Sickle Cell Disease Market Estimated at USD 650 Million in 2024, Set for Steady Growth Through 2034
Get a Sneak Peek at the Latest sickle cell disease market size and insights Report
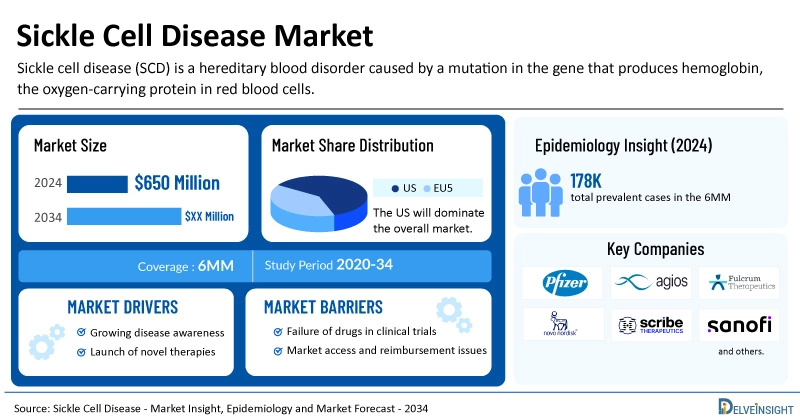
Across the 6MM countries, the Sickle Cell Disease market size was estimated at around USD 650 million in 2024 and is expected to experience steady growth over the forecast period from 2024 to 2034. DelveInsight’s comprehensive market research provides critical insights into such market trends, enabling stakeholders to understand growth drivers, emerging opportunities, and potential challenges within the Sickle Cell Disease landscape.
In 2024, the US represented the largest share of the Sickle Cell Disease market, accounting for approximately 87% compared to the EU4 (Germany, Italy, France, and Spain) and the UK. According to DelveInsight estimates, within the EU4 and the UK, France held the largest market for Sickle Cell Disease, while Spain had the smallest market share.
Estimates indicate that the United States accounted for roughly 73% of the total diagnosed prevalent cases of Sickle Cell Disease (SCD) across the 6MM. According to DelveInsight’s 2024 analysis, the total prevalent SCD cases in the 6MM were approximately 178,500, with numbers expected to increase by 2034. Among the EU4 and the UK, France reported the highest prevalence, with around 26,700 cases in 2024.
According to the analysis, in the United States, the largest proportion of Sickle Cell Disease (SCD) cases occurred in individuals aged 18-44 years, followed by those aged 45–64 years. In France, sickle cell anemia (hemoglobin S/S) was the most common SCD subtype in 2024, with approximately 17,000 cases.
Currently, Sickle Cell Disease is treated using NSAIDs, blood transfusions, chelating agents, nutritional supplements, and broad-spectrum antibiotics. Key established therapies in the Sickle Cell Disease market include DROXIA (hydroxyurea), ENDARI (L-glutamine oral powder), ADAKVEO (crizanlizumab-tmca), among others.
Gene therapy and gene editing approaches show potential in addressing the underlying genetic mutation that causes Sickle Cell Disease. The recent FDA approvals of Vertex/CRISPR’s CASGEVY and Bluebird’s LYFGENIA provide the possibility of a one-time, transformative treatment for eligible Sickle Cell Disease patients.
DelveInsight’s report, “Sickle Cell Disease Market Insights, Epidemiology, and Market Forecast-2034,” provides a comprehensive analysis of the Sickle Cell Disease landscape. The report delivers detailed insights into the disease, including historical and projected epidemiology, helping stakeholders understand the prevalence, incidence, and patient demographics across key regions.
Additionally, it examines Sickle Cell Disease market dynamics, offering a thorough assessment of current and emerging market trends, treatment patterns, and therapeutic developments. The analysis spans major markets, including the United States, EU4 (Germany, Spain, Italy, France, and the United Kingdom), and Japan, equipping decision-makers with actionable intelligence for strategic planning, investment, and research initiatives.
To Know in detail about the Sickle Cell Disease market outlook, drug uptake, treatment scenario and epidemiology trends, Click here; Sickle Cell Disease Market Forecast
Some of the key facts of the Sickle Cell Disease Market Report:
- All OXBRYTA batches for the treatment of Sickle Cell Disease (SCD) were withdrawn from global markets in September 2024. Among the remaining therapies, CASGEVY and LYFGENIA are expected to become key market leaders, significantly influencing the Sickle Cell Disease market across the 6MM countries by 2034.
- Key Sickle Cell Disease Companies: BRL Medicine, Oryzon Genomics, GlaxoSmithKline, Agios Pharmaceuticals, Beam Therapeutics Inc., Graphite Bio, Roche, Quercis Pharma, Editas Medicin, Pfizer, Emmaus Medical, Inc, Vertex Pharmaceuticals, CRISPR therapeutics, Bluebird Bio, Pfizer, Novo Nordisk, Agios Pharmaceuticals, Alexion Pharmaceuticals, Takeda, Prolong Pharmaceuticals, Roche, Beam Therapeutics, Editas Medicine, Sangamo Therapeutics, Bellicum Pharmaceuticals, Invenux, EpiDestiny, Hillhurst Biopharmaceuticals, CSL Behring, Fulcrum Therapeutics, Sana Biotechnology, and others
- Key Sickle Cell Disease Therapies: RL 101, ORY-300, GSK 4172239D, AG-946, BEAM-101, Nula-cel, RG 6107, Isoquercetin, Renizgamglogene autogedtemcel, Inclacumab, L-glutamine, CASGEVY, CTX001, LentiGlobin BB305, Inclacumab, Etavopivat, Mitapivat, ALXN1820, TAK-755, Sanguinate, Crovalimab, BEAM101, EDIT 301, BIVV003, BPX-501, SCD-101, Nicotinamide, HBI-002, CSL889, FTX-6058, SG418n, and others
- The Sickle Cell Disease epidemiology based on gender analyzed that Sickle Cell Disease affects males and females equally
- The Sickle Cell Disease market is expected to surge due to the disease's increasing prevalence and awareness during the forecast period. Furthermore, launching various multiple-stage Sickle Cell Disease pipeline products will significantly revolutionize the Sickle Cell Disease market dynamics.
Sickle Cell Disease Overview
Sickle Cell Disease (SCD) is a hereditary blood disorder caused by a genetic mutation that leads to the production of abnormal hemoglobin (hemoglobin S). This causes red blood cells to become rigid, sticky, and shaped like a sickle (crescent), instead of their normal round shape.
These abnormal cells can block blood flow in small vessels, leading to severe pain episodes (vaso-occlusive crises), anemia, organ damage, and increased risk of infections. Sickle cell disease is most commonly found in individuals of African, Middle Eastern, and South Asian descent.
Get a Free sample for the Sickle Cell Disease Market Forecast, Size & Share Analysis Report:
https://www.delveinsight.com/report-store/sickle-cell-disease-6mm-market
Key Trends in Sickle Cell Disease Therapeutics Market:
- Rise of Gene and Cell Therapies: Increasing focus on CRISPR-based gene editing and stem cell transplantation as potential curative treatments.
- Expansion of Disease-Modifying Drugs: Growth in use of hydroxyurea and emerging pharmacologic agents to reduce vaso-occlusive crises and improve quality of life.
- Early Diagnosis and Newborn Screening Programs: Enhanced screening initiatives are enabling timely intervention and better disease management.
- Adoption of Personalized Medicine: Development of targeted therapies based on individual genetic and clinical profiles.
- Increased Investment in R&D: Pharmaceutical companies are heavily investing in innovative treatments and expanding global clinical trials.
Sickle Cell Disease Epidemiology
The Sickle Cell Disease epidemiology section provides insights into the historical, current, and forecasted epidemiology trends in the seven major countries (7MM) from 2020 to 2034. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. The epidemiology section also provides a detailed analysis of the diagnosed patient pool and future trends.
Sickle Cell Disease Epidemiology Segmentation:
The Sickle Cell Disease market report proffers epidemiological analysis for the study period 2020–2034 in the 7MM segmented into:
- Total Diagnosed Cases of Sickle Cell Disease in the 7MM
- Age-specific Diagnosed Cases of Sickle Cell Disease in the 7MM
- Type-specific Diagnosed Cases of Sickle Cell Disease in the 7MM
Download the report to understand which factors are driving Sickle Cell Disease epidemiology trends @ Sickle Cell Disease Epidemiology Forecast
Recent Development In The Sickle Cell Disease Treatment Landscape:
- In August 2025, Pfizer is reevaluating its strategy after its investigational sickle cell disease therapy did not meet the primary endpoint in the Phase III THRIVE-131 trial (NCT04935879). The study, which assessed the P-selectin inhibitor inclacumab in patients aged 16 and older with sickle cell disease, failed to show a significant reduction in vaso-occlusive crises (VOCs) over 48 weeks, raising uncertainty about the drug’s future development.
- In June 2025, Ellarity has begun dosing the first participant in its Phase I clinical trial of CLY-124, an oral drug candidate for treating sickle cell disease (SCD). This milestone follows the US FDA’s approval of the company’s investigational new drug (IND) application. The global study aims to evaluate the safety, tolerability, and pharmacokinetics of the therapy, initially in healthy volunteers and later in patients with SCD.
- In June 2025, The US FDA has granted a special designation to Vascarta’s lead candidate, VAS-101 (Vasceptor), for treating sickle cell disease (SCD). This designation provides benefits such as seven years of market exclusivity post-approval and exemptions from certain FDA fees. VAS-101 is a patented topical curcumin formulation developed using Vascarta’s transdermal delivery technology to overcome the poor bioavailability and limited efficacy of oral curcumin. Preclinical animal studies suggest that VAS-101 may help reduce chronic pain, enhance red blood cell stability, and lower inflammation in patients with SCD.
- In May 2025, Beam Therapeutics Inc. (Nasdaq: BEAM), a biotechnology company focused on developing precision genetic medicines through base editing, announced that it will present new data from its BEACON Phase 1/2 clinical trial of BEAM-101 at the upcoming European Hematology Association (EHA) 2025 Congress, taking place from June 12–15, 2025, in Milan, Italy. BEAM-101 is an investigational ex vivo genetically modified cell therapy designed to treat sickle cell disease (SCD), particularly in patients suffering from severe vaso-occlusive crises (VOCs).
- In December 2024, Beam Therapeutics Inc. (Nasdaq: BEAM), a biotechnology company focused on precision genetic medicines through base editing, reported new safety and efficacy results from its BEACON Phase 1/2 trial evaluating BEAM-101 in patients with sickle cell disease (SCD) suffering from severe vaso-occlusive crises (VOCs). The findings were featured in the press program at the 66th American Society of Hematology (ASH) Annual Meeting.
- In November 2024, BioLineRx Ltd. (NASDAQ: BLRX) (TASE: BLRX), a commercial-stage biopharmaceutical company specializing in oncology and rare diseases, announced that an abstract presenting initial data from its Phase 1 trial of motixafortide—evaluated alone and in combination with natalizumab for CD34+ hematopoietic stem cell (HSC) mobilization in gene therapies for sickle cell disease (SCD) has been selected for oral presentation at the 66th American Society of Hematology (ASH) Annual Meeting & Exposition, scheduled for December 7–10, 2024, in San Diego, California. Conducted in partnership with Washington University School of Medicine in St. Louis, the study aims to identify alternative HSC mobilization strategies to improve the gene therapy experience for SCD patients
Sickle Cell Disease Drugs Uptake and Pipeline Development Activities
The drugs uptake section focuses on the rate of uptake of the potential drugs recently launched in the Sickle Cell Disease market or expected to get launched during the study period. The analysis covers Sickle Cell Disease market uptake by drugs, patient uptake by therapies, and sales of each drug.
Moreover, the therapeutics assessment section helps understand the drugs with the most rapid uptake and the reasons behind the maximal use of the drugs. Additionally, it compares the drugs based on market share.
The report also covers the Sickle Cell Disease Pipeline Development Activities. It provides valuable insights about different therapeutic candidates in various stages and the key companies involved in developing targeted therapeutics. It also analyzes recent developments such as collaborations, acquisitions, mergers, licensing patent details, and other information for emerging therapies.
Sickle Cell Disease Therapies and Key Companies
- RL 101: BRL Medicine
- ORY-300:1 Oryzon Genomics
- GSK 4172239D: GlaxoSmithKline
- AG-946: Agios Pharmaceuticals
- BEAM-101: Beam Therapeutics Inc.
- Nula-cel: Graphite Bio
- RG 6107: Roche
- Isoquercetin: Quercis Pharma
- Renizgamglogene autogedtemcel: Editas Medicin
- Inclacumab: Pfizer
- L-glutamine: Emmaus Medical, Inc
- CASGEVY: Vertex Pharmaceuticals
- ESCAPE: Beam Therapeutic
- IHP-102: IHP Therapeutics
- HBI-002: Hillhurst Biopharmaceuticals
- BEAM101: Beam Therapeutics
- EPI01: Novo Nordisk
- VIT-2763: CSL Vifor
- Inclacumab: Pfizer
- L-glutamine: Emmaus Medical
- Oxbryta: Pfizer
- Exagamglogene autotemcel: CRISPR Therapeutics/Vertex Pharmaceuticals
- Mitapivat: Agios Pharmaceuticals
- Canakinumab: Novartis
- ALXN1820: Alexion Pharmaceuticals
- Crovalimab: Chugai Pharmaceutical/Roche
- EDIT 301: Editas Medicine
- BIVV003: Sangamo Therapeutics
- BEAM101: Beam Therapeutics
- Hemopexin: CSL Behring
Discover more about therapies set to grab major Sickle Cell Disease market share @ Sickle Cell Disease Treatment Landscape
Sickle Cell Disease Market Drivers
- Rising Disease Burden: Increasing prevalence of Sickle Cell Disease, particularly in Africa, India, and the Middle East, is driving demand for effective therapies.
- Advancements in Gene Therapy: Breakthrough innovations such as gene editing (e.g., CRISPR-based therapies) are transforming treatment potential.
- Growing Awareness & Screening Programs: Government initiatives and newborn screening programs are improving early diagnosis and management.
- Strong Pipeline and R&D Investments: Pharmaceutical companies are actively developing novel therapies, including curative approaches.
- Improved Supportive Care: Advances in pain management, blood transfusion practices, and disease-modifying drugs are enhancing patient outcomes.
Sickle Cell Disease Market Barriers
- High Cost of Advanced Treatments: Gene therapies and novel biologics are extremely expensive, limiting patient access.
- Limited Healthcare Infrastructure: In low- and middle-income countries, inadequate facilities hinder proper diagnosis and treatment.
- Treatment Accessibility Issues: Unequal access to advanced therapies and specialists restricts market growth.
- Safety and Long-Term Efficacy Concerns: Emerging therapies, especially gene editing, face uncertainties regarding long-term outcomes.
- Patient Compliance Challenges: Lifelong disease management, frequent hospital visits, and treatment burden can affect adherence.
Scope of the Sickle Cell Disease Market Report
- Study Period: 2020–2034
- Coverage: 7MM [The United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom)]
- Key Sickle Cell Disease Companies: BRL Medicine, Oryzon Genomics, GlaxoSmithKline, Agios Pharmaceuticals, Beam Therapeutics Inc., Graphite Bio, Roche, Quercis Pharma, Editas Medicin, Pfizer, Emmaus Medical, Inc, Vertex Pharmaceuticals, CRISPR therapeutics, Bluebird Bio, Pfizer, Novo Nordisk, Agios Pharmaceuticals, Alexion Pharmaceuticals, Takeda, Prolong Pharmaceuticals, Roche, Beam Therapeutics, Editas Medicine, Sangamo Therapeutics, Bellicum Pharmaceuticals, Invenux, EpiDestiny, Hillhurst Biopharmaceuticals, CSL Behring, Fulcrum Therapeutics, Sana Biotechnology, and others
- Key Sickle Cell Disease Therapies: RL 101, ORY-300, GSK 4172239D, AG-946, BEAM-101, Nula-cel, RG 6107, Isoquercetin, Renizgamglogene autogedtemcel, Inclacumab, L-glutamine, CASGEVY, CTX001, LentiGlobin BB305, Inclacumab, Etavopivat, Mitapivat, ALXN1820, TAK-755, Sanguinate, Crovalimab, BEAM101, EDIT 301, BIVV003, BPX-501, SCD-101, Nicotinamide, HBI-002, CSL889, FTX-6058, SG418, and others
- Sickle Cell Disease Therapeutic Assessment: Sickle Cell Disease current marketed and Sickle Cell Disease emerging therapies
- Sickle Cell Disease Market Dynamics: Sickle Cell Disease market drivers and Sickle Cell Disease market barriers
- Sickle Cell Disease Competitive Intelligence Analysis: SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies
- Sickle Cell Disease Unmet Needs, KOL’s views, Analyst’s views, Sickle Cell Disease Market Access and Reimbursement
To know more about Sickle Cell Disease companies working in the treatment market, visit @ Sickle Cell Disease Clinical Trials and Therapeutic Assessment
About DelveInsight
DelveInsight is a leading Healthcare Business Consultant, and Market Research firm focused exclusively on life sciences. It supports Pharma companies by providing comprehensive end-to-end solutions to improve their performance.
It also offers Healthcare Consulting Services, which benefits in market analysis to accelerate the business growth and overcome challenges with a practical approach.
Need more?
- ✅ Connect with our analyst to learn how this research was developed
- ✅ Expand the scope with additional segments or countries through free customization
- ✅ Discover how this report can directly influence your business growth
Related Reports
Sickle Cell Disease - Pipeline Insight, 2026
Sickle Cell Disease Pipeline Insight 2026 report by DelveInsight outlays comprehensive insights of present clinical development scenario & growth prospects across the Sickle Cell market
Sickle Cell Disease - Epidemiology Forecast - 2034
DelveInsight's Sickle Cell Disease Epidemiology Forecast 2034 report delivers an in-depth understanding of the disease, historical, & forecasted epidemiology of Sickle Cell Disease..
-pipeline.png&w=256&q=75)

