Sarcopenia Market to Surge from USD 2,276 Million in 2025 to grow at a CAGR of 10..6% to reach USD 5,626 Million by 2034
Get a Sneak Peek at the Latest sarcopenia market size and forecast Report
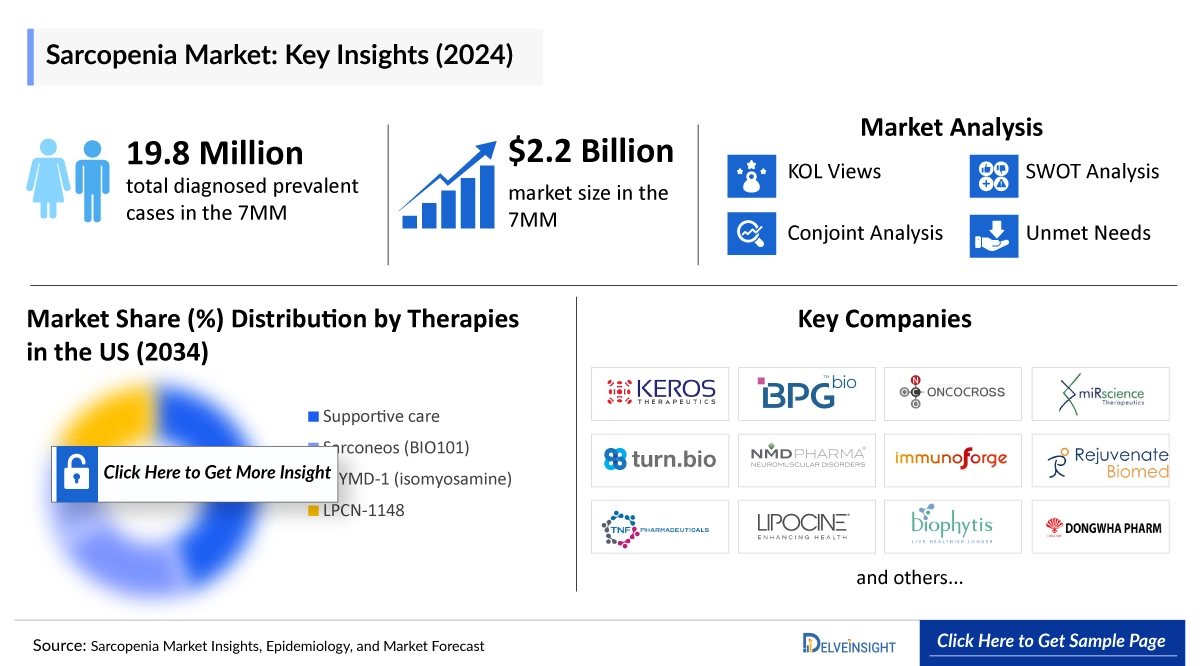
The Sarcopenia market size was estimated at approximately USD 2,276 million in 2025 and is projected to reach USD 5,626 million by 2034, growing at a robust CAGR of 10.6% over the forecast period from 2020 to 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Sarcopenia landscape.
In 2025, the Sarcopenia market in the US was the largest among the 7MM countries, valued at approximately USD 1,500 million, with continued growth expected through 2034.
Major companies developing therapies for Sarcopenia include TNF Pharmaceuticals with MYMD-1/Isomyosamine, Biophytis with Sarconeos, and Lipocine with LPCN 1148. In addition, several firms are advancing early-stage sarcopenia treatments, such as ImmunoForge (Froniglutide), Rejuvenate Biomed (RJx-01), Keros Therapeutics (KER-065), and BPGbio (BPM 31510), alongside other companies progressing candidates through various stages of clinical development.
In 2024, the total diagnosed prevalence of Sarcopenia across the 7MM was estimated at approximately 19,836,200 cases, with projections indicating continued growth over the forecast period from 2020 to 2034. Within the EU4 and the UK, the UK reported the highest number of diagnosed cases, followed by Germany, while Spain had the lowest. In the US, cases are segmented by age groups 65–69 years, 70–79 years, and 80 years and above with the 65–69 age group recording the highest prevalence at around 4,520,200 cases. In Japan, sarcopenia cases were estimated at approximately 5,225,100 in 2024.
DelveInsight’s report, “Sarcopenia Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Sarcopenia landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Sarcopenia market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Sarcopenia market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Sarcopenia Market Forecast
Some of the key facts of the Sarcopenia Market Report:
- The Sarcopenia treatment pipeline is advancing with the development of innovative therapies employing a variety of mechanisms of action. These include TNF-α inhibitors (MYMD-1), androgen receptor agonists (LPCN 1148), proto-oncogene protein c-MAS-1 agonists (Sarconeos), glucagon-like peptide-1 (GLP-1) receptor agonists (Froniglutide), and compounds targeting the aerobic glycolytic pathway (BPM 31510), among several other emerging candidates.
- Key Sarcopenia Companies: TNF Pharmaceuticals, Lipocine, Biophytis, ImmunoForge, Rejuvenate Biomed, Keros Therapeutics, BPGbio, Oncocross, Mirscience Therapeutics, Turn Biotechnologies, NMD PHARMA, ImmunoForge, Dong Wha PHARM, and others
- Key Sarcopenia Therapies: MYMD-1/Isomyosamine, LPCN 1148, Sarconeos (BIO101), Froniglutide (PF-1801), RJx-01, KER-065, BPM 31510 (ubidecarenone), OC514, MT29, TRN-005, NMDP-05, PF1807, DW1030, and others
- The Sarcopenia market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Sarcopenia pipeline products will significantly revolutionize the Sarcopenia market dynamics.
Sarcopenia Overview
Sarcopenia is a progressive condition characterized by the loss of muscle mass, strength, and function, commonly associated with aging. It primarily affects older adults and can significantly impact mobility, balance, and overall quality of life.
The condition is caused by multiple factors, including reduced physical activity, hormonal changes, poor nutrition (especially low protein intake), and chronic illnesses. Individuals with sarcopenia may experience weakness, slower movement, increased risk of falls, and difficulty performing daily activities.
Get a Free sample for the Sarcopenia Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/sarcopenia-market
Key Trends in Sarcopenia Therapeutics Market:
- Rising Focus on Novel Pharmacological Interventions: Development of myostatin inhibitors, selective androgen receptor modulators (SARMs), and other anabolic agents to improve muscle mass and strength.
- Emphasis on Lifestyle and Nutritional Therapies: Integration of protein supplementation, vitamin D, and resistance exercise programs as part of comprehensive management.
- Growing Awareness and Early Diagnosis: Increased screening and diagnostic efforts among aging populations are driving early intervention and treatment adoption.
- Expansion of Digital Health Solutions: Use of wearable devices and telehealth platforms to monitor muscle function, track progress, and enhance patient compliance.
- Investment in R&D and Clinical Trials: Pharmaceutical companies are heavily investing in late-stage trials to develop disease-modifying treatments targeting sarcopenia.
Sarcopenia Epidemiology
The Sarcopenia epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Sarcopenia Epidemiology Segmentation:
The Sarcopenia market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Prevalence of Sarcopenia
- Prevalent Cases of Sarcopenia by severity
- Gender-specific Prevalence of Sarcopenia
- Diagnosed Cases of Episodic and Chronic Sarcopenia
Download the report to understand which factors are driving Sarcopenia epidemiology trends @ Sarcopenia Epidemiology Forecast
Recent Development In The Sarcopenia Treatment Landscape:
- In March 2026, Epirium Bio Inc., a clinical-stage biopharmaceutical company developing therapies for neuromuscular and fibrotic disorders, announced that new clinical and translational findings for MF-300 will be showcased in two oral presentations at the Intrinsic Capacity, Frailty and Sarcopenia Research Conference (ICFSR) 2026, scheduled for March 10–12 in Washington, DC.
- In September 2025, Epirium Bio Inc. (Epirium), a clinical-stage biopharmaceutical company focused on treatments for neuromuscular and fibrotic diseases, announced positive Phase 1 trial results for MF-300, a novel therapy under development for sarcopenia. The trial met its primary safety endpoint, with all tested doses of MF-300 generally well tolerated and no participant discontinuations. MF-300 showed dose-dependent pharmacodynamic (PD) responses that were evident early and maintained over time, while the placebo group showed no significant changes, confirming target engagement and biological activity. Pharmacokinetic (PK) analysis revealed dose-related increases in exposure, with a half-life supporting convenient once-daily oral dosing.
- In July 2025, Epirium Bio Inc., a clinical-stage biopharmaceutical company focused on neuromuscular and fibrotic diseases, announced the completion of dosing in its Phase 1 trial of MF-300. This randomized, double-blind, placebo-controlled single and multiple ascending dose (SAD/MAD) study evaluated the safety, tolerability, pharmacokinetics, and pharmacodynamics of MF-300 in healthy adults. MF-300, an investigational first-in-class oral inhibitor of the 15-hydroxyprostaglandin dehydrogenase (15-PGDH) enzyme, is being developed to treat sarcopenia, or age-related muscle weakness. Preliminary findings showed all adverse events were mild to moderate, with no severe or serious cases, no early discontinuations, and no stopping criteria met.
- In April 2025, TNF Pharmaceuticals, Inc. announced that Mitchell Glass, M.D., President and Chief Medical Officer of TNF, delivered a platform presentation on the abstract titled “Isomyosamine for the Treatment of Sarcopenia in Older Adults” at the British Geriatrics Society (BGS) Spring Meeting 2025, which took place from April 9–11 in Belfast, Ireland, and online.
- In December 2024, The US FDA awarded Fast Track Designation (FTD) to LPCN 1148 for the treatment of sarcopenia in patients with decompensated cirrhosis.
- In December 2024, Epirium Bio, Inc. (Epirium) announced that the US Food and Drug Administration (FDA) has approved the Investigational New Drug (IND) application for MF-300, a novel, orally administered 15-hydroxyprostaglandin dehydrogenase (15-PGDH) enzyme inhibitor being developed to treat sarcopenia, which is age-related muscle weakness.
- In December 2024, Lipocine announced that the US Food and Drug Administration (FDA) has awarded Fast Track Designation to LPCN1148 for the treatment of sarcopenia in patients suffering from decompensated cirrhosis.
Sarcopenia Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Sarcopenia market or expected to get launched during the study period. The analysis covers Sarcopenia market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Sarcopenia Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Sarcopenia Therapies and Key Companies
- MYMD-1/Isomyosamine: TNF Pharmaceuticals
- LPCN 1148: Lipocine
- Sarconeos (BIO101): Biophytis
- Froniglutide (PF-1801): ImmunoForge
- RJx-01: Rejuvenate Biomed
- KER-065: Keros Therapeutics
- BPM 31510 (ubidecarenone): BPGbio
- OC514: Oncocross
- MT29: Mirscience Therapeutics
- TRN-005: Turn Biotechnologies
- NMDP-05: NMD PHARMA
- PF1807: ImmunoForge
- DW1030: Dong Wha PHARM
Discover more about therapies set to grab major Sarcopenia market share @ Sarcopenia Treatment Landscape
Sarcopenia Market Drivers
- Rapidly Aging Population: The rising prevalence of Sarcopenia is strongly linked to the growing global elderly population, increasing demand for diagnosis and treatment.
- Increased Awareness of Muscle Health: Greater focus on age-related muscle loss and preventive healthcare is boosting early diagnosis and intervention.
- Advancements in Therapeutics & Nutrition: Development of novel drugs, protein supplements, and nutraceuticals is supporting better disease management.
- Rising Focus on Preventive Healthcare: Adoption of fitness programs, rehabilitation therapies, and lifestyle interventions is contributing to market growth.
- Growing Research and Clinical Trials: Increased R&D investments and clinical studies are expanding the pipeline for innovative sarcopenia treatments.
Sarcopenia Market Barriers
- Lack of Standardized Diagnostic Criteria: Variability in diagnosis and absence of universally accepted guidelines hinder accurate disease identification.
- Limited Approved Pharmacological Treatments: Few approved drugs specifically targeting sarcopenia restrict market expansion.
- Low Awareness in Developing Regions: Underdiagnosis and limited healthcare access reduce treatment uptake.
- High Treatment and Management Costs: Long-term therapies, supplements, and rehabilitation programs can be expensive.
- Complex Disease Mechanism: Multifactorial causes, including aging, inactivity, and comorbidities, make treatment challenging and slow drug development.
Scope of the Sarcopenia Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom), and Japan]
- Key Sarcopenia Companies: TNF Pharmaceuticals, Lipocine, Biophytis, ImmunoForge, Rejuvenate Biomed, Keros Therapeutics, BPGbio, Oncocross, Mirscience Therapeutics, Turn Biotechnologies, NMD PHARMA, ImmunoForge, Dong Wha PHARM, and others
- Key Sarcopenia Therapies: MYMD-1/Isomyosamine, LPCN 1148, Sarconeos (BIO101), Froniglutide (PF-1801), RJx-01, KER-065, BPM 31510 (ubidecarenone), OC514, MT29, TRN-005, NMDP-05, PF1807, DW1030, and others
- Sarcopenia Therapeutic Assessment: Sarcopenia current marketed and Sarcopenia emerging therapies
- Sarcopenia Market Dynamics: Sarcopenia market drivers and Sarcopenia market barriers
- Sarcopenia Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Sarcopenia Unmet Needs, KOL’s views, Analyst’s views, Sarcopenia Market Access and Reimbursement
To know more about Sarcopenia companies working in the treatment market, visit @ Sarcopenia Clinical Trials and Therapeutic Assessment
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Sarcopenia Epidemiology Forecast - 2034
DelveInsight's Sarcopenia - Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, and forecasted epidemiology of Sarcopenia in the United States
Sarcopenia Pipeline Insight, 2026
Sarcopenia Pipeline Insights, 2026 report by DelveInsight outlays comprehensive insights of present clinical development scenario and growth prospects across the Sarcopenia market.