Gastroparesis Insights and Trends
- Diabetic gastroparesis is the most common subtype, followed by postsurgical cases, with most pipeline therapies targeting idiopathic and diabetic forms due to their higher prevalence and commercial potential.
- As per NORD, gastroparesis demonstrates a gender bias affecting more women than men. Approximately 80% of idiopathic cases are women.
- In the US, the highest etiology-specific cases accounted for diabetes (57%), followed by postsurgical cases (15%) and drug-induced cases (11%) in 2025.
- Gastroparesis cases can be categorized based on the severity of delayed gastric emptying as mild (<20% gastric retention), moderate (20–29% gastric retention), and severe (≥30% gastric retention). As per the estimates, in the US, mild cases represent the largest share of gastroparesis at 43%, followed by severe cases.
- There's limited research on the prevalence of gastroparesis in Asian populations due to potential underdiagnosis and lack of interest.
- Oral metoclopramide has long been the sole US FDA-approved option for diabetic gastroparesis. In June 2020, GIMOTI became the first FDA-approved nasal formulation, providing faster symptom relief and overcoming absorption issues tied to delayed gastric emptying, while its use is still limited by tardive dyskinesia risks and treatment duration guidelines.
- Several drugs approved for other conditions are utilized by clinician’s off-label to alleviate gastroparesis symptoms. These include medications such as nortriptyline (a tricyclic antidepressant), mirtazapine, buspirone, and others.
- Dietary modifications represent the first line of treatment for gastroparesis and are generally used for all patients, regardless of disease severity.
- Therapeutic approaches for gastroparesis include lifestyle and dietary modifications, medications such as prokinetics and antiemetics, as well as pain management and surgical options.
- While current treatment options remain limited, ongoing clinical development and innovation in prokinetic and antiemetic therapies are likely to expand the therapeutic landscape, creating new opportunities across major markets in the coming years.
- In the gatroparesis market, a few of the emerging therapies are Naronapride, CIN-102, PCS12852, PCS12852 (YH12852), and others are being developed in the mid-late-stage clinical development.
Gastroparesis Market Size and Forecast in the US
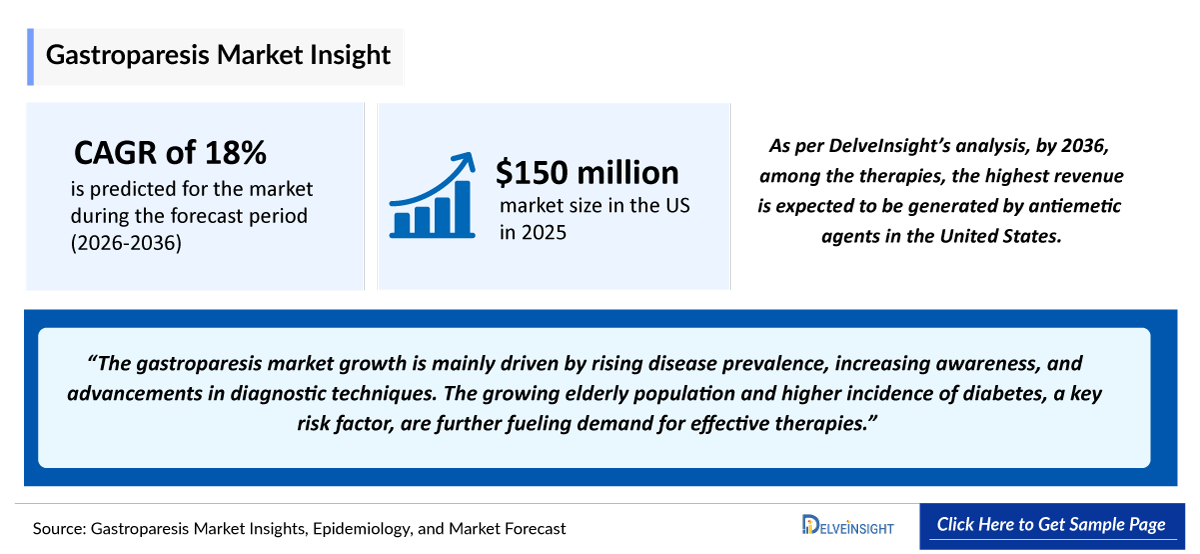
- 2025 Gastroparesis Market Size: ~150 million
- 2036 Projected Gastroparesis Market Size: ~970 million
- Gastroparesis Growth Rate (2026–2036): ~18% CAGR
DelveInsight's ‘Gastroparesis – Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of the gastroparesis, historical and forecasted epidemiology, as well as the gastroparesis market trends in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
The gastroparesis market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates gastroparesis patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across global regions. The report highlights key unmet medical needs in gastroparesis and maps the competitive and clinical landscape to uncover high‑value opportunities, providing a clear outlook on future market growth potential.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
Gastroparesis Market CAGR (Forecast period) |
~18% (2026–2036) |
|
Gastroparesis Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Gastroparesis Companies |
|
|
Gastroparesis Therapies |
|
|
Gastroparesis Market |
Segmented by
|
|
Analysis
|
|
Key Factors Driving the Gastroparesis Market
Rising Prevalence of Diabetes
Gastroparesis is strongly linked to diabetes, particularly long-standing cases. As diabetes rates increase globally, the number of gastroparesis patients grows, boosting demand for diagnosis and treatment.
Aging Population
Older adults are more prone to gastrointestinal disorders, including gastroparesis. The growing elderly population contributes significantly to rising case numbers and healthcare demand.
Unmet Medical Needs
Current treatments often provide limited relief and may have side effects. The need for more effective and safer therapies continues to drive innovation and market interest.
Gastroparesis Understanding and Treatment Algorithm
Gastroparesis Overview and Diagnosis
Gastroparesis is a chronic gastrointestinal disorder characterized by delayed gastric emptying without any mechanical obstruction. It commonly occurs in people with long-standing diabetes but can also arise from post-surgical complications, medications, or idiopathic causes. Patients typically experience nausea, vomiting, early satiety, bloating, and abdominal pain, which can significantly impact nutrition and quality of life. The condition can lead to complications such as malnutrition, dehydration, and erratic blood glucose levels, particularly in diabetic patients. Its chronic and relapsing nature often requires long-term management and monitoring.
Diagnosis involves ruling out mechanical obstruction and confirming delayed gastric emptying. Common tools include gastric emptying scintigraphy (the gold standard), breath tests, and upper endoscopy. Physicians also assess underlying causes such as diabetes and review medications that may impair gastric motility, ensuring a comprehensive evaluation before confirming gastroparesis. Additional tests like wireless motility capsules and imaging studies may be used in complex cases. Accurate diagnosis is crucial to differentiate gastroparesis from other functional gastrointestinal disorders with similar symptoms.
Further details are provided in the report.
Current Gastroparesis Treatment Landscape
Treatment focuses on symptom management, dietary modifications, and improving gastric motility. Patients are advised to consume small, low-fat, low-fiber meals. Pharmacologic therapy includes prokinetic and antiemetic agents such as Metoclopramide (GIMOTI), the only FDA-approved drug specifically indicated for gastroparesis, and Erythromycin, used off-label. Other supportive medications, like antiemetics, may help control nausea and vomiting. In severe or refractory cases, options such as gastric electrical stimulation, jejunostomy feeding tubes, or surgical interventions may be considered to maintain nutrition and improve quality of life. Continuous monitoring and individualized treatment plans are essential due to variability in patient response.
Further details related to country-based variations are provided in the report.
Gastroparesis Unmet Needs
The section “unmet needs of gastroparesis” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Limited effective treatment option
- Insufficient clinical guidelines and standardization
- Need for better biomarkers and diagnostic tools
- High symptom burden and poor quality of life, and others…..
Note: Comprehensive unmet needs insights in Gastroparesis and their strategic implications are provided in the full report.
Gastroparesis Epidemiology
Key Findings from Gastroparesis Epidemiological Analysis and Forecast
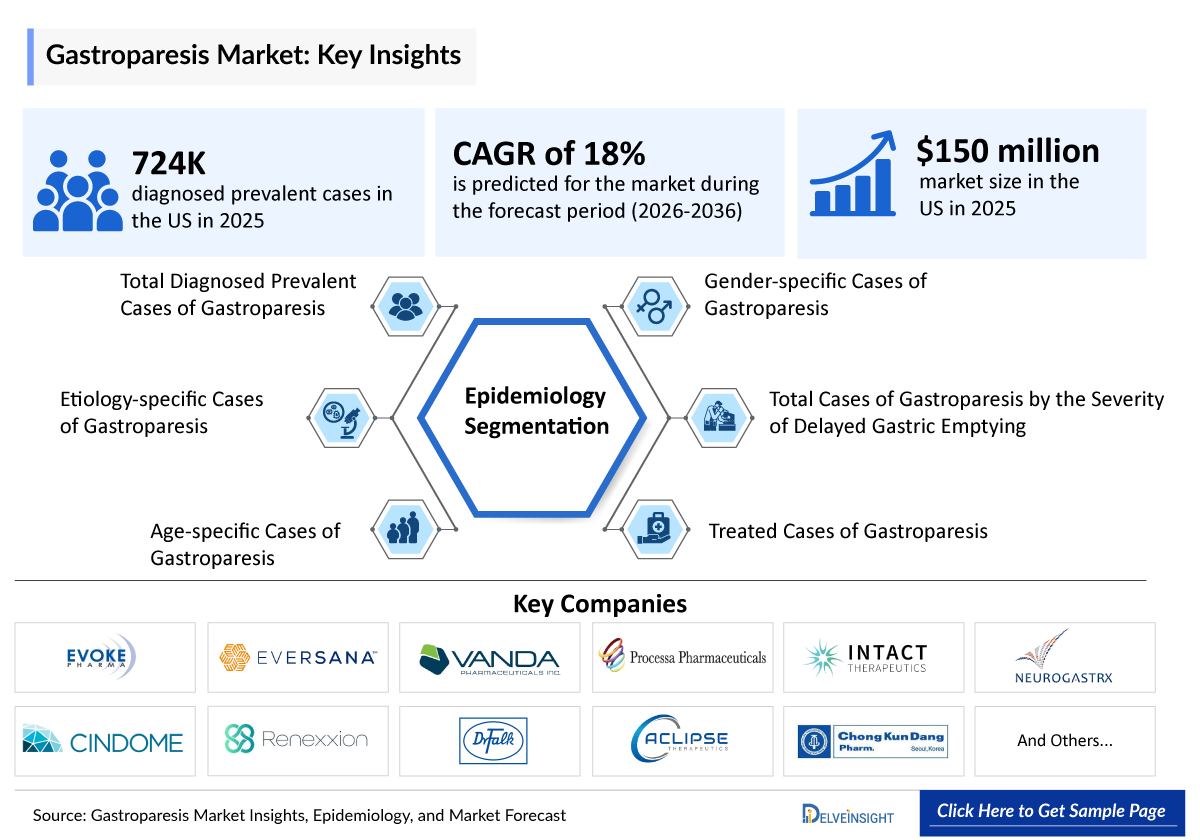
- Among the 7MM, the US accounted for the highest number of diagnosed prevalent cases of gastroparesis with ~724,000 cases in 2025.
- In the US in 2025, the highest age-specific cases were seen in the 65+ age group, followed by those aged 48–57, while the lowest were in the 18–27 age group.
- Gastroparesis as a disease predominantly affects women, with ~70% of the diagnosed patients being females, while only ~30% of the diagnosed patients are males.
- Among etiology-specific cases across the 7MM, diabetes has the highest prevalence, followed by postsurgical causes, drug-induced cases, and then idiopathic cases.
Gastroparesis Drug Analysis & Competitive Landscape
The gastroparesis drug chapter provides a detailed, market-focused review of the emerging pipeline across Phase I-III clinical trials. It covers the mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, and strategic partnerships for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the gastroparesis treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the gastroparesis therapeutics market.
Approved Therapies for Gastroparesis
Metoclopramide (GIMOTI): EVOKE Pharma/EVERSANA
GIMOTI nasal spray is the first and only FDA-approved non-oral treatment for the relief of symptoms in adults with acute and recurrent diabetic gastroparesis. Metoclopramide hydrochloride is the active ingredient in GIMOTI. It is a dopamine antagonist, 5-HT3 antagonist, and 5-HT4 agonist with promotility and anti-emetic effects, for the relief of symptoms associated with acute and recurrent diabetic gastroparesis. In June 2020, the US FDA approved metoclopramide (GIMOTI) nasal spray. In January 2020, EVOKE Pharma entered into a commercial services agreement with Eversana Life Sciences Services for the commercialization of GIMOTI. According to the Eversana Agreement, Eversana will commercialize and distribute GIMOTI in the US.
|
Acute Myeloid Leukemia (AML) Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Metoclopramide (GIMOTI) |
EVOKE Pharma/ EVERSANA |
Diabtetic gastroparesis |
Small molecule |
Dopamine-2 receptor antagonist/serotonin 5-HT4 receptor agonist |
Oral |
US: 2020 |
Note: Detailed marketed therapies assessment will be provided in the final report.
Gastroparesis Pipeline Analysis
Naronapride (ATI-7505): Renexxion
Naronapride is an investigational gastrointestinal prokinetic drug. It is designed to treat disorders of gut motility, including gastroparesis. Naronapride works through a differentiated dual mechanism of action- 5-HT4 receptor agonism and dopamine D2 receptor antagonism. Inhibiting D2 receptors removes the “brakes” on the motility of the gut. The high 5-HT4 receptor selectivity minimizes the potential for off-target pharmacological effects. Its dual mechanism of action, minimal systemic bioavailability, and differentiated pharmaceutical and pharmacokinetic profile are designed to deliver targeted activity within the GI tract while limiting systemic exposure. In October 2021, Renexxion Ireland announced that it had entered into a Licensing and Collaboration Agreement with Dr. Falk Pharma GmbH to jointly develop and commercialize Naronapride, a unique late-stage GI prokinetic, initially for gastroparesis.
CIN-102 (deudomperidone): CinDome Pharma
Deudomperidone an oral, small-molecule dopamine D2/D3 receptor antagonist. As an antiemetic and prokinetic, deudomperidone improves gastric emptying and relieves the symptoms of nausea and vomiting associated with gastroparesis. Deudomperidone has completed its Phase I program, demonstrated a negative thorough QT study, and conducted a proof-of-concept Phase II trial demonstrating a reduction in gastric emptying. Topline safety and efficacy results from EnvisionGI are expected in the 2H of 2026. The topline safety and efficacy results of Envision3D are anticipated in Q1 2026.
|
Competitive Landscape of Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Naronapride (ATI-7505) |
Renexxion |
II |
Gastroparesis |
Oral |
5-HT4 receptor agonist and dopamine D2 receptor antagonist |
Information is available in the full report |
|
CIN-102 (deudomperidone) |
CinDome Pharma |
II |
Idiopathic gastroparesis |
Oral |
Dopamine D2/3 receptor antagonists |
Information is available in the full report |
|
NG101 (metopimazine) |
Neurogastrx |
II |
Gastroparesis |
Oral |
Dopamine D2/3 receptor antagonists |
Information is available in the full report |
|
PCS12852 |
Processa Pharma/Intact Therapeutics |
II |
Moderate-to-severe gastroparesis |
Oral |
5-hydroxytryptamine-4 (5-HT4) receptor agonist |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Note: Detailed emerging therapies assessment will be provided in the final report.
Gastroparesis Key Players, Market Leaders, and Emerging Companies
- EVOKE Pharma
- Renexxion
- CinDome Pharma
- Neurogastrx
- Processa Pharmaceuticals
- Intact Therapeutics, and others
Gastroparesis Drug Updates
- In April 2026, Dr. Falk Pharma GmbH and Renexxion Ireland announced that data from the Global Phase IIb (MOVE-IT) study of naronapride in 328 adults with moderate-to-severe idiopathic or diabetic gastroparesis had been selected for a late-breaking oral presentation at DDW 2026 in the US.
- In March 2026, CinDome Pharma announced that enrollment in the Phase II (envisionGI) trial of deudomperidone (CIN-102) for adults with idiopathic gastroparesis had been completed.
- In September 2025, CinDome Pharma announced that it had completed enrollment in the (envision3D) Phase II clinical trial of deudomperidone in adults with diabetic gastroparesis.
- In June 2025, Processa Pharmaceuticals announced that it had entered into a binding term sheet with Intact Therapeutics, granting Intact an exclusive option to license PCS12852, a best-in-class 5-HT4 receptor agonist with the potential to become a first meaningful treatment for gastroparesis and other gastrointestinal motility disorders.
- In March 2024, Renexxion Ireland and Dr. Falk Pharma GmbH announced US FDA clearance of the INDA for naronapride to treat patients with gastroparesis.
Gastroparesis Market Outlook
The treatment landscape for gastroparesis has historically relied on symptomatic management, including dietary modifications, antiemetics, and prokinetic agents. A key milestone in therapy is the approval of metoclopramide (GIMOTI), which enhances gastric motility through dopamine receptor antagonism. Despite its effectiveness, long-term use is limited by safety concerns, highlighting the need for better-tolerated and more durable therapies.
The gastroparesis pipeline is gradually evolving, with investigational agents targeting gastrointestinal motility and symptom control through novel mechanisms. Naronapride (ATI-7505), a selective 5-HT4 receptor agonist, is being developed to enhance gastric emptying with improved cardiac safety versus earlier agents. CIN-102 (deudomperidone) aims to deliver prokinetic effects with fewer central nervous system side effects. NG101 (metopimazine) is also under investigation for its antiemetic action, potentially relieving persistent nausea and vomiting associated with gastroparesis.
Overall, the emergence of safer and more targeted therapies, along with improvements in diagnostic capabilities and increasing disease awareness, is projected to propel steady market growth across the 7MM from 2022 to 2036, yielding significant commercial opportunities for existing products and pipeline candidates.
- Among the 7MM, the US represented the largest gastroparesis market, valued at approximately USD ~150 million in 2025, and is projected to grow at a significant CAGR through 2036.
- Currently, the majorly prescribed class of therapies is prokinetics, such as oral or IV metoclopramide antiemetics and neuromodulators to manage symptoms.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Gastroparesis (2022–2036 Forecast)
The gastroparesis market (2022–2036 forecast) is shifting toward mechanism-based therapies that aim to improve gastric motility and control core symptoms such as nausea and vomiting, rather than relying solely on general supportive care.
- Small molecules: The current gastroparesis pipeline is dominated by small-molecule therapies designed to modulate gastrointestinal motility and neurotransmitter pathways. Naronapride (ATI-7505) is a selective 5-HT4 receptor agonist that stimulates acetylcholine release, enhancing gastric emptying while aiming to minimize cardiovascular risks seen with earlier agents. CIN-102 (deudomperidone) and NG101 (metopimazine) target dopamine D2 receptors to improve motility and control nausea and vomiting, respectively.
Overall, innovation in gastroparesis is driven by approved and investigational small-molecule therapies targeting complementary mechanisms, including prokinetic and antiemetic effects. As pipeline candidates advance, they are expected to broaden treatment options beyond metoclopramide, address key unmet needs, and support steady market growth across major regions.
Gastroparesis Drug Uptake
This section focuses on the uptake rate of potential drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the gastroparesis drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
During the forecast period, the currently FDA-approved therapy Metoclopramide (GIMOTI) is expected to maintain baseline utilization as the first-line pharmacologic option. However, its uptake is likely to plateau due to safety concerns such as extrapyramidal side effects and limitations on long-term use. It will continue to be widely used in acute and short-term management, particularly in newly diagnosed patients and in settings where newer therapies are not yet accessible.
Among pipeline agents, Naronapride (ATI-7505) is expected to see gradual uptake as a next-generation 5-HT4 agonist, driven by its potential to improve gastric motility with a more favorable cardiovascular safety profile. CIN-102 (domperidone) is expected to follow a moderate adoption curve, particularly in regions where domperidone use is established, benefiting from improved pharmacokinetic stability and reduced CNS penetration. Its uptake will likely be driven by chronic symptom management needs. NG101 (metopimazine) is expected to achieve faster early adoption in select patient segments due to its strong antiemetic effect, positioning it as an add-on therapy for refractory nausea and vomiting. Overall uptake will depend on clinical efficacy, safety outcomes, and positioning alongside standard of care therapies.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved Therapies in Gastroparesis
The United States
|
US Reimbursement of Therapies Approved for Gastroparesis | |
|
Drug/Therapy |
Access Program |
|
GIMOTI |
Evoke Pharma Saving Program for GIMOTI |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Gastroparesis Therapies Price Scenario & Trends
Pricing and analogue assessment of gastroparesis therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market gastroparesis, adherence, and long-term uptake.
- Pricing of Gastroparesis-Approved Drugs
The recommended dosage of GIMOTI for adults with acute and recurrent diabetic gastroparesis is one spray (15 mg) administered into one nostril, 30 minutes before each meal and at bedtime, up to a maximum of four times daily. Treatment duration ranges from 2 to 8 weeks, based on the patient's symptomatic response. A single bottle provides sufficient medication for 28 days of use at the maximum recommended frequency. The cost per bottle is approximately USD 2,276.03, resulting in an estimated total therapy cost of around USD 4,552 for an 8-week course.
Industry Experts and Physician Views for Gastroparesis
To keep up with the gastroparesis market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry experts were contacted for insights on the Gastroparesis emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in gastroparesis, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts connected with 15+ KOLs to gather insights at the country level. Centers such as the Duke Molecular Physiology Institute, Department of Nephrology, Hospital Universitario Fundación Alcorcón, and Nippon Medical School, etc. were contacted. Their opinion helps understand and validate current and emerging gastroparesis therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in gastroparesis.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Gastroparesis remains a significant unmet clinical need, presenting a major opportunity for new drug development. As a result, there is growing interest from federal funding agencies, regulatory bodies, and the pharmaceutical industry.” |
|
Germany |
“Although surgery can serve as a rescue therapy in those who do not respond to medication and pyloromyotomy, it is imperative to reevaluate these patients preoperatively and exclude other causes of gastroparesis-like symptoms, such as small bowel dysmotility, overlapped functional dyspepsia, etc. Surgical intervention includes surgical pyloroplasty, total gastrectomy, and subtotal gastrectomy with Roux-en-Y gastrojejunostomy reconstruction.” |
Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of gastroparesis, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided.
Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Gastroparesis, explaining its causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Gastroparesis market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Gastroparesis market.
Report Insights
- Gastroparesis Patient Population Forecast
- Gastroparesis Therapeutics Market Size
- Gastroparesis Pipeline Analysis
- Gastroparesis Market Size and Trends
- Gastroparesis Market Opportunity (Current and Forecasted)
Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- Artificial Intelligence (AI)-Enabled Market Research Report
- 11-Year Forecast
- Gastroparesis Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (By Geography)
- Gastroparesis Treatment Addressable Market (TAM)
- Gastroparesis Competitive Landscape
- Gastroparesis Major Companies Insights
- Gastroparesis Price Trends and Analogue Assessment
- Gastroparesis Therapies Drug Adoption/Uptake
- Gastroparesis Therapies Peak Patient Share Analysis
Report Assessment
- Gastroparesis Current Treatment Practices
- Gastroparesis Unmet Needs
- Gastroparesis Clinical Development Analysis
- Gastroparesis Emerging Drugs Product Profiles
- Gastroparesis Market Attractiveness
- Gastroparesis Qualitative Analysis (SWOT and Conjoint Analysis)
FAQs
Market Insights
- What was the Gastroparesis market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Gastroparesis?
- What are the disease risks, burdens, and unmet needs of Gastroparesis? What will be the growth opportunities across the 7MM concerning the patient population with Gastroparesis?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Gastroparesis? What are the current guidelines for treating Gastroparesis in the US, Europe, and Japan?
Reasons to Buy
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Gastroparesis market.
- Bottom‑up forecasting builds from the affected population to product forecasts, delivering a robust, data‑driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease incidence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights into the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI)‑enabled report summarize and simplify complex datasets with in the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data‑driven decisions.