Alcohol Use Disorder Market Summary
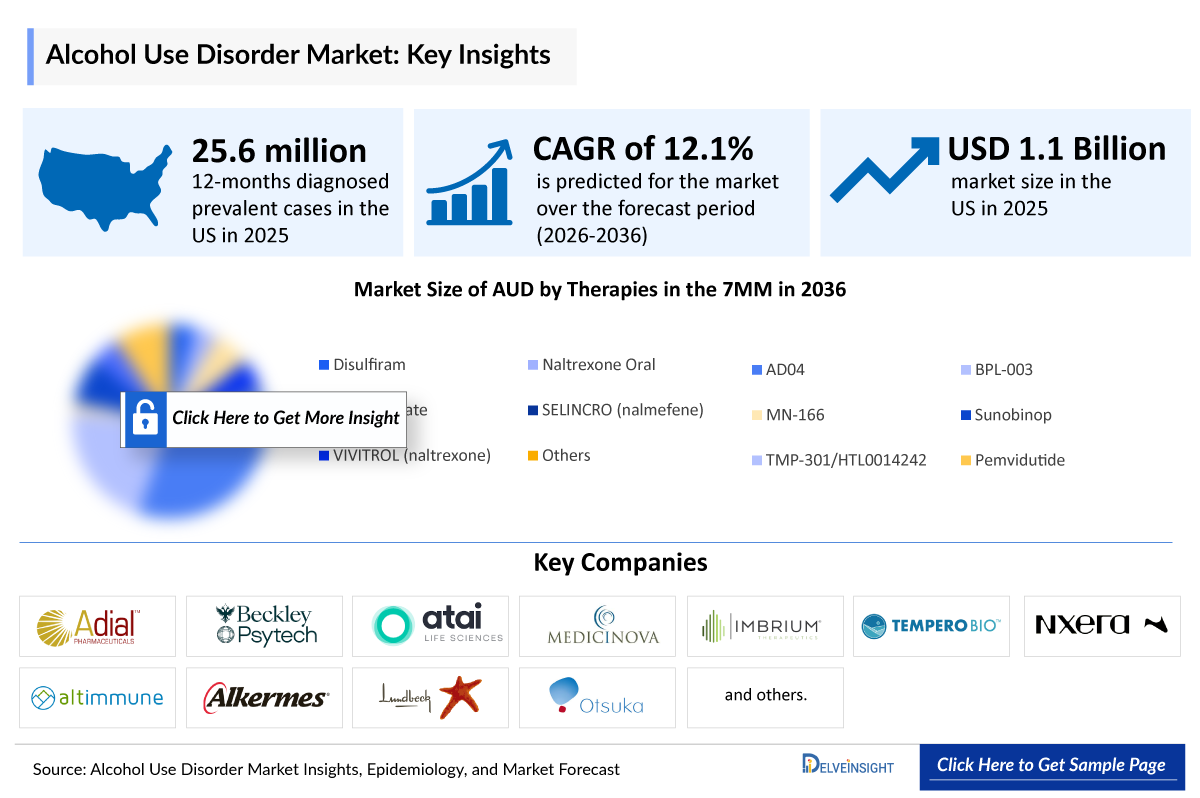
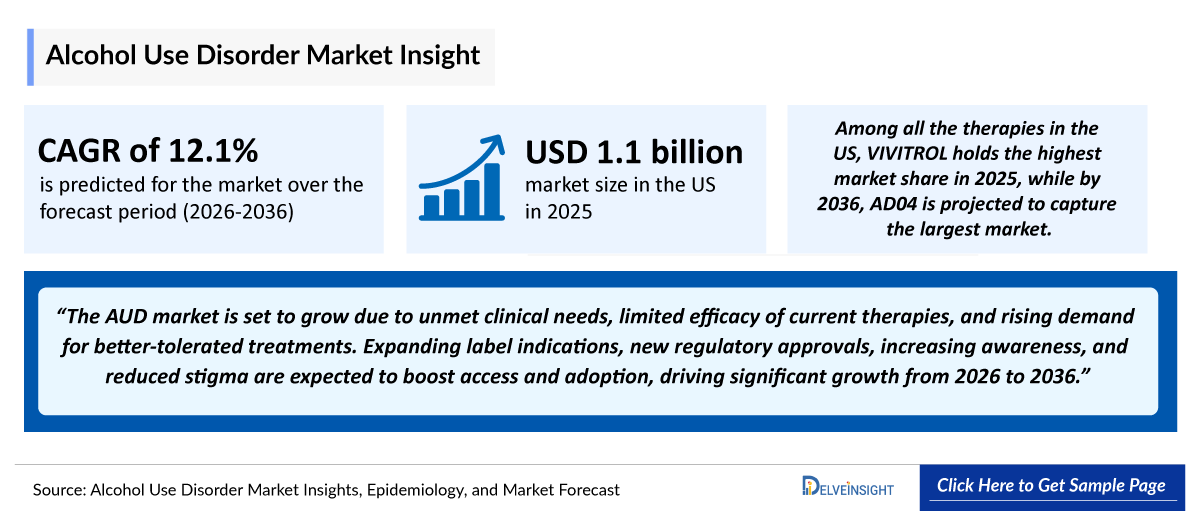
- The Alcohol Use Disorder Market Size was valued ~USD 1,100 million is anticipated to reach ~USD 3,600 million growing with a significant CAGRof 12.1% during the study period (2022-2036).
- The leading Alcohol Use Disorder companies developing therapies in the treatment market include - Alkermes , Adial Pharmaceuticals, Eli Lilly and Company, MediciNova, Imbrium Therapeutics (subsidiary of Purdue Pharma), and others.
Alcohol Use Disorder Market and Epidemiology Analysis
- The US has experienced a statistically significant decline in AUD, accompanied by parallel reductions in binge drinking, heavy alcohol use, and underage alcohol consumption among individuals aged 12–20. Reflecting these positive shifts, the 2025 landscape highlights a strong and growing momentum toward recovery.
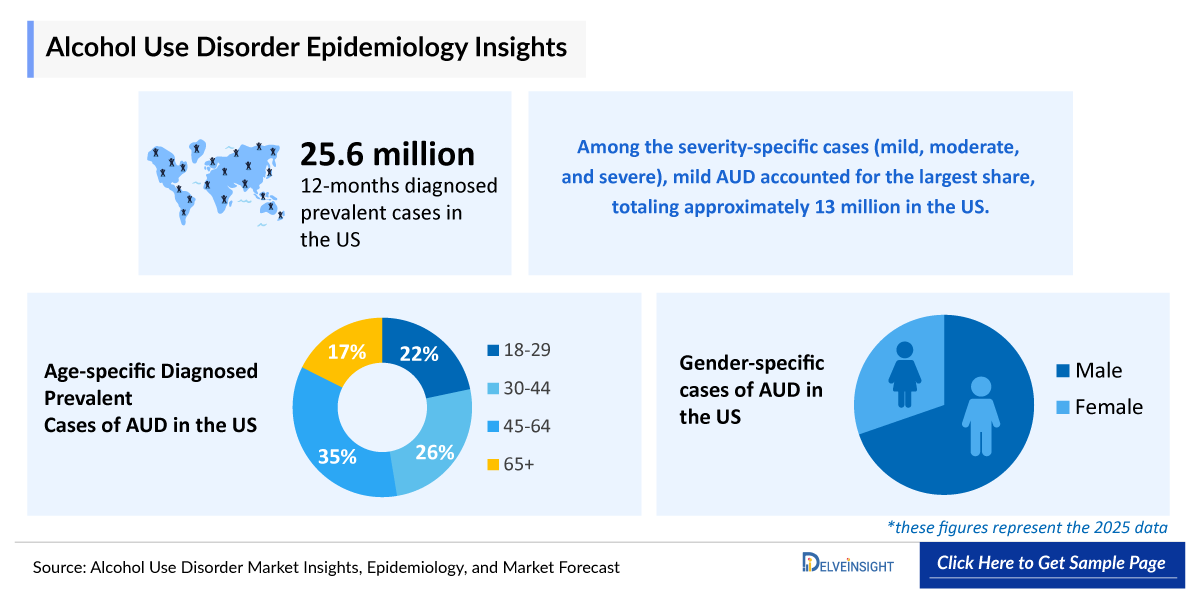
- The United States has a substantial burden of AUD, with recent estimates suggesting ~25 million Americans are affected, potentially reflecting comparatively permissive cultural norms around alcohol consumption.
- AUD remains largely untreated, with fears of stigmatization and perceptions that treatment is ineffective contributing to low treatment uptake. Additionally, existing therapeutic options show limited long-term effectiveness, as nearly 90% of patients do not achieve sustained abstinence.
- Patients with AUD are classified as mild, moderate, or severe under DSM-5 criteria, with nearly 14 million individuals experiencing moderate to severe disease. Approved treatments have limited efficacy for AUD. Consequently, there is a substantial unmet need for more effective therapies that reduce alcohol cravings and heavy drinking while addressing associated comorbidities.
- Currently approved therapies include CAMPRAL (acamprosate), ANTABUSE (disulfiram), REVIA (oral naltrexone), and VIVITROL (extended-release injectable naltrexone). Among all, generic versions are widely available for all drugs except for VIVITROL, enhancing accessibility.
- Current AUD treatments lack biomarker guidance, leading to trial-and-error care. Adial Pharmaceuticals (AD04) introduces genetically targeted, oral precision therapy, currently in clinical trials. The drug is administered twice daily, with plans to transition to once-daily dosing, positioning it as the only late-stage asset with launch expected by 2028.
- Currently, no approved treatment is tailored to AUD severity; patients generally receive uniform therapy regardless of individual risk. However, emerging candidates like Sunobinop, Pemvidutide, and others are being evaluated for moderate-to-severe AUD.
Alcohol Use Disorder Market size and forecast in the 7MM
- 2025 AUD Market Size: USD 1,100 million
- 2036 Projected AUD Market Size: USD 3,600 million
- AUD Growth Rate (2026–2036): 12.1% CAGR
Request for unlocking the Sample Page of the "Alcohol Use Disorder Market Insights"
Key Factors Driving the Alcohol Use Disorder Market
- Rising Global Alcohol Consumption: Increasing alcohol intake, particularly in emerging economies and younger populations, is expanding the prevalence of Alcohol Use Disorder.
- Growing Awareness and Diagnosis: Improved recognition of mental health and substance abuse disorders is leading to higher diagnosis and treatment rates.
- Advancements in Pharmacological Treatments: Development of novel therapies, including anti-craving medications and neurobiological-targeted treatments, is enhancing patient outcomes.
- Government Initiatives and Support Programs: Public health campaigns, rehabilitation programs, and favorable policies are encouraging treatment adoption and market growth.
- High Unmet Medical Need: Limited efficacy and adherence issues with existing therapies are driving demand for more effective, long-term, and personalized treatment options.
DelveInsight's ‘Alcohol Use Disorder Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the AUD, historical and forecasted epidemiology, as well as the AUD therapeutics market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The AUD market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, AUD patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in AUD and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Scope of the Alcohol Use Disorder Market | |
|
Study Period |
2022 to 2036 |
|
Forecast Period |
2026-2036 |
|
Geographies Covered |
|
|
Alcohol Use Disorder Market |
|
|
Alcohol Use Disorders Market Size | |
|
Alcohol Use Disorder Companies |
Calliditas Therapeutics AB, Travere Therapeutics Inc., Omeros Corporation, Novartis Pharmaceuticals, Chinook Therapeutics Inc., Vera Therapeutics Inc., Otsuka Pharmaceutical, and others. |
|
Alcohol Use Disorder Epidemiology Segmentation |
|
Alcohol Use Disorder Understanding
Alcohol Use Disorder Overview and Diagnosis
Alcohol Use Disorder, or alcoholism, is a condition where excessive drinking affects physical health, thinking, and behavior, with individuals continuing to drink despite harmful consequences. It is characterized by loss of control over alcohol use, inability to quit, cravings, increased tolerance, withdrawal symptoms, and neglect of responsibilities. Over time, individuals may become physically and psychologically dependent as the brain adapts to alcohol, leading to withdrawal when intake stops. AUD is linked to serious health, social, and behavioral issues, including liver and heart disease, mental health disorders, accidents, and risky behaviors. It arises from a combination of genetic susceptibility especially with a family history and environmental factors such as social influences, stress, and lack of support.
Alcohol Use Disorders are common but often underrecognized, making routine screening important. Diagnosis is mainly clinical, based on symptoms like loss of control, cravings, continued use despite harm, tolerance, and withdrawal, with severity classified by DSM-5. Tools such as AUDIT and CAGE help identify risky drinking, while blood tests and biomarkers can support assessment and monitor alcohol use or relapse.
Further details are provided in the report...
Alcohol Use Disorder Treatment
AUD treatment combines behavioral therapies, complementary approaches, and medications, with no single method being superior. Cognitive-behavioral therapies help change harmful thoughts and behaviors, while holistic approaches like mindfulness, exercise, and social support aid recovery. Approved medications disulfiram, naltrexone, and acamprosate help reduce drinking or cravings, and some off-label drugs may also be used to support treatment.
Further details related to country-based variations are provided in the report...
Alcohol Use Disorder Unmet Needs
The section “unmet needs of AUD” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Limited efficacy of existing behavioral and pharmacological treatments
- Limited understanding of the neuropsychological basis and complexity of AUD
- Lack of predictive tools for treatment response
- Social stigma and lack of awareness
and others…..
Note: Comprehensive unmet needs insights in AUD and their strategic implications are provided in the full report....
Alcohol Use Disorder Epidemiology
Key Findings from AUD Epidemiological Analysis and Forecast
- According to DelveInsight estimates, there were 25,648,000 12-months diagnosed prevalent cases of AUD in the US in 2025, which are projected to decrease by 2036.
- Among EU4 and the UK, Germany accounted for maximum cases of AUD, followed by France and least in Spain, in 2025.
- The 12-month diagnosed prevalence of AUD in the United States has shown a modest decline in recent years, decreasing from ~10.6% in 2021 to ~9.7% in 2025. This trend aligns with reductions in heavy and binge drinking rates, particularly among young adults, rather than solely cultural or lifestyle shifts.
- AUD affects approximately 70% of males and 30% of females in the EU4 and the UK. Projections for the gender-specific diagnosed prevalence indicate a decrease during the forecast period (2026–2036).
- DelveInsight analysts have categorized the diagnosed cases of AUD into five age groups: 18–29 years, 30–44 years, 45–64 years, and 65+ years, respectively, during the study period (2022–2036). In 2025, the highest number of AUD cases was accounted for the age group 45–64 years, with 8,977,000 cases.
Alcohol Use Disorder Epidemiology Segmentation
- 12-months Diagnosed Prevalent Cases of AUD
- Gender-specific Diagnosed Prevalent Cases of AUD
- Age-specific Diagnosed Prevalent Cases of AUD
- Severity-specific Diagnosed Prevalent Cases of AUD
- Total Treated Cases of AUD
Recent Developments In The Alcohol Use Disorder Treatment Market:
- According to the SEC filing published in September 2025, Adial Pharmaceuticals plans to initiate a Phase III study of AD04 in the first-half of 2026, assuming the availability of adequate funding, the conclusion of ongoing discussions with regulatory authorities, and finalization of the trial design, and the availability of sufficient drug product to carry out the study.
- Additionally, according to Adial Pharmaceuticals’ annual presentation, the company is scheduled to review the study design, protocol, and statistical analysis plan with the FDA on July 29, 2025, and anticipates achieving the potential commercial launch of AD04 by 2027. The company is seeking 505(B)(2) path to regulatory approval for this drug.
- According to a news release in June 2025, a University of California, Los Angeles (UCLA) clinical trial found that ibudilast did not significantly reduce alcohol consumption compared to a placebo; however, the drug demonstrated a positive effect among female participants.
Alcohol Use Disorder Drug Analysis
The AUD drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the AUD treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the AUD market.
Approved Therapies for Alcohol Use Disorder
VIVITROL (naltrexone): Alkermes
VIVITROL is the first and only extended-release medication for the treatment of alcohol dependence. It is a once-monthly, intramuscular injectable medication indicated for patients who are able to abstain from drinking alcohol in an outpatient setting prior to beginning treatment. VIVITROL is a potent opioid antagonist, which is nonaddictive and nonaversive, is administered by a healthcare provider, and should be used in combination with psychosocial support. The proprietary Medisorb(R) drug delivery technology in VIVITROL enables the medication to be gradually released into the body at a controlled rate over 1 month.
Alcohol Use Disorder Pipeline Analysis
AD04 (ondansetron): Adial Pharmaceuticals
AD04 is a genetically targeted therapeutic agent for the treatment of AUD and is currently being investigated in a Phase III clinical trial for the potential treatment of AUD in subjects with certain target genotypes, which are to be identified using the company’s proprietary companion diagnostic genetic test. Adial has already developed a companion diagnostic test to select patients with the specific genomic biomarker that may benefit from AD04. It was successfully used in a large-scale RCT, the ONWARD study, and will be advanced in future clinical studies, to support FDA approval and commercial availability at the time of AD04’s launch.
Brenipatide (LY3537031): Eli Lilly and Company
Brenipatide is a biologic entity that acts as an agonist of both glucose-dependent insulinotropic polypeptide (GIP) receptor and the GLP-1 receptor and is being studied for the treatment of AUD. Eli Lilly has introduced Brenipatide for AUD, opioid dependence, and asthma, marking a strategic move beyond obesity treatment. It is currently being evaluated in Phase III of clinical trials.
Comparison of Alcohol Use Disorder Emerging Drugs Under Development | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
AD04 (ondansetron) |
Adial Pharmaceuticals |
III |
Moderate-to-severe AUD 18 years and older (adult, older adult) |
Oral |
Serotonin-3 5-Hydroxytryptamine Type 3 (5HT3) receptor antagonist |
Small molecule |
2028 |
|
Brenipatide (LY3537031) |
Eli Lilly and Company |
III |
Moderate-to-severe AUD (Adult) |
Subcutaneous |
Dual GIP/ Glucagon-like peptide-1 (GLP-1) receptor agonist |
Peptide-based biologic |
Information is available in the full report |
|
MN-166 (ibudilast) |
MediciNova |
II |
Alcohol dependence 18–70 years (adult, older adult) |
Oral |
Inhibits PDE4 and inflammatory cytokines, including macrophage Migration Inhibitory Factor (MIF) |
Small molecule |
Information is available in the full report |
|
Sunobinop (V117957) |
Imbrium Therapeutics (subsidiary of Purdue Pharma) |
II |
Moderate-to-severe AUD 18 years and older (adult, older adult) |
Oral |
Activate the nociceptin/orphanin-FQ peptide (NOP) receptor |
Small molecule |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Alcohol Use Disorder Market Outlook
AUD is a common and complex condition, with treatment goals ranging from abstinence to harm reduction. Management typically combines behavioral therapies such as CBT, motivational approaches, and 12-step programs with medications like disulfiram, naltrexone, and acamprosate, while agents like topiramate and gabapentin are used off-label. As no single therapy is clearly superior, research continues to refine treatment strategies; however, low treatment uptake and adherence highlight the need for more effective, accessible, and personalized care.
The AUD treatment market remains largely dependent on established pharmacotherapies with modest efficacy, yet is poised for potential disruption from emerging metabolic and neurological approaches. Current branded options, including VIVITROL, continue to generate steady demand within the non-addictive medication-assisted treatment (MAT) segment. However, the development pipeline is increasingly focused on novel mechanisms, particularly GIP/GLP-1 receptor agonists such as brenipatide that are gaining attention for their potential to reduce cravings and alcohol consumption, supported by growing clinical and real-world evidence.
The AUD treatment market includes a broad range of off-label and generic therapies, such as topiramate (TOPAMAX), gabapentin, benzodiazepines, memantine, ondansetron (ZOFRAN), varenicline, aripiprazole, and suvorexant, reflecting efforts to address unmet clinical needs beyond approved options. Among these, topiramate has gained recognition and is endorsed by the National Institute on Alcohol Abuse and Alcoholism.
The US AUD market remains significantly underpenetrated despite a large patient population. Existing treatments are low-cost generics, limiting branded market value. Pharmacotherapy uptake is extremely low, with very few patients receiving approved medications underscoring a substantial treatment gap.
- It is notably seen that the US AUD market is expected to grow from USD 1,100 million in 2025 and about to increase by 2036.
- Among all the therapies in the US, VIVITROL (naltrexone) holds the highest market share in 2025, while by 2036, AD04 is projected to capture the largest market.
Alcohol Use Disorder Competitive Landscape
The competitive landscape of the alcohol use disorder (AUD) market is moderately fragmented and evolving, with a mix of global pharmaceutical companies, biotech firms, and healthcare service providers. Key players such as AbbVie, Alkermes, Otsuka Pharmaceutical, Teva Pharmaceutical Industries, and Lundbeck maintain a strong presence through established medication-assisted treatments like naltrexone, acamprosate, and disulfiram.
The market is characterized by a dual structure, combining pharmacological therapies with behavioral and support-based interventions, including rehabilitation programs and community organizations, which play a crucial role in patient management.
Competition is increasingly shifting toward innovation and pipeline development, with emerging companies focusing on novel mechanisms such as genetically targeted therapies, neurobiological treatments, and psychedelic-based approaches. Several candidates, including AD04, BPL-003, and Ibudilast, are advancing through clinical trials, intensifying competition.
Additionally, digital health integration including telemedicine, mobile apps, and virtual therapy platforms is reshaping the competitive dynamics by improving treatment access and patient adherence.
Key Alcohol Use Disorder Companies
The Key Alcohol Use Disorder companies actively involved in the Alcohol Use Disorder treatment landscape include -
- Adial Pharmaceuticals
- Eli Lilly and Company
- MediciNova
- Imbrium Therapeutics (subsidiary of Purdue Pharma)
- Tempero Bio and Nxera Pharma
- Altimmune, and others
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Alcohol Use Disorder (2022–2036 Forecast)
The AUD market comprises targeted small molecules, and Peptide-based biologic alongside conventional and off-label therapies, each addressing distinct immunologic pathways and mechanisms underlying AUD.
- Targeted small molecules: VIVITROL is an extended-release opioid receptor antagonist that supports treatment of Alcohol Use Disorder by blocking the rewarding effects of alcohol, reducing cravings, and improving adherence through once-monthly dosing highlighting the clinical value of mechanism-driven, long-acting pharmacotherapy in addiction management.
- Small molecules: AD04 (ondansetron) by Adial Pharmaceuticals is a serotonin-3 (5-HT3) receptor antagonist being developed for Alcohol Use Disorder, aiming to reduce alcohol consumption and cravings in genetically defined patient subgroups highlighting the potential of precision, mechanism-driven therapies in addiction treatment.
Further details will be provided in the report….
Alcohol Use Disorder Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the AUD drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
Detailed insights of emerging therapies' drug uptake is included in the report...
Alcohol Use Disorder Market Access and Reimbursement of Approved therapies
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
AUD therapies Price Scenario & Trends
Pricing and analogue assessment of AUD therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most approproiate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of AUD Approved Drugs
VIVITROL (naltrexone), approved only in the US, is administered via intramuscular injection. The recommended dose is 380 mg given as a deep gluteal injection every four weeks or once a month. The wholesale acquisition cost (WAC) per monthly dose is USD 1,309, resulting in an estimated six-month treatment cost of approximately USD 8,840.
Industry Experts and Physician Views for Alcohol Use Disorder
To keep up with AUD market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the AUD emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in AUD, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights at country level. Centers such as the University of California, Imperial College London, Hokkaido University, and Cholangiocarcinoma Foundation, etc. were contacted. Their opinion helps understand and validate current and emerging AUD therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in AUD.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Several medications including topiramate, gabapentin, baclofen, ondansetron, and varenicline have been used off-label to manage Alcohol Use Disorder. While some patients show positive responses, the overall efficacy of these agents remains limited. To date, none have consistently and significantly outperformed placebo in reducing AUD symptoms, underscoring the need for more robust and targeted pharmacological options in the treatment landscape.” |
|
Italy |
“The negative effects of alcohol use can transmit intergenerational harm if AUD occurs during pregnancy and/or while parenting a child. Prenatal alcohol exposure is the leading preventable cause of congenital anomalies in the US, and heavy drinking in women has been on the rise, further accelerated by the COVID-19 pandemic.” |
Alcohol Use Disorder Report Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of AUD, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided..
Scope of the Alcohol Use Disorder Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of AUD, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the AUD market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM AUD market.
Alcohol Use Disorder Market Report Insights
- Alcohol Use Disorder Patient Population Forecast
- Alcohol Use Disorder Therapeutics Market Size
- Alcohol Use Disorder Pipeline Analysis
- Alcohol Use Disorder Market Size And Trends
- Alcohol Use Disorder Market Opportunity (current and forecasted)
Alcohol Use Disorder Market Report Key Strengths
- Epidemiology-based (Epi-based) bottom-up forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- Alcohol Use Disorder market outlook (North America, Europe, Asia-Pacific)
- Patient Burden trends (by geography)
- Alcohol Use Disorder Treatment addressable Market (TAM)
- Alcohol Use Disorder Competitve Landscape
- Alcohol Use Disorder major companies Insights
- Alcohol Use Disorder price trends and analogue assessment
- Alcohol Use Disorder therapies and Drug Adoption/Uptake
- Alcohol Use Disorder therapies Peak Patient Share Analysis
Alcohol Use Disorder Market Report Assessment
- Alcohol Use Disorder Current treatment practices
- Alcohol Use Disorder Unmet needs
- Alcohol Use Disorder Clinical development Analysis
- Alcohol Use Disorder Emerging drugs product profiles
- Alcohol Use Disorder Market attractiveness
- Alcohol Use Disorder Qualitative analysis (SWOT and conjoint analysis)
- Alcohol Use Disorder Market Drivers
- Alcohol Use Disorder Market Barriers
FAQs Related To Alcohol Use Disorder Market Report:
Alcohol Use Disorder Market Insights
- What was the AUD market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of AUD?
- What are the disease risks, burdens, and unmet needs of AUD? What will be the growth opportunities across the 7MM concerning the patient population with AUD?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of AUD? What are the current guidelines for treating AUD in the US, Europe, and Japan?
Reasons to Buy Alcohol Use Disorder Market Forecast Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the AUD market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets withing the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.