Castrate-Sensitive Prostate Cancer Market Summary
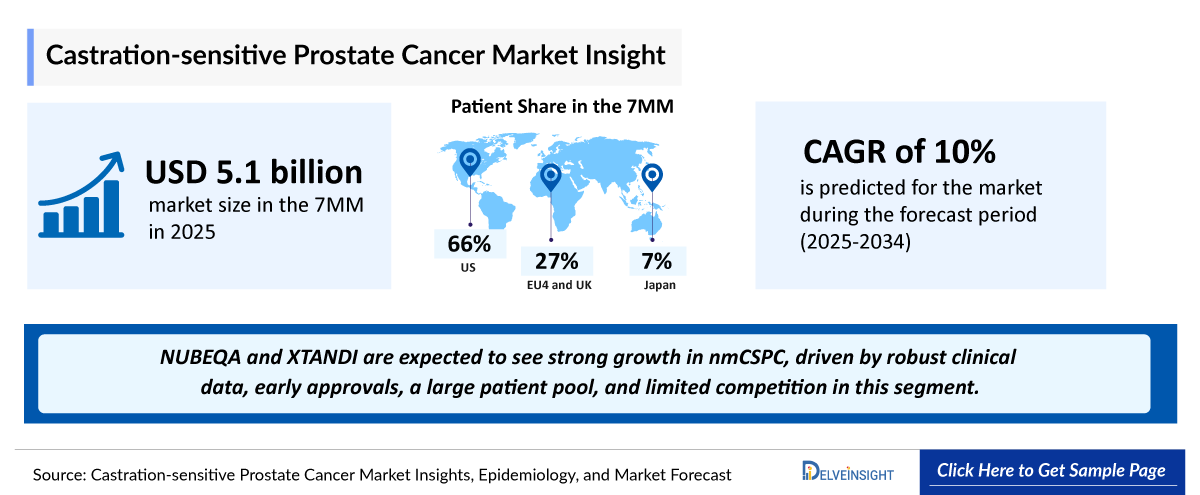
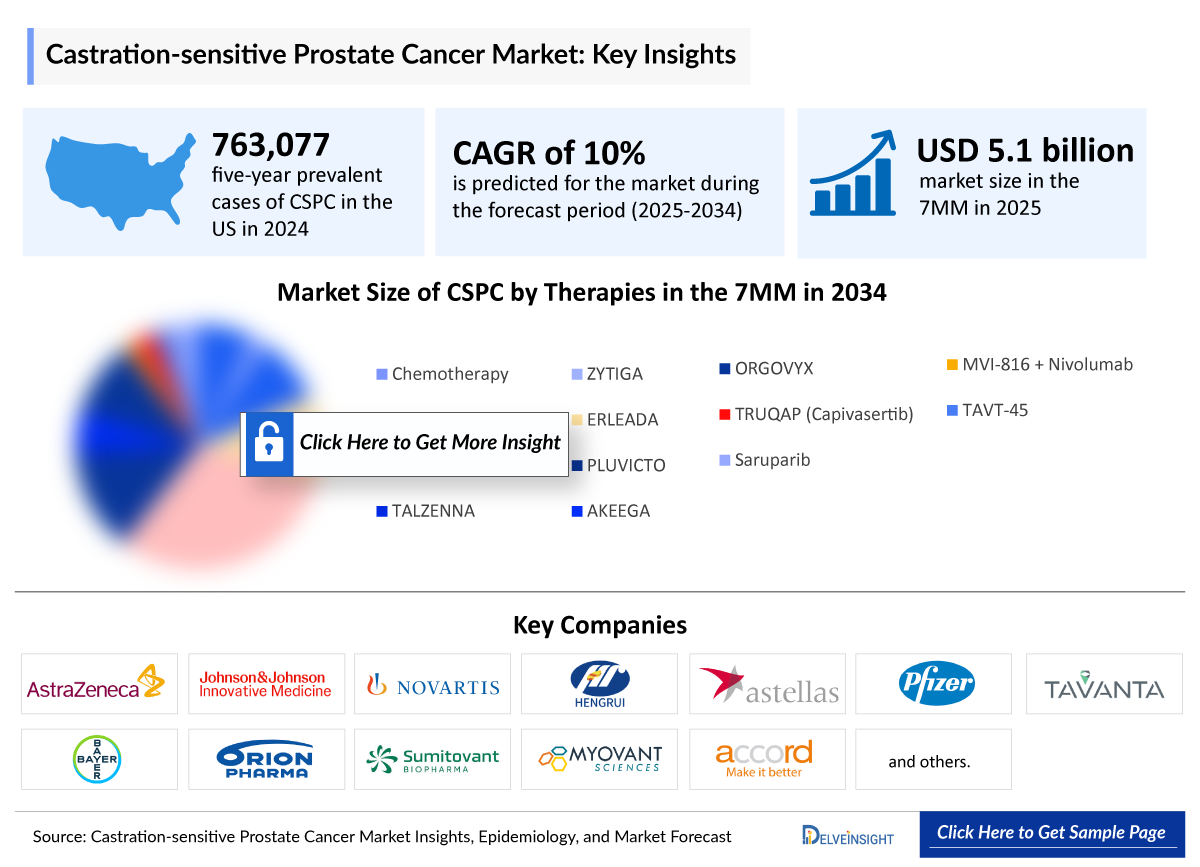
- The Castrate-Sensitive Prostate Cancer market in the 7MM was USD 5143 million in 2025 and projected to reach USD 11753 million by 2034.
- The Castrate-Sensitive Prostate Cancer market is projected to grow at a CAGR of 10% by 2034 in leading countries (US, EU4, UK and Japan)
Castrate-Sensitive Prostate Cancer (CSPC) Market and Epidemiology Analysis
- In 2024, the total market size of CSPC therapies was USD 415 million in the United States which is expected to increase in the study period (2024–2034).
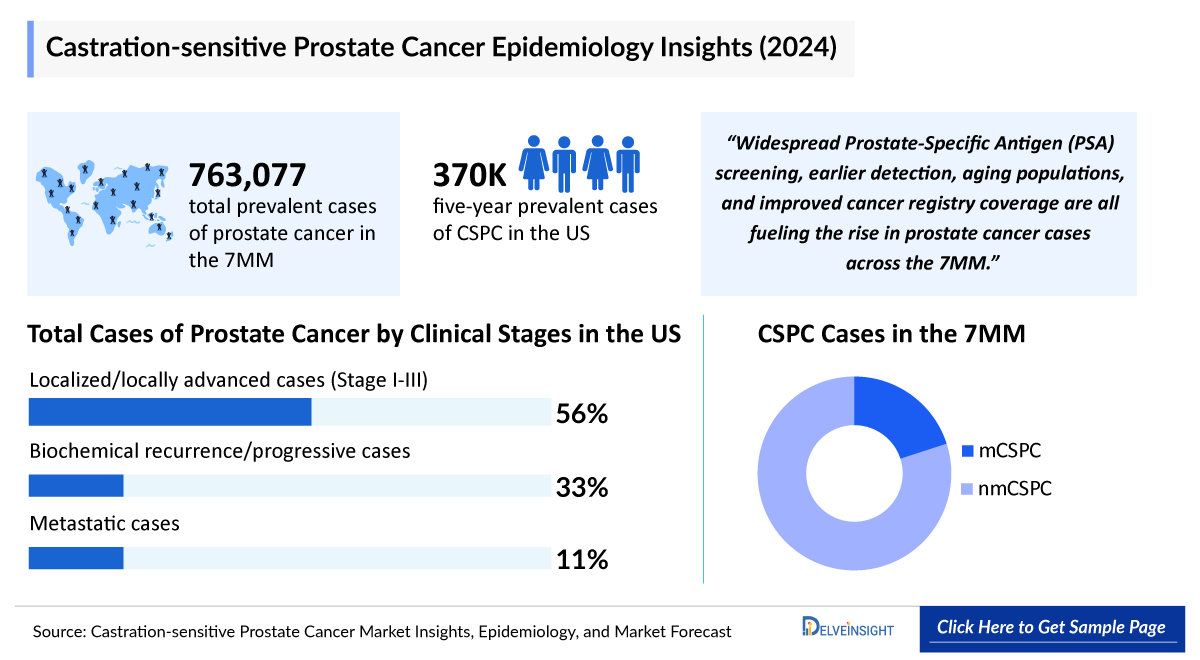
- The total cases of CSPC in the 7MM were found to be 763,077 in 2024 which is expected to grow during the study period.
- The total prevalent cases of CSPC in the 7MM were found to be 935,398 in 2025 which is expected to grow during the study period.
- On June 3, 2025, the Food and Drug Administration (FDA) approved darolutamide (Nubeqa, Bayer Healthcare Pharmaceuticals Inc.) for metastatic castration-sensitive prostate cancer (mCSPC). The FDA previously approved darolutamide in combination with docetaxel for mCSPC.
 Castrate-Sensitive Prostate Cancer Market size and forecast
Castrate-Sensitive Prostate Cancer Market size and forecast
- 2025 Castrate-Sensitive Prostate Cancer Market Size: USD 5143 million
- 2034 Projected Castrate-Sensitive Prostate Cancer Market Size: USD 11753 million
- Castrate-Sensitive Prostate Cancer Growth Rate (2025-2034): 10% CAGR
- Largest Castrate-Sensitive Prostate Cancer Market: United States
DelveInsight’s ‘Castrate-Sensitive Prostate Cancer (CSPC) Market Insights, Epidemiology and Market Forecast - 2034’ report delivers an in-depth understanding of the CSPC, historical and forecasted epidemiology as well as the CSPC market trends in the United States, EU5 (Germany, France, Italy, Spain, and United Kingdom), and Japan.
The CSPC market report provides current treatment practices, emerging drugs, CSPC market share of the individual therapies, current and forecasted CSPC market size from 2024 to 2034 segmented by seven major markets. The Report also covers current CSPC treatment practice/algorithm, market drivers, market barriers and unmet medical needs to curate best of the opportunities and assesses underlying potential of the market
Scope of the Castrate-Sensitive Prostate Cancer Market | |
|
Study Period |
2020–2034 |
|
Forecast Period |
2024–2034 |
|
Geographies Covered |
US, EU4 (Germany, France, Italy, and Spain) and the UK, and Japan |
|
Castrate-Sensitive Prostate Cancer Epidemiology |
|
|
Castrate-Sensitive Prostate Cancer Market |
|
|
Castrate-Sensitive Prostate Cancer Market Analysis |
|
|
Castrate-Sensitive Prostate Cancer Market players |
|
Castrate-Sensitive Prostate Cancer Disease Understanding
CSPC Overview
Prostate cancer is a type of malignancy that occurs in the prostate gland. It is one of the most common types of cancer found in men. This cancer usually grows slowly and is confined to the prostate gland initially, where it may not cause serious harm. According to the Cancer Treatment Centres of America (CTCA), more than 99% of prostate cancers are adenocarcinomas, which develop in the gland cells. The exact cause of prostate cancer is unknown.
However, several things can increase the risk of developing this condition like age, family history, diet, high testosterone level, genome changes, and race.
In the majority of the cases, the symptoms of prostate cancer are not apparent in the early stages of the disease. The symptoms of prostate cancer may be different for each man, and other conditions may cause some of these symptoms like blood in semen, erectile dysfunction, painful ejaculation, pain in pelvic area, and numbness in legs.
“Patients who have never received i.e. are sensitive to ADT known as hormone-sensitive prostate cancer (HSPC) or castrate-sensitive prostate cancer (CSPC). And it was accessed that around 78% of biochemical recurrence/ progressive cases are non-metastatic CSPC/ HSPC.”
CSPC Diagnosis
Patients undergo regular prostate cancer screening before symptoms appear. Screening may involve one or more of the following tests: prostate-specific antigen (PSA), digital rectal exam (DRE), prostate ultrasound, prostate MRI, prostate Mp-MRI.
CSPC Treatment
Prostate cancer is the second most common cancer in men can be categorized into castrate-sensitive (castration-sensitive prostate cancer [CSPC], and castration-resistant prostate cancer (mCRPC). This type of cancer only considered to be CSPC if it responds to medical or surgical treatment to lower a patient’s testosterone levels. For the treatment of CSPC, Androgen-deprivation therapy (ADT) has been the cornerstone of therapy. However, most metastatic CSPC ( mCSPC/HSPC ) cannot be cured by ADT, and re‐emerges as metastatic castration-resistant prostate cancer (mCRPC). The treatment landscape of CSPC was not evolved until recent years. Docetaxel was the first chemotherapy for metastatic prostate cancer that showed a survival benefit. Since then pharmaceutical industries had indulged themselves into the investigation of new therapies for this type of Prostate Cancer. Lately, developments of androgen receptor targeted therapies have influenced the treatment paradigm of CSPC/HSPC
Get More Insights of this Report @ Prostate Cancer Treatment Market Size
Castrate-Sensitive Prostate Cancer Epidemiology
The CSPC epidemiology division provides the insights about historical and current CSPC patient pool and forecasted trend for each seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the DelveInsight report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings
The total cases of CSPC in the 7MM were found to be 763,077 in 2024 which is expected to grow during the study period, i.e., 2024–2034.
The disease epidemiology covered in the report provides historical as well as forecasted CSPC epidemiology [segmented as Total Prevalent Cases of Prostate Cancer, Total Diagnosed Cases of Prostate Cancer, Age-specific Cases of Prostate Cancer, Total Diagnosed Cases of Prostate Cancer by Clinical Stages, Total Non-metastatic and Metastatic Cases of CSPC, and Total Treated Cases of Non-metastatic and Metastatic CSPC] scenario of CSPC in the 7MM covering United States, EU5 countries (Germany, France, Italy, Spain, and United Kingdom), and Japan from 2024 to 2034.
Country Wise- CSPC Epidemiology
Estimates show that the highest cases of CSPC were in the United States, followed by Germany, France, Japan, and Italy in 2024.
Castrate-Sensitive Prostate Cancer Epidemiology Segmentation
- Total Prevalent Cases of Prostate Cancer
- Total Diagnosed Cases of Prostate Cancer
- Age-specific Cases of Prostate Cancer
- Total Diagnosed Cases of Prostate Cancer by Clinical Stages
- Total Non-metastatic and Metastatic Cases of CSPC
- Total Treated Cases of Non-metastatic and Metastatic CSPC] scenario of CSPC
Castrate-Sensitive Prostate Cancer Market Recent Developments and Breakthroughs
- In September 2024, Ipsen announced that the Phase III CONTACT-02 trial for Cabometyx® and atezolizumab in mCRPC showed a non-significant improvement in overall survival but met the progression-free survival (PFS) endpoint.
- In September 2024, Foundation Medicine received FDA approval for FoundationOne®CDx and Liquid CDx as companion diagnostics for Lynparza® in BRCA-mutated mCRPC.
- In July 2024, the ARANOTE trial showed that NUBEQA® plus ADT significantly improved radiological PFS in metastatic hormone-sensitive castrate-resistant prostate cancer (mHSPC).
- In July 2024, the FDA granted fast-track designation to SYNC-T SV-102 for metastatic castrate-resistant prostate cancer (mCRPC).
- In June 2024, Kangpu Biopharmaceuticals received FDA approval for a Phase II/III trial of KPG-121 with Abiraterone for mCRPC.
- In May 2024, Fusion Pharmaceuticals began the Phase 2 AlphaBreak trial of FPI-2265 in mCRPC patients.
- In April 2024, Astellas Pharma received European approval for XTANDI™ in high-risk biochemical recurrent non-metastatic hormone-sensitive prostate cancer (nmHSPC).
- In April 2024, FibroGen announced positive Phase 1 trial data for FG-3246 in mCRPC.
- On November 16, 2023, the FDA approved enzalutamide (Xtandi) for non-metastatic castration-sensitive prostate cancer (nmCSPC) with high-risk biochemical recurrence.
Castrate-Sensitive Prostate Cancer Drugs Analysis
Drug chapter segment of the CSPC market report encloses the detailed analysis of CSPC marketed drugs and late stage (Phase-III and Phase-II) pipeline drugs. It also helps to understand the CSPC clinical trial details, expressive pharmacological action, agreements and collaborations, approval and patent details, advantages and disadvantages of each included drug and the latest news and press releases.
CSPC Marketed Drugs
Erleada (apalutamide): Janssen Pharmaceuticals
Erleada (apalutamide; Janssen Pharmaceutical) is a next-generation oral androgen receptor (AR) inhibitor that blocks the androgen signaling pathway in prostate cancer cells. It is indicated for the treatment of patients with non-metastatic castration-resistant prostate cancer (nmCRPC) and for the treatment of patients with metastatic castration-sensitive prostate cancer (mCSPC/HSPC).
In January 2020, the European Commission has approved apalutamide (Erleada) for use in combination with androgen deprivation therapy (ADT) for the treatment of adult men with metastatic hormone-sensitive prostate cancer. The EC approval is based on data from the phase III TITAN study, which assessed the addition of apalutamide to ADT in a broad range of patients with mHSPC, regardless of disease volume, prior treatment with docetaxel or staging at initial diagnosis.
Xtandi (Enzalutamide): Astellas Pharma/Pfizer
It is an orally bioavailable, organic, non-steroidal small molecule targeting the androgen receptor (AR) with potential antineoplastic activity. In December 2019, the US FDA granted approval to Xtandi (enzalutamide; Astellas Pharma) for use in the treatment of patients with metastatic CSPC. Prior to this, Xtandi was approved for castration-resistant prostate cancer. This approval was based on findings from the ARCHES clinical trial. The median rPFS rates in the Xtandi were not reached and for placebo arms was 19.4 months, respectively. In addition to this, in ARCHES trial statistically significant improvement in time to initiation of a new antineoplastic therapy with enzalutamide vs. placebo. The most common adverse reactions that occurred more frequently in enzalutamide-treated patients in ARCHES were hot flush, asthenia/fatigue, hypertension, fractures, and musculoskeletal pain.
Products detail in the report…
CSPC Emerging Drugs
Keytruda (pembrolizumab): Merck Sharp & Dohme
Keytruda, also known as pembrolizumab, is a PD-1 blocking antibody. It is product of Merck Sharp & Dohme. It is mainly used for cancers that are advanced, have spread to other parts of the body or are not responding to other treatments. In some cancers, it is only given to patients whose tumors produce high levels of a protein known as PD-L1. It is approved for multiple types of cancer. This is currently being investigated in Phase III clinical trials in combination with Enzalutamide for the treatment of patients affected by mHSPC.
Opdivo (nivolumab): Bristol-Myers Squibb
Opdivo (Nivolumab) by Bristol-Myers Squibb is a human immunoglobulin (Ig) G4 monoclonal antibody directed against the negative immunoregulatory human cell surface receptor PD-1, with immune checkpoint inhibitory and antineoplastic activities. This drug is designed to uniquely harness the body’s own immune system to help restore anti-tumor immune response. By harnessing the body’s own immune system to fight cancer, Opdivo has become an important treatment option across multiple cancers.
BMS is working on assessing the clinical activity seen with Opdivo in combination with in combination with Ipilimumab, Docetaxel, and ADT for mHSPC in Phase II/III clinical trial.
Relugolix: Myovant Sciences
Myovant Sciences also investigating their candidate Relugolix for Androgen-sensitive advanced prostate cancer. A phase III HERO study (NCT03085095; Recruiting) currently ongoing to determine the benefit and safety of Relugolix 120 mg orally once daily for 48 weeks on maintaining serum testosterone suppression to castrate levels (≤ 50 ng/dL [1.7 nmol/L] in participants with androgen-sensitive advanced prostate cancer. This is an international, randomized, open-label, parallel group efficacy and safety study.
Topsalysin (PRX302): Sophiris Bio
Topsalysin (PRX302), an investigative agent of Sophiris Bio for localized prostate cancer. It is first-in-class transmembrane pore-forming protein, was genetically modified to be activated only by enzymatically-active PSA, which is produced in large quantities within the prostate of men with prostate cancer.
ProstAtak: Advantagene
Advantagene is developing ProstAtak for the treatment of patients suffering from prostate cancer. ProstAtak jump-starts the body’s own immune system to detect and destroy recurring cancer cells. The treatment is done together with standard radiation therapy. Last year, Candel Therapeutics (Advantagene) announced that it has completed the enrollment of the "ULYSSES" trial, a Phase II study of ProstAtak for prostate cancer patients choosing Active Surveillance (PrTK04- NCT02768363). However, enrollment is still open for prostate cancer patients choosing radiation therapy as their primary treatment (PrTK03- NCT01436968).
Products detail in the report…
Castrate-Sensitive Prostate Cancer Market Outlook
Key Findings
The market size of CSPC in the 7MM countries is expected to increase by 2034 from USD 638 million in 2024, which is expected to increase during study period (2024–2034).
The United States Castrate-Sensitive Prostate Cancer Market Outlook
In 2024, the total market size of CSPC therapies was USD 415 million in the United States which is expected to increase in the study period (2024–2034).
EU-5 Countries Castrate-Sensitive Prostate Cancer Market Outlook
In 2024, the total market size of CSPC therapies was USD 180 million in the EU-5 countries which is expected to increase in the study period (2024–2034).
Japan Castrate-Sensitive Prostate Cancer Market Outlook
The total market size of CSPC therapies in Japan was USD 43 million in 2024.
Castrate-Sensitive Prostate Cancer Pipeline Development Activities
The drugs which are in pipeline include:
- Keytruda (Pembrolizumab/MK-3475) + Enzalutamide + ADT: Phase III
- Opdivo (nivolumab) ± ADT ± docetaxel ± Ipilimumab: Phase II/III
- Relugolix: Phase III
- ProstAtak (AdV-tk + valacyclovir): Phase III
- PRX302 (topsalysin): Phase II
- RV001: Phase III
Product details and complete list of emerging products will be provided in the full report...
CSPC Drugs Uptake
- In mCSPC, currently market is dominated by hormonal therapies, mainly the first-generation hormonal therapies.
- Erleada owing to better clinical profile expected to generate the maximum sales within the metastatic CSPC.
- Like mCSPC, in nmCSPC/HSPC also market is also mainly dominated by first generation hormonal therapies.
CSPC Market Access and Reimbursement
- Health technology assessment (HTA) agencies are evaluating the drugs based on segments they are targeting. In case of CSPC setting, drugs will be compared against the cheaper hormonal therapies especially first-generation therapies.
- And it will be challenging for the drugs to show add on benefit compared to first generation hormonal therapies in given price. And unavailability of precedent in this setting makes it even more difficult to predict the HTA agencies decisions.
Latest KOL Views on CSPC
To keep up with current market trends, we take KOLs and SME’s opinion working in CSPC domain through primary research to fill the data gaps and validate our secondary research. Their opinion helps to understand and validate current and emerging therapies treatment patterns or CSPC market trend. This will support the clients in potential upcoming novel treatment by identifying the overall scenario of the market and the unmet needs.
CSPC Competitive Intelligence Analysis
We perform Competitive and Market Intelligence analysis of the CSPC Market by using various Competitive Intelligence tools that includes – SWOT analysis, PESTLE analysis, Porter’s five forces, BCG Matrix, Market entry strategies etc. The inclusion of the analysis entirely depends upon the data availability.
Scope of the CSPC Market Report
- The report covers the descriptive overview of CSPC, explaining its causes, signs and symptoms, pathophysiology, and currently available therapies.
- Comprehensive insight has been provided into the CSPC epidemiology and treatment in the 7MM.
- Additionally, an all-inclusive account of both the current and emerging therapies for CSPC is provided, along with the assessment of new therapies, which will have an impact on the current treatment landscape.
- A detailed review of CSPC market; historical and forecasted is included in the report, covering drug outreach in the 7MM.
- The report provides an edge while developing business strategies, by understanding trends shaping and driving the global CSPC market.
CSPC Market Report Highlights
- In the coming years, CSPC market is set to change due to the rising awareness of the disease and incremental healthcare spending across the world; which would expand the size of the market to enable the drug manufacturers to penetrate more into the market.
- The companies and academics are working to assess challenges and seek opportunities that could influence CSPC R&D. The therapies under development are focused on novel approaches to treat/improve the disease condition.
- Major players are involved in developing therapies for CSPC. Launch of emerging therapies, will significantly impact the CSPC market.
- A better understanding of disease pathogenesis will also contribute to the development of novel therapeutics for CSPC.
- Our in-depth analysis of the pipeline assets across different stages of development (Phase III and Phase II), different emerging trends and comparative analysis of pipeline products with detailed clinical profiles, key cross-competition, launch date along with product development activities will support the clients in the decision-making process regarding their therapeutic portfolio by identifying the overall scenario of the research and development activities.
Castrate-Sensitive Prostate Cancer Market Report Insights
- Patient Population
- Therapeutic Approaches
- CSPC Pipeline Analysis
- CSPC Market Size and Trends
- Market Opportunities
- Impact of upcoming Therapies
Castrate-Sensitive Prostate Cancer Market Report Key Strengths
- 11 Years Forecast
- 7MMCoverage
- CSPC Epidemiology Segmentation
- Key Cross Competition
- Highly Analyzed Market
- Drugs Uptake
Castrate-Sensitive Prostate Cancer Market Report Assessment
- SWOT Analysis
- Current Treatment Practices
- Unmet Needs
- Pipeline Product Profiles
- Conjoint Analysis
- Castrate-Sensitive Prostate Cancer Market Attractiveness
- Castrate-Sensitive Prostate Cancer Market Drivers
- Castrate-Sensitive Prostate Cancer Market Barriers
Key Questions Answered in the Castrate-Sensitive Prostate Cancer Market Report
Castrate-Sensitive Prostate Cancer Market Insights:
- What was the CSPC Market share (%) distribution in 2024 and how it would look like in 2034?
- What would be the CSPC total market Size as well as market size by therapies across the 7MM during the study period (2024–2034)?
- What are the key findings pertaining to the market across the 7MM and which country will have the largest CSPC market size during the study period (2024–2034)?
- At what CAGR, the CSPC market is expected to grow in 7MM during the study period (2024–2034)?
- What would be the CSPC market outlook across the 7MM during the study period (2024–2034)?
- What would be the CSPC market growth till 2034 and what will be the resultant market size in the year 2034?
- How would the market drivers, barriers and future opportunities affect the market dynamics and a subsequent analysis of the associated trends?
- CSPC patient types/ pool where unmet need is more and whether emerging therapies will be able to address the residual unmet need?
- How emerging therapies are performing on the parameters like efficacy, safety, route of administration (RoA), treatment duration and frequencies on the basis of their clinical trial results?
- Among the emerging therapies, what are the potential therapies which are expected to disrupt the CSPC market?
Castrate-Sensitive Prostate Cancer Epidemiology Insights:
- What is the disease risk, burden, and unmet needs of the CSPC?
- What is the historical CSPC patient pool in seven major markets covering the United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom) and Japan?
- What would be the forecasted patient pool of CSPC in 7 major markets covering the United States, EU5 (Germany, France, Italy, Spain, and the United Kingdom) and Japan?
- What will be the growth opportunities in the 7MM with respect to the patient population pertaining to CSPC?
- Out of all the 7MM countries, which country would have the highest prevalent population of CSPC during the study period (2024–2034)?
- At what CAGR the population is expected to grow in the 7MM during the study period (2024–2034)?
- What are the various recent and upcoming events which are expected to improve the diagnosis of CSPC?
Current Treatment Scenario, Marketed Drugs and Emerging Therapies:
- What are the current options for the treatment of CSPC along with the approved therapy?
- What are the current treatment guidelines for the treatment of CSPC in the US, Europe and Japan?
- What are the CSPC marketed drugs and their MOA, regulatory milestones, product development activities, advantages, disadvantages, safety and efficacy, etc.?
- How many companies are developing therapies for the treatment of CSPC?
- How many therapies are developed by each company for the treatment of CSPC?
- How many emerging therapies are in mid stage, and late stage of development for the treatment of CSPC?
- What are the key collaborations (Industry–Industry, Industry–Academia), Mergers and acquisitions, licensing activities related to the CSPC therapies?
- What are the recent novel therapies, targets, mechanisms of action and technologies developed to overcome the limitation of existing therapies?
- What are the clinical studies going on for CSPC and their status?
- What are the key designations that have been granted for the emerging therapies for CSPC?
- What is the global historical and forecasted market of CSPC?
Reasons to Buy CSPC Report
- The report will help in developing business strategies by understanding trends shaping and driving the CSPC market.
- To understand the future market competition in the CSPC market and Insightful review of the key market drivers and barriers.
- Organize sales and marketing efforts by identifying the best opportunities for CSPC in the US, Europe (Germany, France, Italy, Spain, and the United Kingdom) and Japan.
- Identification of strong upcoming players in market will help in devising strategies that will help in getting ahead of competitors.
- Organize sales and marketing efforts by identifying the best opportunities for CSPC market.
- To understand the future market competition in the CSPC market.
Stay Updated with us for Recent Articles