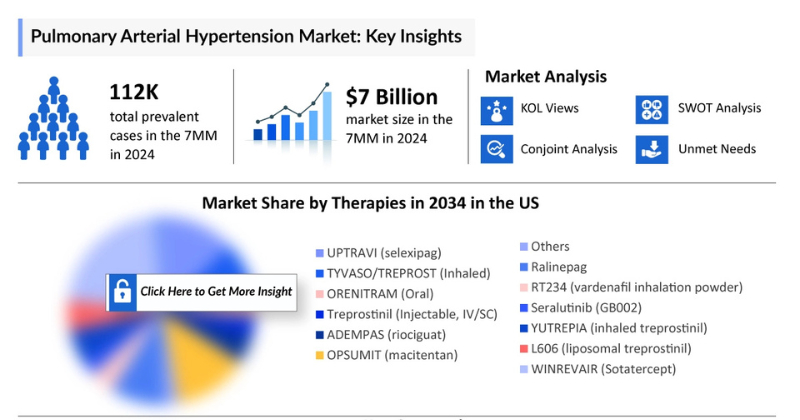
The Pulmonary Arterial Hypertension (PAH) market size across the seven major markets (7MM) was estimated at around USD 6,971 million in 2025 and is expected to expand at a CAGR of 6.3% between 2024 and 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Pulmonary Arterial Hypertension landscape.
As per DelveInsight’s estimates, the US held the largest Pulmonary Arterial Hypertension (PAH) market among the 7MM, valued at approximately USD 6,000 million in 2024. As per DelveInsight’s estimates, Germany led the Pulmonary Arterial Hypertension (PAH) market within the EU4 and UK, valued at around USD 80 million in 2024. France followed with an estimated market size of USD 70 million, while Spain reported the smallest PAH treatment market at approximately USD 45 million during the same year.
Several new therapies are expected to enter the Pulmonary Arterial Hypertension (PAH) market during the forecast period (2025–2034), including ralinepag (United Therapeutics), RT234 (vardenafil inhalation powder) (Respira Therapeutics), Seralutinib (GB002) (Gossamer Bio), YUTREPIA (treprostinil) (Liquidia Technologies), and L606 (liposomal treprostinil) (Pharmosa Biopharm/Liquidia).
According to DelveInsight’s estimates, the total prevalence of Pulmonary Arterial Hypertension (PAH) across the 7MM was approximately 112,700 cases in 2024, with projections indicating an increase by 2034. The United States accounted for the largest share of diagnosed PAH cases, representing about 45% of the total patient population. In 2024, Japan recorded the second-highest number of diagnosed cases, with around 23,000 patients. The EU4 and UK collectively reported approximately 26,000 treated PAH cases in 2024, with numbers expected to grow over the forecast period.
DelveInsight’s report “Pulmonary Arterial Hypertension Market Insights, Epidemiology, and Market Forecast-2034” provides a comprehensive analysis of the Pulmonary Arterial Hypertension landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Pulmonary Arterial Hypertension market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Pulmonary Arterial Hypertension market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Pulmonary Arterial Hypertension Market Forecast
Some of the key facts of the Pulmonary Arterial Hypertension Market Report:
- Key Pulmonary Arterial Hypertension Companies: Aerovate Therapeutics, Novartis, Keros Therapeutics, Vigonvita Life Sciences, Insmed Incorporated, Chugai Pharmaceutical, Merck Sharp & Dohme, Janssen Pharmaceutical, Actelion, Gossamer Bio, Lung Biotechnology PBC, United Therapeutics, Gilead Science, Acceleron Pharma, Eiger BioPharmaceuticals, AstraZeneca, and others.
- Key Pulmonary Arterial Hypertension Therapies: AV-101, LTP001, KER-012, TPN171H, Treprostinil Palmitil, Satralizumab (Genetical Recombination), Sotatercept, Macitentan, ACT-293987, GB002 (seralutinib), Parenteral Treprostinil, Ralinepag, Selonsertib, Sotatercept, ubenimex, Olaparib, and others
- The Pulmonary Arterial Hypertension epidemiology based on gender analyzed that females are more affected with Pulmonary Arterial Hypertension than males
- The Pulmonary Arterial Hypertension market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Pulmonary Arterial Hypertension pipeline products will significantly revolutionize the Pulmonary Arterial Hypertension market dynamics.
Pulmonary Arterial Hypertension Overview
Pulmonary Arterial Hypertension (PAH) is a rare, progressive disorder characterized by high blood pressure in the arteries that carry blood from the heart to the lungs. This increased pressure strains the right side of the heart, potentially leading to heart failure. Symptoms include shortness of breath, fatigue, chest pain, and swelling in the legs or ankles. PAH can be idiopathic, hereditary, or associated with other conditions such as connective tissue diseases or congenital heart defects. Treatment focuses on medications that dilate blood vessels, improve heart function, and slow disease progression, as there is currently no cure.
Get a Free sample for the Pulmonary Arterial Hypertension Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/pulmonary-arterial-hypertension-market
Key Trends in Pulmonary Arterial Hypertension Therapeutics Market:
- Growing adoption of combination therapies as standard of care: Clinicians increasingly use drug combinations (e.g. endothelin‑receptor antagonists + PDE‑5 inhibitors) rather than monotherapy to target multiple disease pathways, improve efficacy, slow disease progression, and enhance patient outcomes.
- Rise of novel and disease‑modifying therapies (beyond symptom management): Emergence of drugs that target vascular remodelling and underlying pathology for example, first‑in‑class therapies addressing signaling pathways is transforming PAH treatment from symptomatic relief toward potentially modifying disease course.
- Shift toward patient‑friendly routes of administration (oral, inhaled, simplified dosing): While historically many PAH therapies were injectable or infusible, there is increasing development and adoption of oral agents and inhaled formulations that offer greater convenience, better adherence, and improved quality of life.
- Regional growth in emerging markets and expanding access in Asia-Pacific: Markets in Asia‑Pacific (including emerging economies) are witnessing faster growth, driven by increasing awareness, rising healthcare investments, better access to diagnostics and treatments, and expanding availability of affordable therapies.
- Increasing focus on personalized medicine & targeted treatment strategies: Advances in molecular research, biomarkers, and genetic profiling are enabling more tailored therapeutic approaches, helping to match patients with the most effective drugs and optimizing treatment strategies.
Pulmonary Arterial Hypertension Epidemiology
The Pulmonary Arterial Hypertension epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Pulmonary Arterial Hypertension Epidemiology Segmentation:
The Pulmonary Arterial Hypertension market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalence of Pulmonary Arterial Hypertension
- Prevalent Cases of Pulmonary Arterial Hypertension by severity
- Gender-specific Prevalence of Pulmonary Arterial Hypertension
- Diagnosed Cases of Episodic and Chronic Pulmonary Arterial Hypertension
Download the report to understand which factors are driving Pulmonary Arterial Hypertension epidemiology trends @ Pulmonary Arterial Hypertension Epidemiology Forecast
Recent Development In The Pulmonary Arterial Hypertension Treatment Landscape:
- In November 2025, At the 2025 American Heart Association (AHA) Scientific Sessions in New Orleans, Japanese researchers presented findings from the SATISFY-JP trial, a Phase II study evaluating satralizumab, a targeted anti-interleukin-6 (IL-6) receptor antibody, in patients with pulmonary arterial hypertension (PAH) selected for immune activation. This trial is among the first in PAH to adopt a biomarker-driven, precision medicine approach based on elevated IL-6 levels. PAH is a rare, progressive condition characterized by high blood pressure in the lung arteries, with limited survival despite current vasodilator therapies. Emerging evidence indicates that immune system activity, particularly the IL-6 pathway, significantly contributes to disease severity in a subset of patients.
- In November 2025, Inhibikase Therapeutics, Inc. (Nasdaq: IKT), a clinical-stage biopharmaceutical company focused on developing treatments for cardiopulmonary diseases such as pulmonary arterial hypertension (PAH), announced that it plans to advance IKT-001 into a global pivotal Phase 3 clinical trial for PAH. The Phase 3 study, titled IMPROVE-PAH (IKT-001 for Measuring Pulmonary Vascular Resistance and Outcome Variables in a Phase 3 Evaluation of PAH), is expected to begin in the first quarter of 2026.
- In September 2025, AllRock Bio Inc., a clinical-stage biotech company developing therapies for cardiopulmonary and fibrotic diseases, announced a $50 million Series A financing round co-led by Versant Ventures and Westlake BioPartners. The funding will support the advancement of its lead candidate, ROC-101—an oral, first-in-class pan-rho-associated protein kinase (ROCK) inhibitor exclusively licensed from Sanofi—into Phase 2 clinical trials. ROC-101 is being developed to treat pulmonary arterial hypertension (PAH) and pulmonary hypertension associated with interstitial lung disease (ILD-PH), both life-threatening conditions with poor five-year survival rates of 57% and 38%. By targeting inflammatory, proliferative, and fibrotic remodeling driven by both ROCK1 and ROCK2, ROC-101 holds promise to meet a major unmet need in these patient populations.
- In August 2025, Cereno Scientific’s lead candidate, CS1, has been granted FDA Fast Track designation for pulmonary arterial hypertension, acknowledging its promise as a novel and potentially disease-modifying therapy for this rare and severe condition.
- In August 2025, VASTHERA Co., Ltd. announced FDA clearance of its Investigational New Drug (IND) application to begin a Phase 1 trial for VTB-10, a novel candidate for pulmonary arterial hypertension (PAH). Developed via the company’s proprietary Redoxizyme™ platform, VTB-10 is a first-in-class small molecule enzyme (Chemzyme) designed to mimic the function of peroxiredoxin (PRX), which is deficient in PAH lesions. Preclinical data showed that VTB-10 not only reversed abnormal vascular remodeling but also restored healthy endothelial function, offering a dual mechanism of action that sets it apart from current treatment options.
- In March 2025, Merck (NYSE: MRK), also known as MSD outside the U.S. and Canada, has shared initial results from its Phase 3 ZENITH trial evaluating WINREVAIR™ (sotatercept-csrk) versus placebo in adults with pulmonary arterial hypertension (PAH, Group 1 PH), classified as WHO functional class III or IV and at high risk of mortality, despite receiving the maximum tolerated background PAH therapy. With a median follow-up of 10.6 months (range: 0.3–26.1), WINREVAIR significantly reduced the relative risk of major morbidity and mortality events—including all-cause death, lung transplantation, or PAH-related hospitalization (≥24 hours)—by 76% (HR=0.24 [95% CI, 0.13–0.43]; p<0.0001 [1-sided]) compared to placebo. In the WINREVAIR group, 17.4% (15/86) of patients experienced one or more such events, versus 54.7% (47/86) in the placebo group. The safety profile of WINREVAIR in the ZENITH trial remained consistent with findings from earlier studies.
- In January 2025, Merck (NYSE: MRK), known as MSD outside the United States and Canada, announced preliminary findings from its Phase 3 ZENITH trial assessing WINREVAIR™ (sotatercept-csrk) against placebo in adults with pulmonary arterial hypertension (PAH, Group 1 PH), specifically those categorized as WHO functional class III or IV and considered at high risk of mortality despite being on optimized background PAH treatment. Over a median follow-up period of 10.6 months (range: 0.3–26.1), WINREVAIR demonstrated a 76% reduction in the relative risk of major morbidity and mortality events—defined as all-cause death, lung transplant, or PAH-related hospitalization lasting 24 hours or more compared to placebo (HR=0.24 [95% CI, 0.13–0.43]; p<0.0001 [1-sided]). In the WINREVAIR arm, 17.4% (15 of 86) of patients experienced one or more such events, compared to 54.7% (47 of 86) in the placebo group. The safety profile of WINREVAIR was generally in line with previous clinical studies.
Pulmonary Arterial Hypertension Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Pulmonary Arterial Hypertension market or expected to get launched during the study period. The analysis covers Pulmonary Arterial Hypertension market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Pulmonary Arterial Hypertension Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Pulmonary Arterial Hypertension Therapies and Key Companies
- AV-101: Aerovate Therapeutics
- LTP001: Novartis
- KER-012: Keros Therapeutics
- TPN171H: Vigonvita Life Sciences
- Treprostinil Palmitil: Insmed Incorporated
- Satralizumab (Genetical Recombination): Chugai Pharmaceutical
- Sotatercept: Merck Sharp & Dohme
- Macitentan: Janssen Pharmaceutical
- ACT-293987: Actelion
- GB002 (seralutinib): Gossamer Bio
- Parenteral Treprostinil: Lung Biotechnology PBC
- Ralinepag: United Therapeutics
- Selonsertib: Gilead Sciences
- Sotatercept: Acceleron Pharma
- ubenimex: Eiger BioPharmaceuticals
- Olaparib: AstraZeneca
Discover more about therapies set to grab major Pulmonary Arterial Hypertension market share @ Pulmonary Arterial Hypertension Treatment Landscape
Pulmonary Arterial Hypertension Market Drivers
- Rising Disease Prevalence: Increasing number of diagnosed PAH cases globally is boosting market demand.
- Advancements in Targeted Therapies: Development of novel drugs such as ralinepag, RT234, and Seralutinib improving treatment outcomes.
- Improved Diagnostic Capabilities: Enhanced imaging and biomarker-based diagnostics enable early detection and personalized treatment.
- Growing R&D Investment: Biopharmaceutical companies are heavily investing in clinical trials for innovative PAH therapies.
- Regulatory Support: Orphan drug designations and accelerated approval pathways are facilitating faster market entry of new therapies.
Pulmonary Arterial Hypertension Market Barriers
- High Treatment Costs: Expensive therapies and long-term treatment regimens limit patient access, especially in developing regions.
- Complex Disease Pathophysiology: Heterogeneity of PAH and underlying comorbidities make treatment challenging.
- Limited Curative Options: Current therapies mainly manage symptoms and slow disease progression rather than provide a cure.
- Side Effects and Safety Concerns: Adverse reactions associated with PAH drugs can impact patient compliance.
- Market Fragmentation: Variability in treatment availability and healthcare infrastructure across regions restricts uniform adoption.
Scope of the Pulmonary Arterial Hypertension Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Pulmonary Arterial Hypertension Companies: Aerovate Therapeutics, Novartis, Keros Therapeutics, Vigonvita Life Sciences, Insmed Incorporated, Chugai Pharmaceutical, Merck Sharp & Dohme, Janssen Pharmaceutical, Actelion, Gossamer Bio, Lung Biotechnology PBC, United Therapeutics, Gilead Science, Acceleron Pharma, Eiger BioPharmaceuticals, AstraZeneca, and others
- Key Pulmonary Arterial Hypertension Therapies: AV-101, LTP001, KER-012, TPN171H, Treprostinil Palmitil, Satralizumab (Genetical Recombination), Sotatercept, Macitentan, ACT-293987, GB002 (seralutinib), Parenteral Treprostinil, Ralinepag, Selonsertib, Sotatercept, ubenimex, Olaparib, and others
- Pulmonary Arterial Hypertension Therapeutic Assessment: Pulmonary Arterial Hypertension current marketed and Pulmonary Arterial Hypertension emerging therapies
- Pulmonary Arterial Hypertension Market Dynamics: Pulmonary Arterial Hypertension market drivers and Pulmonary Arterial Hypertension market barriers
- Pulmonary Arterial Hypertension Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Pulmonary Arterial Hypertension Unmet Needs, KOL’s views, Analyst’s views, Pulmonary Arterial Hypertension Market Access and Reimbursement
To know more about Pulmonary Arterial Hypertension companies working in the treatment market, visit @ Pulmonary Arterial Hypertension Clinical Trials and Therapeutic Assessment
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Pulmonary Arterial Hypertension - Epidemiology Forecast to 2034
DelveInsight's Pulmonary Arterial Hypertension - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of the disease...
Pulmonary Arterial Hypertension - Pipeline Insight, 2025
Pulmonary Arterial Hypertension Pipeline Insights, 2025 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across..