Food Allergy Market Summary
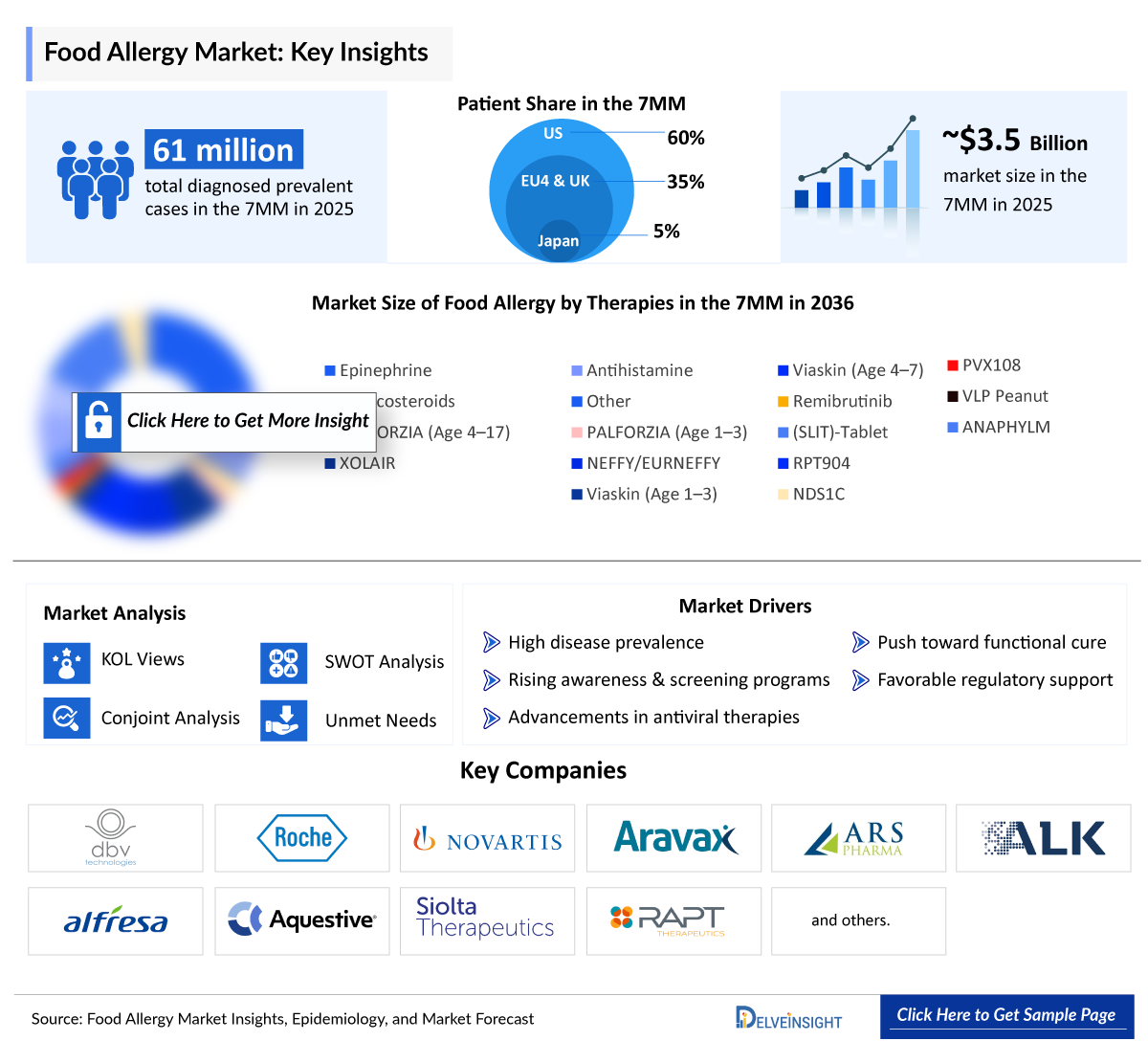
- The Food Allergy market size was valued at approximately USD 3,500 million in 2025 and is projected to reach nearly USD 10,500 million by 2036, growing at a CAGR of 9% during 2026–2036.
- The leading Food Allergy companies developing therapies in the market include - Novartis, Roche, ARS Pharmaceuticals, ALK-Abelló , Alfresa, Stallergenes Greer, DBV Technologies, Aravax, Aquestive Therapeutics, and others.
Food Allergy Insights and Trends
- A significant proportion of the food allergy burden is associated with comorbid atopic conditions such as Asthma, Atopic Dermatitis, and Allergic Rhinitis, along with pediatric predisposition and genetic factors, highlighting the need for integrated disease management, early diagnosis, and preventive strategies to reduce severe reactions and improve patient outcomes.
- Epinephrine is the first-line treatment for anaphylaxis. Antihistamines, corticosteroids, and bronchodilators may also be used, but they do not treat anaphylaxis itself and serve only as adjunctive therapies.
- Desensitization improves food allergy care by raising reaction thresholds, reducing accidental anaphylaxis risk, and enhancing patient protection and confidence.
- Currently, omalizumab (XOLAIR), epinephrine nasal spray (NEFFY), and peanut allergen powder (PALFORZIA) are the only approved medications offering important treatment options for patients. Stallergenes Greer will discontinue the commercialization of PALFORZIA as of July 31, 2026. This voluntary discontinuation is not related to product safety, quality, or efficacy.
- The evolving food allergy treatment landscape features clinical trials led by DBV Technologies (Viaskin [DBV712] peanut patch), Aravax (PVX108), Aquestive Therapeutics (ANAPHYLM (epinephrine sublingual film), and others.
Food Allergy Market Size and Forecast in the 7MM
- 2025 Food Allergy Market Size: ~USD 3,500 million
- 2036 Projected Food Allergy Market Size: USD ~10,500 million
- Food Allergy Growth Rate (2026–2036): ~9% CAGR
Request a sample to unlock the CAGR for "Food Allergy Market Forecast"
Key Factors Driving the Food Allergy Market
- Rising Food Allergy Prevalence: The Rising food allergy prevalence, particularly among males, is a key driver of market growth. In 2025, the 7MM reported approximately 61 million diagnosed prevalent cases.
- Rising Opportunities in Targeted Food Allergy Therapies: Emerging evidence of improved safety, tolerability, and potential disease-modifying effects of novel immunotherapies over traditional allergen avoidance strategies highlights a growing opportunity for drug developers to focus on approaches that induce immune tolerance, reduce the risk of severe reactions, and provide long-term protection in food allergy patients.
- Emerging Food Allergy Competitive Landscape: The evolving food allergy pipeline is a key market driver, with increasing focus on immunotherapies and novel desensitization approaches. Promising candidates such as DBV712, PVX108 (AVX-201), and ANAPHYLM are advancing through clinical trials, supporting future market growth.
DelveInsight's ‘Food Allergy Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the Food Allergy, historical and forecasted epidemiology, as well as the Food Allergy therapeutics market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The Food Allergy market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates Food Allergy patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in Food Allergy and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
Scope of the Food Allergy Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Food Allergy Market CAGR (Study period/Forecast period) |
~9% (2026–2036) |
|
Food Allergy Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Food Allergy Companies |
|
|
Food Allergy Therapies |
|
|
Food Allergy Market |
Segmented by
|
|
Analysis |
|
Food Allergy Disease Understanding
Food Allergy Overview
Food allergy is an immune-mediated condition that occurs when the body mistakenly identifies certain food proteins as harmful, triggering an abnormal immune response. It can range from mild to severe, with symptoms such as itching, hives, swelling, abdominal pain, vomiting, and diarrhea, and in severe cases, anaphylaxis, which may involve difficulty breathing, a drop in blood pressure, and can be life-threatening. Food allergy is primarily caused by exposure to common allergens such as peanuts, tree nuts, milk, eggs, wheat, soy, fish, and shellfish. It typically develops due to a combination of genetic and environmental factors.
Food Allergy Diagnosis
Food allergy is diagnosed primarily through clinical evaluation and detailed patient history, focusing on reproducible symptoms such as urticaria, gastrointestinal distress, respiratory difficulty, or anaphylaxis following food exposure. Diagnostic tests, including skin prick testing and serum-specific immunoglobulin E (IgE) measurements, help confirm sensitization, while oral food challenge remains the gold standard for definitive diagnosis.
Further details are provided in the report...
Food Allergy Treatment
The primary goals of food allergy treatment are to prevent allergic reactions, reduce the risk of severe outcomes such as anaphylaxis, improve patient quality of life, and, where possible, induce long-term immune tolerance. Management is largely centered on strict allergen avoidance and patient education, including careful label reading and prevention of cross-contamination. Emergency preparedness is essential, with epinephrine (e.g., EpiPen, AUVI-Q) as the first-line treatment for anaphylaxis, supported by adjunctive therapies such as antihistamines (e.g., cetirizine, diphenhydramine) and corticosteroids for symptom control.
For long-term management, immunotherapy approaches are increasingly adopted in a step-wise manner. Oral immunotherapy (OIT), such as PALFORZIA, is used to achieve desensitization through controlled allergen exposure. Emerging alternatives include epicutaneous immunotherapy (EPIT) like Viaskin Peanut Patch (DBV712) and sublingual immunotherapy (SLIT), which offer improved safety profiles. In patients with severe or multiple food allergies, biologic therapies such as XOLAIR may be considered to target IgE-mediated pathways and enhance treatment outcomes. Supportive strategies, including dietary modification, regular monitoring, and management of comorbid allergic conditions, remain critical to ensure sustained protection and reduce the risk of recurrence.
Further details related to country-based variations are provided in the report.
Food Allergy Unmet Needs
The section “unmet needs of Food Allergy” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of curative therapies; current management relies on avoidance
- Risk of severe reactions (anaphylaxis) and unpredictable triggers
- Limitations in diagnostic accuracy and standardization, and others…..
Comprehensive unmet needs insights in Food Allergy and their strategic implications are provided in the full report.
Food Allergy Epidemiology
The Food Allergy epidemiology section provides insights about the historical and current Food Allergy patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the Food Allergy market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings from Food Allergy Epidemiological Analysis and Forecast
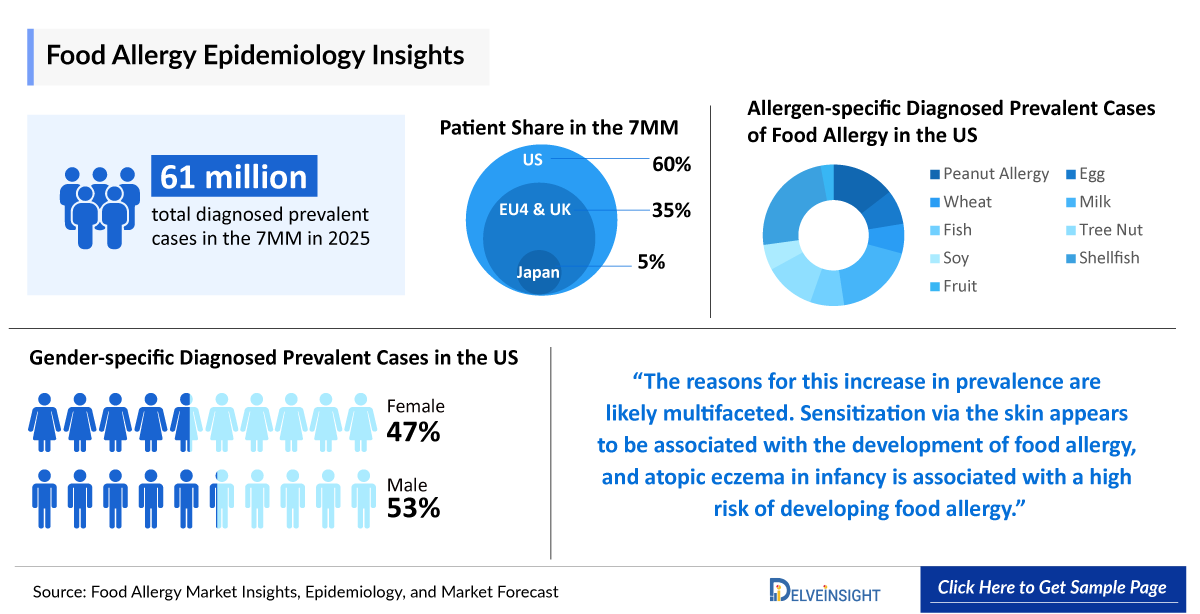
- According to DelveInsight’s estimates, the total diagnosed prevalent cases of Food Allergy in the 7MM were approximately 61,000,000 in 2025.
- Among the EU4 and the UK, Spain accounted for the highest number of diagnosed prevalent cases of Food Allergy, followed by Germany, whereas France accounted for the lowest number of cases in 2025.
- In 2025, among gender-specific cases of food allergy in 7 million individuals, males accounted for 53% while females made up 47%.
- In Japan in 2025, egg allergy had the highest diagnosed prevalence among food allergens, followed by milk allergy, while fish allergy had the lowest prevalence.
Food Allergy Epidemiology Segmentation
- Total Diagnosed Prevalent Cases of Food Allergy
- Allergen-specific Diagnosed Prevalent Cases of Food Allergy
- Gender-Specific Diagnosed Prevalent Cases of Food Allergy
- Severity-specific Diagnosed Prevalent Cases of Food Allergy
- Total treated cases of Food Allergy
Recent Developments in the Food Allergy Treatment Landscape
- In February 2026, Aquestive Therapeutics announced that it received a Complete Response Letter (CRL) from the FDA on January 30, 2026, for the New Drug Application (NDA) seeking approval of ANAPHYLM (dibutepinephrine) Sublingual Film for the treatment of Type I allergic reactions, including anaphylaxis, in patients weighing 30kg or more (approximately 66 pounds).
- In February 2026, according to Novartis’ Q4 2025 presentation, initiation of the Phase III program for remibrutinib is anticipated in the second half of 2026.
- In August 2025, Stallergenes Greer announced to voluntarily discontinue PALFORZIA by July 31, 2026, not due to safety, quality, or efficacy concerns. The company will fulfill regulatory obligations and support a smooth transition for patients and healthcare providers.
Food Allergy Drug Chapters & Competitive Analysis
The Food Allergy drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase I–III Food Allergy clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Food Allergy treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Food Allergy therapeutics market.
Approved Therapies for Food Allergy
Omalizumab (XOLAIR): Novartis/ Roche
XOLAIR, developed by Novartis and Roche, is a monoclonal anti-IgE antibody that targets the underlying allergic pathway by binding to free IgE and preventing its interaction with receptors on mast cells and basophils. Initially approved for allergic asthma and chronic spontaneous urticaria, XOLAIR received US FDA approval in 2024 for the treatment of food allergy to reduce the risk of allergic reactions, including anaphylaxis, following accidental exposure to one or more foods. By lowering circulating IgE levels, it helps decrease hypersensitivity responses and provides a novel, mechanism-driven approach for food allergy management, reflecting its expanded clinical utility and significance in addressing an area of high unmet need.
Epinephrine /Adrenaline (NEFFY/ EURNEFFY): ARS Pharmaceuticals /ALK Abelló /Alfresa Pharma
NEFFY/EURNEFFY, developed by ARS Pharmaceuticals and partnered with ALK-Abelló and Alfresa Pharma for global commercialization, is a needle-free, intranasal formulation of epinephrine designed for the emergency treatment of allergic reactions, including anaphylaxis associated with food allergy. As a fast-acting sympathomimetic agent, epinephrine works by constricting blood vessels, relaxing airway muscles, and reducing swelling, thereby rapidly reversing life-threatening symptoms. NEFFY has gained regulatory approvals in key markets, including the US and Europe, offering a convenient and user-friendly alternative to traditional injectable auto-injectors, potentially improving adherence and timely administration during acute allergic emergencies.
Food Allergy Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
RoA |
MoA |
Marketed Region |
|
Omalizumab (XOLAIR) |
Novartis/ Roche |
Food allergy |
Monoclonal antibody |
SC |
Target and block IgE |
US: 2024 Europe: 2024 Japan: 2025 |
|
Epinephrine /Adrenaline (NEFFY/ EURNEFFY) |
ARS Pharma, ALK-Abelló, Alfresa Pharma |
Food allergy |
Small molecule |
Intranasal |
Activating both alpha and beta receptors |
US: 2024 Europe: 2024 |
|
Peanut allergen powder-dnfp (PALFORZIA) |
Stallergenes Greer |
Peanut allergy |
Immunotherapy |
Oral |
Immune desensitization |
US: 2020 Europe: 2020 |
Note: Detailed marketed therapies assessment will be provided in the final report...
Food Allergy Pipeline Analysis
Viaskin (DBV712) Peanut Patch: DBV Technologies
DBV712, developed by DBV Technologies, is an innovative epicutaneous immunotherapy designed for the treatment of peanut allergy. It delivers small, controlled amounts of peanut protein through the skin using a non-invasive patch, promoting immune tolerance while minimizing systemic exposure. This approach targets Langerhans cells in the epidermis to modulate the immune response without entering the bloodstream in significant quantities. DBV712 aims to reduce the risk of severe allergic reactions upon accidental exposure, offering a safer and more convenient alternative to oral immunotherapy, particularly for pediatric patients.
PVX108 (AVX-201): Aravax
PVX108, developed by Aravax, is an investigational peptide-based immunotherapy for peanut allergy designed to induce immune tolerance by targeting specific T-cell epitopes of peanut allergens. Unlike traditional allergen exposure approaches, it uses short synthetic peptides that do not cross-link IgE, thereby reducing the risk of allergic reactions during treatment. This targeted mechanism aims to modulate the immune response, promoting long-term desensitization while improving safety and tolerability. By addressing the underlying immune dysfunction, PVX108 has the potential to offer a disease-modifying approach for food allergy management.
Comparison of Emerging Food Allergy Drugs Under Development | |||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Molecule Type |
Anticipated Launch in the US |
|
Viaskin Peanut Patch (DBV712) |
DBV Technologies |
III |
Peanut Allergy |
Epicutaneous |
Immunomodulator |
Immunotherapy |
Information is available in the full report |
|
PVX108 (AVX-201) |
Aravax |
III |
Peanut Allergy |
Intradermal |
Target T cells and reverse the course of allergic disease |
Immunotherapy |
Information is available in the full report |
|
ANAPHYLM |
Aquestive Therapeutics |
II |
Anaphylaxis |
Sublingual |
Adrenergic receptor agonists |
Small molecule |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | |||||||
Note: A detailed emerging therapies assessment will be provided in the final report
Food Allergy Key Players, Market Leaders and Emerging Companies
- Novartis
- Roche
- ARS Pharmaceuticals
- ALK-Abelló
- Alfresa
- Stallergenes Greer
- DBV Technologies
- Aravax
- Aquestive Therapeutics, and others
Food Allergy Market Outlook
The food allergy market is undergoing a significant transformation, shifting from strict allergen avoidance and emergency management toward more proactive, disease-modifying therapeutic approaches. The emergence of novel immunotherapies such as PALFORZIA, Viaskin Peanut Patch (DBV712), and PVX108 (AVX-201) reflects a move toward targeted immune modulation aimed at inducing long-term tolerance rather than temporary desensitization. These advancements highlight mechanism-driven strategies focused on improving safety, reducing the risk of severe reactions, and enhancing patient adherence, thereby strengthening clinical confidence and reshaping the competitive landscape.
With the continued development of innovative pipeline candidates, including PVX108 (AVX-201), Viaskin Peanut Patch (DBV712), and ANAPHYLM (epinephrine sublingual film), the food allergy treatment landscape is evolving in line with other immune-mediated conditions where precision-based therapies are gaining traction. Additionally, biologics targeting key immune pathways, such as XOLAIR, are further expanding therapeutic possibilities. The United States currently represents the largest food allergy market, supported by higher awareness, improved diagnosis rates, strong regulatory support, and early adoption of novel therapies compared with Europe and other regions.
Overall, the introduction of differentiated therapies such as PALFORZIA, alongside emerging options like Viaskin Peanut Patch, PVX108, and ANAPHYLM, improving diagnostic practices, and rising awareness among patients and caregivers are expected to drive steady growth in the global food allergy market over the forecast period. This evolving landscape presents substantial commercial opportunities for both approved products and pipeline candidates, particularly as the field moves closer to achieving long-term disease control and improved quality of life for patients.
- According to estimates, the total Food Allergy market size in 7MM was approximately USD 3,500 million in 2025, with the United States accounting for the largest share, and the market is projected to grow at a significant CAGR through 2036.
- In 2025, among the EU4 and the UK, Germany held the largest market share, followed by the UK, while France accounted for the smallest share.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Food Allergy (2022–2036 Forecast)
The food allergy market comprises emergency pharmacologic treatments, allergen-specific immunotherapies, biologics, and supportive/non-drug approaches, each addressing distinct aspects of acute reaction management, desensitization, and long-term immune modulation.
- Allergen-specific immunotherapies: Products such as PALFORZIA and Viaskin Peanut Patch (DBV712) aim to desensitize patients through controlled allergen exposure, increasing tolerance thresholds while requiring careful monitoring for adverse events.
- Biologics: Therapies such as XOLAIR target IgE–mediated pathways, reducing allergic response severity and emerging as adjunctive or standalone options in food allergy management.
Emergency treatments and immunotherapies together form the current standard of care, while biologics and next-generation immunomodulatory therapies are shaping the future innovation landscape in food allergy.
Further details will be provided in the report….
Food Allergy Drug Uptake
This section focuses on the uptake rate of potential Food Allergy drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Food Allergy market's uptake by drugs, patient uptake by therapy, and sales of each drug.
The uptake of therapies in food allergy is expected to vary across allergen avoidance strategies, emergency treatments, immunotherapies, and emerging disease-modifying agents. Currently available options, such as epinephrine for acute reactions and oral immunotherapy (e.g., peanut allergen products), demonstrate moderate uptake, while newer approaches, including epicutaneous patches and peptide-based immunotherapies, are anticipated to show progressive adoption, supported by improved safety profiles and better patient compliance. Their uptake is likely to be driven by the limitations of strict dietary avoidance and the persistent risk of accidental exposure, along with the burden of anxiety and reduced quality of life. In comparison, next-generation therapies targeting immune tolerance and novel delivery approaches are expected to witness gradual but steady uptake as clinical evidence strengthens and these therapies become increasingly integrated into treatment guidelines for diverse food allergy populations.
Further detailed analysis of emerging therapies' drug uptake in the report…
Food Allergy Market Access and Reimbursement Scenario
-
US
The US Reimbursement for Food Allergy Therapies | |
|
Drug |
Access Program |
|
Omalizumab (XOLAIR) |
|
|
Epinephrine /Adrenaline (NEFFY/ EURNEFFY) |
|
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Further details are provided in the final report….
Food Allergy Therapies Price Scenario & Trends
Pricing and analogue assessment of Food Allergy therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, the closest and most appropriate analogue selection for emerging therapies, and the understanding of how pricing influences market access, adherence, and long-term uptake.
-
Pricing of Juvenile Food Allergy Approved Drugs
At a list price of approximately USD 890 per month in the US, PALFORZIA results in an estimated annual therapy cost of around USD 10,680.
Further details are provided in the final report….
Industry Experts and Physician Views on Food Allergy
To keep up with Food Allergy market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the Food Allergy emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Food Allergy, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 10+ KOLs to gather insights; however, interviews were conducted with 6+ KOLs in the 7MM. Centers such as the Harvard Medical School, University of Nottingham, and Japanese Red Cross Central Blood Institute, etc. were contacted. Their opinion helps understand and validate current and emerging Food Allergy therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Food Allergy.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Food allergy, diet, and nutrition are deeply interconnected, with growing evidence pointing to a complex, bidirectional relationship between nutrition and psychological well-being, potentially influenced by the gut microbiome. Emerging data also suggest that dietary patterns may have long-term implications for both health and disease, revealing areas of opportunity for further exploration and intervention.” |
|
Italy |
“Over-diagnosis remains a significant challenge in pediatric food allergy, often fueled by the dependence on specialized tertiary centers for diagnostic confirmation, the inherent risks of Oral Food Challenges (OFC), psychological strain on families, and the high financial burden. A European systematic review highlighted a striking gap between self-reported food allergy (17.3%) and cases confirmed through OFC (0.9%). Similarly, our findings showed that only about half of the children referred for suspected food allergy were ultimately diagnosed with a true allergy after comprehensive evaluation.” |
Food Allergy Report Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Food Allergy, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are mainly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Food Allergy Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Food Allergy, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Food Allergy market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Food Allergy market.
Food Allergy Market Report Insights
- Food Allergy Patient Population Forecast
- Food Allergy Therapeutics Market Size
- Food Allergy Pipeline Analysis
- Food Allergy Market Size and Trends
- Food Allergy Market Opportunity (Current and Forecasted)
Food Allergy Market Report Key Strengths
- Food Allergy Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-enabled Market Research Report
- 11-year Forecast
- Food Allergy Market Outlook (North America, Europe, Asia-Pacific)
- Food Allergy Patient Burden Trends (by geography)
- Food Allergy Treatment Addressable Market (TAM)
- Food Allergy Competitive Landscape
- Food Allergy Major Companies Insights
- Food Allergy Price Trends and Analogue Assessment
- Food Allergy Therapies Drug Adoption/Uptake
- Food Allergy Therapies Peak Patient Share analysis
Food Allergy Market Report Assessment
- Food Allergy Current Treatment Practices
- Food Allergy Unmet Needs
- Food Allergy Clinical Development Analysis
- Food Allergy Emerging Drugs Product Profiles
- Food Allergy Market Attractiveness
- Food Allergy Qualitative Analysis (SWOT and Conjoint Analysis)
- Food Allergy Market Drivers
- Food Allergy Market Barriers
FAQs Related to the Food Allergy Market Report:
Food Allergy Market Insights
- What was the Food Allergy market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Food Allergy?
- What are the disease risks, burdens, and unmet needs of Food Allergy? What will be the growth opportunities across the 7MM concerning the Food Allergy patient population?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Food Allergy? What are the current guidelines for treating Food Allergy in the US, Europe, and Japan?
Reasons to Buy Food Allergy Market Forecast Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Food Allergy market.
- Bottom-up forecasting builds from the affected population to product forecasts, delivering a robust, data-driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) - enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data-driven decisions.