Generalized Myasthenia Gravis Market Summary
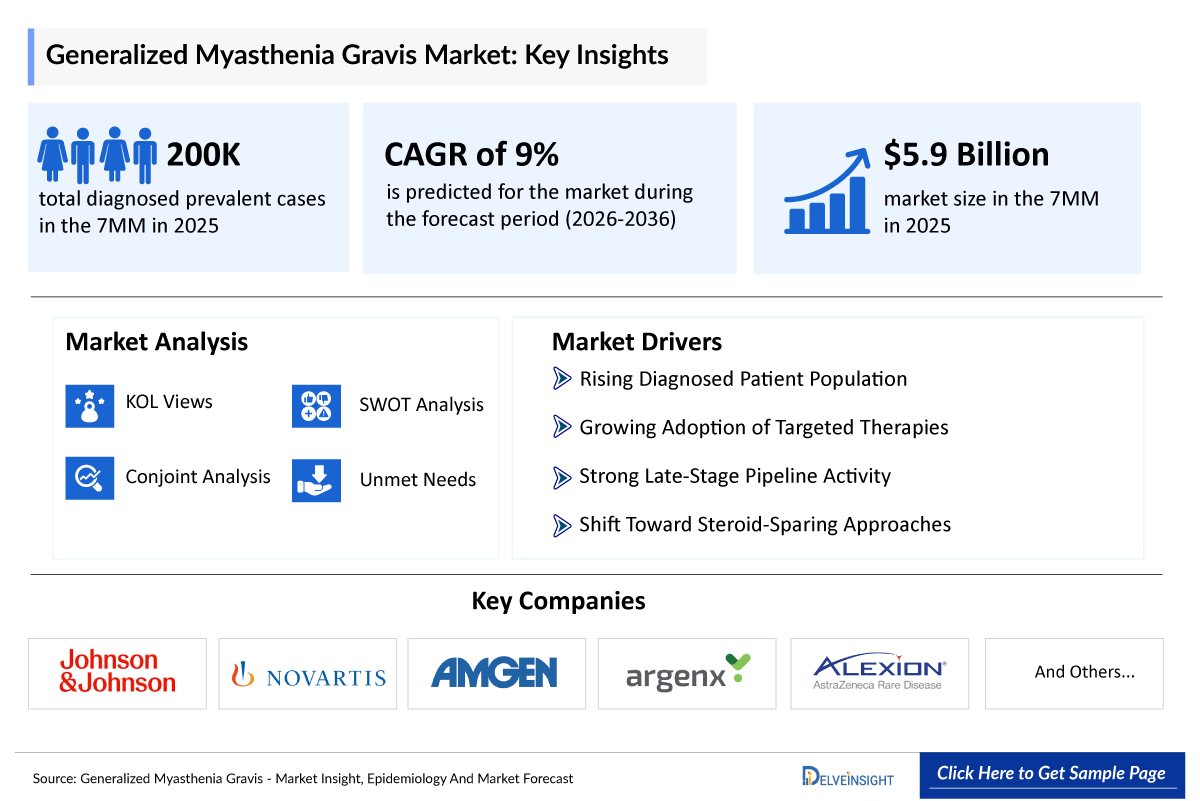
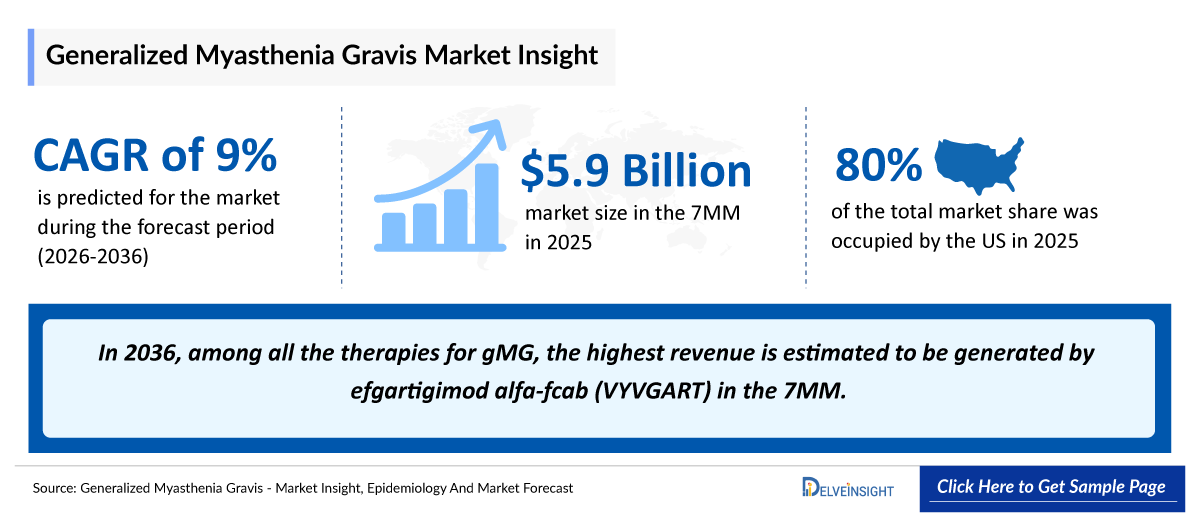
- The generalized Myasthenia Gravis (gMG) market size across the 7MM was valued at approximately USD 5,900 million in 2025 and is projected to reach nearly USD 15,600 million by 2036, growing at a CAGR of 9% during the forecast period from 2026 to 2036.
- The leading generalized Myasthenia Gravis companies developing therapies in the treatment market include - Johnson&Johnson, Novartis, AMGEN, Argenx, Alexion and others.
Generalized Myasthenia Gravis (gMG) Market & Epidemiology Insights
- gMG treatment gaps persist in achieving durable remission and rapid control in refractory patients, creating strong opportunities for differentiated therapies delivering sustained efficacy, faster onset, and improved tolerability across disease stages.
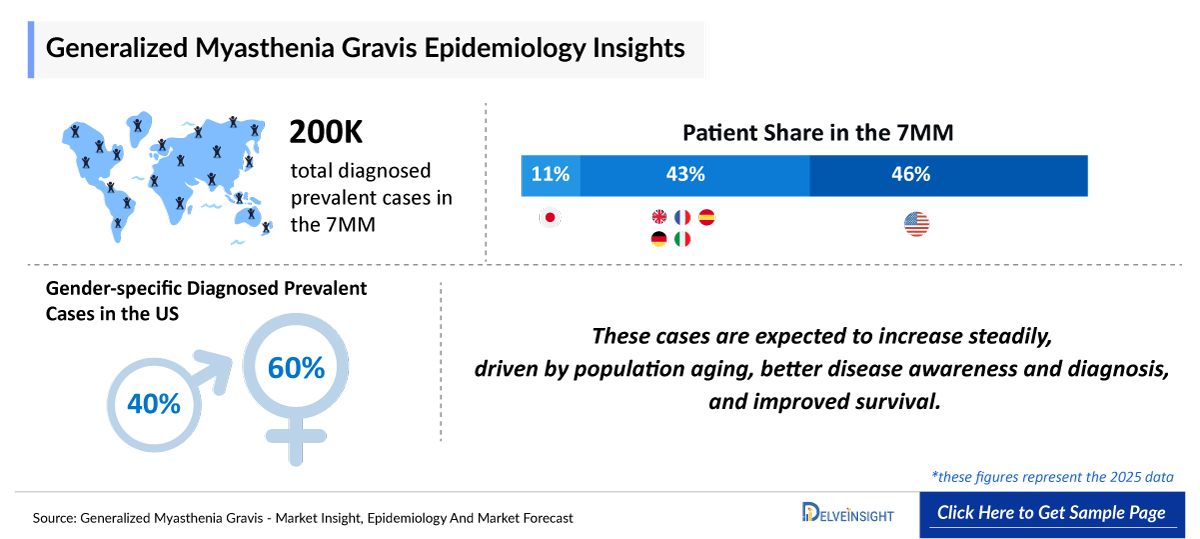
- In 2025, the United States accounted for ~90,000 diagnosed prevalent cases of gMG, underscoring a substantial patient base driving sustained demand.
- Companies such as Immunovant, Alexion, Cartesian Therapeutics, and others are advancing late-stage Phase III assets such as IMVT-1402, Gefurulimab, Descartes-08, and others; coupled with regulatory designations like ODD, FTD, Priority Review, and RMAT, these therapies are well-positioned to accelerate market growth and reshape the gMG treatment landscape.
- A key growth driver in gMG is the rapid advancement and uptake of targeted biologics and novel immunotherapies, supported by expedited regulatory pathways, enabling faster market entry and improved patient outcomes.
- gMG management is transitioning from broad immunosuppression to targeted biologics such as VYVGART, RYSTIGGO, ZILBRYSQ, ULTOMIRIS, and others driving mechanism-specific control, improved durability.
- The loss of exclusivity of SOLIRIS has catalyzed the first meaningful biosimilar wave in gMG, introducing tangible pricing pressure and access expansion, while simultaneously accelerating strategic migration toward next-generation complement inhibitors with improved durability and lifecycle protection.
Generalized Myasthenia Gravis (gMG) Market size and forecast
- 2025 gMG Market Size in the 7MM: ~USD 5,900 million
- 2036 Projected gMG Market Size in the 7MM: ~USD 15,600 million
- gMG Growth Rate (2026–2036) in the 7MM: ~9% CAGR
Request for unlocking the Sample Page of the "Generalized Myasthenia Gravis Market Insights"
Key Factors Driving the Generalized Myasthenia Gravis (gMG) Market:
- Rising Prevalence of Autoimmune Disorders: Increasing incidence and diagnosis of generalized Myasthenia Gravis (gMG) are contributing to higher treatment demand globally.
- Advancements in Targeted Therapies: The emergence of biologics, FcRn inhibitors, and complement inhibitors is significantly improving treatment outcomes and expanding the therapeutic landscape.
- Growing Awareness and Early Diagnosis: Improved awareness among healthcare professionals and patients is leading to earlier detection and timely treatment initiation.
- Strong Pipeline and Regulatory Approvals: Ongoing clinical trials and recent approvals of innovative therapies are accelerating market growth and creating new commercial opportunities.
- Expanding Healthcare Infrastructure and Reimbursement Support: Better access to specialty care, advanced diagnostics, and favorable reimbursement policies are supporting increased adoption of premium gMG therapies.
DelveInsight's ‘Generalized Myasthenia Gravis (gMG) Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of gMG, historical and forecasted epidemiology, as well as the gMG therapeutics market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The gMG market report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, gMG patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key unmet medical needs in gMG and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Scope of the Generalized Myasthenia Gravis Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Generalized Myasthenia Gravis Market CAGR (Forecast period) |
9.1% (2026-2036) |
|
Generalized Myasthenia Gravis Epidemiology Segmentation Analysis |
Patient Burden Assesment
|
|
Generalized Myasthenia Gravis Companies |
|
|
Generalized Myasthenia Gravis Therapies |
|
|
Generalized Myasthenia Gravis Market |
Segmented by
|
|
Analysis |
|
Generalized Myasthenia Gravis (gMG) Disease Understanding
Generalized Myasthenia Gravis (gMG) Overview and Diagnosis
Generalized Myasthenia Gravis (gMG), is an autoimmune disease in which the immune system attacks the body’s tissues. In myasthenia gravis, the attack interrupts the connection between nerve and muscle the neuromuscular junction. Myasthenia gravis is characterized by autoantibodies against the Acetylcholine Receptor Antibody (AChR-Ab) or against a receptor-associated protein called Muscle-specific tyrosine Kinase Antibody (MuSK-Ab).
The clinical diagnosis of gMG is confirmed by Electromyography (EMG) studies, pharmacologic testing, and serum Ab assay. Positive results on EMG confirm a postsynaptic defect of the NMT, the clinical response to Cholinesterase Inhibitors (ChE-Is) supports myasthenia gravis diagnosis, and detection of specific Abs confirms gMG and identifies ab-related subgroups. EMG confirmation is crucial in patients with neither AChR nor MuSK Abs on the standard assay.
Further details are provided in the report...
Current Generalized Myasthenia Gravis (gMG) Treatment Landscape
Current management of gMG has traditionally relied on symptomatic relief and broad immunosuppression, with acetylcholinesterase inhibitors such as pyridostigmine providing immediate symptom control, while corticosteroids and immunosuppressants like azathioprine and MMF enable longer-term disease stabilization; rapid-acting interventions such as IVIg and plasma exchange are reserved for acute exacerbations, and thymectomy remains an option for select patients. However, the treatment paradigm is increasingly shifting toward targeted biologics, with agents such as VYVGART, RYSTIGGO, ZILBRYSQ, ULTOMIRIS, and UPLIZNA enabling mechanism-specific modulation through FcRn blockade and complement inhibition, thereby addressing underlying disease pathology with improved precision and reduced reliance on chronic immunosuppression.
Further details related to country-based variations are provided in the report.
Generalized Myasthenia Gravis (gMG) Unmet Needs
The section “unmet needs of gMG” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Lack of Therapies Ensuring Sustained Remission
- Limited Fast-acting and Tolerable Options for Refractory gMG
- Lack of Disease-modifying Therapies (DMTs) to Improve Survival
- Financial Barriers and Delayed Access to Effective gMG Treatments
- Lack of Convenient Routes and Flexible Administration Schedules
and others…
Note: Comprehensive unmet needs insights in gMG and their strategic implications are provided in the full report...
Generalized Myasthenia Gravis (gMG) Epidemiology
The Generalized Myasthenia Gravis epidemiology section provides insights about the historical and current Generalized Myasthenia Gravis patient pool and forecasted trends for individual seven major countries. It helps to recognize the causes of current and forecasted trends by exploring numerous studies and views of key opinion leaders. This part of the Generalized Myasthenia Gravis market report also provides the diagnosed patient pool and their trends along with assumptions undertaken.
Key Findings from Generalized Myasthenia Gravis (gMG) Epidemiological Analysis and Forecast
- In 2025, Myasthenia Gravis accounted for ~135,000 diagnosed prevalent cases in the US.
- Among the gender-specific cases of gMG, the majority was occupied by female accounting for ~60% of the cases in the US in 2025.
- In 2025, the highest disease burden of gMG was observed in patients aged =65 years, accounting for ~108,000 cases across the 7MM, highlighting a predominantly elderly patient population.
- In 2025, by antibody serology, Anti-AChR antibody–positive cases accounted for ~80% of patients with gMG in the EU4 and the UK, representing the dominant disease subtype.
Generalized Myasthenia Gravis (gMG) Epidemiology Segmentation
- Total Diagnosed Prevalent Cases of Myasthenia Gravis
- Severity-specific Diagnosed Prevalent Cases of Myasthenia Gravis by MGFA Classification
- Total Diagnosed Prevalent Cases of gMG
- Gender-specific Diagnosed Prevalent Cases of gMG
- Age-specific Diagnosed Prevalent Cases of gMG
- Diagnosed Prevalent Cases of gMG by Antibody Serology
- gMG Treated Cases
- Treated Cases of gMG
Recent Devleopments in the Generalized Myasthenia Gravis (gMG) Treatment Landscape:
- In December 2025, Johnson & Johnson reported that the European Commission approved a Marketing Authorisation for IMAAVY (nipocalimab) as an add-on to standard therapy for gMG, making it the first FcRn blocker approved for both adults and adolescents aged 12 years and older who are anti-AChR or anti-MuSK antibody-positive; this approval builds on its existing authorisations in the US, Brazil, and Japan for the treatment of gMG.
- In May 2025, UCB reported that it had obtained approval Pharmaceuticals and Medical Devices Agency (PMDA) for rozanolixizumab to be self-administered at home by patients using either an infusion pump or a newly authorized manual push syringe technique.
- In December 2023, the EC approved ZILBRYSQ for the treatment of gMG in adult patients who are anti-AChR antibody-positive1 .
Generalized Myasthenia Gravis (gMG) Drug Analysis & Competitive Landscape
The Generalized Myasthenia Gravis (gMG) drug chapter provides a detailed, market-focused review of approved therapies and the emerging pipeline across Phase II Generalized Myasthenia Gravis clinical trials. It covers mechanism of action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming Key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Generalized Myasthenia Gravis (gMG) treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the (gMG) therapeutics market.
Approved Therapies for Generalized Myasthenia Gravis (gMG)
Rozanolixizumab (RYSTIGGO): UCB
Rozanolixizumab is a subcutaneous (SC) humanized monoclonal IgG4 antibody designed to bind with high affinity to the neonatal Fc receptor (FcRn). By blocking the interaction between FcRn and IgG, it enhances IgG degradation, thereby reducing circulating pathogenic IgG autoantibodies key contributors to autoimmune diseases such as Gmg. The therapy is FDA-approved for adult patients with gMG who are positive for anti-AChR or anti-MuSK antibodies, the two most prevalent gMG subtypes. Ongoing studies are also evaluating its safety and efficacy in pediatric populations.
In May 2025, UCB reported that it had obtained approval Pharmaceuticals and Medical Devices Agency (PMDA) for rozanolixizumab to be self-administered at home by patients using either an infusion pump or a newly authorized manual push syringe technique.
Generalized Myasthenia Gravis (gMG) Marketed/Approved Therapies | ||||||
|
Drug/Therapy |
Company |
Indication |
Molecule Type |
MoA |
RoA |
Marketed Region |
|
Zilucoplan (ZILBRYSQ) |
UCB |
gMG |
Macrocyclic peptide |
Complement C5 inhibitor |
SC injection |
US: 2023 EU: 2023 JP: 2023 |
|
Nipocalimab-aahu (IMAAVY) |
Johnson & Johnson |
gMG |
mAb |
FcRn antagonist |
IV infusion |
US: 2025 EU: 2025 JP: 2025 |
|
Note: To be continued in the final report.. | ||||||
Generalized Myasthenia Gravis (gMG) Pipeline Analysis
IMVT-1402: Immunovant/ Roivant Sciences
IMVT-1402 is a next-generation, SC administered anti-FcRn mAb developed by Immunovant for the treatment of IgG-mediated autoimmune diseases. It effectively lowers pathogenic IgG while preserving normal albumin and cholesterol levels, mitigating safety issues seen with previous FcRn inhibitors.
In December 2025, Immunovant reported the pricing of an underwritten common stock offering expected to generate approximately USD 550 million in gross proceeds, with Roivant Sciences, its controlling stockholder, committing to participate in the offering.
Competitive Landscape of Generalized Myasthenia Gravis Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Descartes-08 |
Cartesian Therapeutics |
III |
gMG |
IV infusion |
RNA CAR T-cell therapy |
2028 |
|
(FABHALTA) Iptacopan |
Novartis |
III |
gMG |
Oral |
CFB inhibitor |
Information is available in the full report |
|
Gefurulimab |
Alexion AstraZeneca Rare Disease |
III |
gMG |
SC |
Humanised bispecific VHH antibody Complement C5 inhibitors |
Information is available in the full report |
|
MAVENCLAD |
Merck KGaA |
III |
gMG |
Oral |
Cytotoxic effects on B |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Generalized Myasthenia Gravis (gMG) Key Players, Market Leaders and Emerging Companies
- Johnson&Johnson
- Novartis
- AMGEN
- Argenx
- Alexion and others
Generalized Myasthenia Gravis (gMG) Market Outlook
The market outlook for gMG remains highly positive, driven by rapid adoption of targeted biologics and expanding diagnosed patient populations. Transition from broad immunosuppression to mechanism-specific therapies particularly FcRn inhibitors and complement inhibitors is reshaping treatment paradigms and enabling premium pricing. Continued pipeline innovation, including oral and next-generation agents, is expected to enhance convenience and long-term disease control. Overall growth will be sustained by strong clinical differentiation, earlier-line use, and increasing physician preference for durable, targeted therapies across diverse patient segments.
Key marketed therapies shaping current management
- Rozanolixizumab-noli (RYSTIGGO)-UCB: Rozanolixizumab is a subcutaneous (SC) humanized monoclonal IgG4 antibody designed to bind with high affinity to the neonatal Fc receptor (FcRn). By blocking the interaction between FcRn and IgG, it enhances IgG degradation, thereby reducing circulating pathogenic IgG autoantibodies—key contributors to autoimmune diseases such as gMG.
- Zilucoplan (ZILBRYSQ)-UCB: Zilucoplan is a once-daily SC peptide therapy designed for self-administration by adults with anti-AChR antibody-positive gMG. As a targeted inhibitor of C5, it works by binding to C5 and blocking its enzymatic cleavage into C5a and C5b.
- Nipocalimab-aahu (IMAAVY)-Johnson & Johnson: IMAAVY is an FDA-approved monoclonal antibody therapy for the treatment of gMG in adults and pediatric patients aged 12 and older who test positive for either anti-AChR or anti-MuSK antibodies. As the first FcRn blocker approved for both antibody subtypes, IMAAVY is administered IV and functions by binding with high affinity to the FcRn, effectively lowering levels of circulating IgG—including disease-causing autoantibodies and alloantibodies.
And more
- Overall, in gMG, the launch targeted biologics, improved diagnosis through autoantibody testing (e.g., Anti-AChR), and increasing disease awareness are expected to drive steady growth in the 7MM gMG market from 2022–2036, with strong commercial implications for both marketed products and emerging pipelines.
- Among the 7MM, the US accounted for the largest market size of gMG, i.e., USD ~4,700 million in 2025.
- In 2036, among all the therapies for gMG, the highest revenue is estimated to be generated by efgartigimod alfa-fcab (VYVGART) in the 7MM.
- The most meaningful recent shift in Europe has been the approval and launch of multiple targeted biologics with improved delivery profiles. Efgartigimod alfa (VYVGART) and Ravulizumab (ULTOMIRIS) have been approved across EU markets, offering advanced mechanisms like FcRn blockade and complement C5 inhibition that support better disease control and improved quality of life for patients.
Further details will be provided in the report….
Drug Class/Insights into Leading Emerging and Marketed Therapies in Generalized Myasthenia Gravis (gMG) (2022–2036 Forecast)
The treatment landscape of gMG is rapidly evolving, with a diverse pipeline spanning small molecules, monoclonal antibodies, and advanced cell therapies, collectively aiming to deliver more targeted, durable, and potentially disease-modifying outcomes beyond conventional immunosuppression.
Monoclonal antibodies: Monoclonal antibodies continue to drive innovation in generalized Myasthenia Gravis, offering targeted modulation of key immune pathways with improved precision and durability. Emerging agents such as IMVT-1402, Pozelimab + Cemdisiran, and DNTH103 (Claseprubart) are advancing next-generation strategies in FcRn blockade and complement inhibition. These therapies aim to enhance efficacy, extend dosing intervals, and reduce treatment burden, positioning them as key competitors in a rapidly evolving, biologics-driven gMG landscape.
CAR-T cell therapies: CAR-T cell therapies are emerging as a potentially transformative approach in generalized Myasthenia Gravis, aiming to achieve deep and durable immune reset rather than chronic suppression. Agents such as KYV-101 and Descartes-08 target autoreactive B cells and plasma cells implicated in disease pathology. Unlike conventional therapies, these one-time or limited-duration treatments hold the potential for sustained remission, particularly in refractory patients, although challenges related to cost, safety, and scalability may influence their broader adoption in the near term.
Generalized Myasthenia Gravis (gMG) Drug Uptake
This section focuses on the uptake rate of potential Generalized Myasthenia Gravis drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the gMG drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug.
The emergence of next-generation pipeline therapies is expanding the treatment paradigm in generalized Myasthenia Gravis, with a focus on precise FcRn modulation and improved safety profiles. IMVT-1402, developed by Immunovant/Roivant Sciences, is a subcutaneously administered anti-FcRn monoclonal antibody designed to reduce pathogenic IgG while preserving albumin and cholesterol levels, thereby addressing key limitations of earlier agents. Positioned as one of a lead asset, it is advancing through clinical development in gMG and other autoimmune indications, with an anticipated medium uptake trajectory, reaching peak adoption in approximately seven years.
Detailed insights of emerging therapies' drug uptake is included in the report
Market Access and Reimbursement of Approved therapies in Generalized Myasthenia Gravis (gMG)
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of approved therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc.
Germany
Germany Reimbursement of Therapies Approved for Generalized Myasthenia Gravis (gMG) | |
|
Drug/Therapy |
Indication |
|
ULTOMIRIS (Ravulizumab) |
Adults with anti-AChR antibody-positive gMG who are still eligible for standard treatment. |
Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
NOTE: Further Details are provided in the final report….
Generalized Myasthenia Gravis (gMG) therapies Price Scenario & Trends
Pricing and analogue assessment of gMG therapies highlights evolving price dynamics structures. This section summarizes the cost of approved treatments, closest and most appropriate analogue selection for emerging therapies, and understanding of how pricing influences market access, adherence, and long-term uptake.
Pricing of Generalized Myasthenia Gravis (gMG) Approved Drugs
To calculate the price of VYVGART, the recommended dose of 700 mg once weekly for 4 weeks/cycle was used with a gap of 50 days between two cycles. A vial of 400 mg/20 mL costs USD 6,618. Based on 38 vials (four doses/cycle for 4.7 cycles/year) per year, the estimated annual cost is USD 248,850.
Price of VYVGART Hytrulo was calculated with the recommended dose of 1,008 mg efgartigimod alfa and 11,200 units hyaluronidase per 5.6 mL (180 mg/2,000 units per mL) in a single-dose vial once weekly for 4 weeks/cycle, and was used with a gap of 50 days between two cycles. A vial of 5.6 ml costs USD 17,185. Based on 19 vials required per year, the estimated annual cost is USD 323,073.
The weighted average of VYVGART and VYVGART Hytrulo was used as a reference price at approximately USD 300,000.
Industry Experts and Physician Views for Generalized Myasthenia Gravis (gMG)
To keep up with gMG market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the gMG emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts engaged with 8+ key opinion leaders (KOLs) across major markets to capture country-level insights in generalized Myasthenia Gravis. Leading centers such as University of California and Royal College of Physicians, among others, were consulted to validate clinical practices, treatment patterns, and emerging therapeutic perspectives.
Their opinion helps understand and validate current and emerging gMG, therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for Market access, therapy adoption, and pipeline prioritization in gMG.
|
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“Many individuals with gMG experience diagnostic delays due to symptom overlap with other neurological disorders. A lack of widespread awareness among clinicians and limited access to specialized testing contribute to misdiagnoses. Enhanced clinical education and improved screening strategies are vital for timely, accurate detection and better patient care.” |
|
Japan |
Japan approved eculizumab as the first molecular-targeted therapy for gMG, marking a pivotal advancement in disease management. Its introduction provided a novel, effective treatment alternative for patients with refractory generalized myasthenia gravis, addressing a critical unmet need and reshaping therapeutic strategies for a previously difficult-to-treat population.” |
Generalized Myasthenia Gravis Report Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis.
In the SWOT analysis of Generalized Myasthenia Gravis (gMG), strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. Attribute analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Generalized Myasthenia Gravis Market Report
- The report covers a segment of key events, an executive summary, a descriptive overview of Generalized Myasthenia Gravis (gMG), explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current treatment landscape.
- A detailed review of the Generalized Myasthenia Gravis (gMG) market, historical and forecasted market size, market share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Generalized Myasthenia Gravis (gMG) market.
Generalized Myasthenia Gravis Market Report Insights
- Generalized Myasthenia Gravis (gMG) Patient Population Forecast
- Generalized Myasthenia Gravis (gMG) Therapeutics Market Size
- Generalized Myasthenia Gravis (gMG) Pipeline Analysis
- Generalized Myasthenia Gravis (gMG) Market Size and Trends
- Generalized Myasthenia Gravis (gMG) Market Opportunity (Current and forecasted)
Generalized Myasthenia Gravis Market Report Key Strengths
- Epidemiology-Based (Epi-based) Bottom-up Forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- Generalized Myasthenia Gravis (gMG) Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Generalized Myasthenia Gravis (gMG) Treatment Addressable Market (TAM)
- Generalized Myasthenia Gravis (gMG) Competitve Landscape
- Generalized Myasthenia Gravis (gMG) Major Companies Insights
- Generalized Myasthenia Gravis (gMG) Price trends and Analogue Assessment
- Generalized Myasthenia Gravis (gMG) Therapies Drug Adoption/Uptake
- Generalized Myasthenia Gravis (gMG) Therapies Peak Patient Share analysis
Generalized Myasthenia Gravis Market Report Assessment
- Generalized Myasthenia Gravis (gMG) Current Treatment Practices
- Generalized Myasthenia Gravis (gMG) Unmet Needs
- Generalized Myasthenia Gravis (gMG) Clinical Development Analysis
- Generalized Myasthenia Gravis (gMG) Emerging Drugs Product Profiles
- Generalized Myasthenia Gravis (gMG) Market Attractiveness
- Generalized Myasthenia Gravis (gMG) Qualitative Analysis (SWOT and conjoint analysis)
- Generalized Myasthenia Gravis Market Drivers
- Generalized Myasthenia Gravis Market Barriers
FAQs Related to the Generalized Myasthenia Gravis Market Report:
Generalized Myasthenia Gravis Market Insights
- What was the Generalized Myasthenia Gravis (gMG) market size, the market size by therapies, market share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Generalized Myasthenia Gravis (gMG)?
- What are the disease risks, burdens, and unmet needs of Generalized Myasthenia Gravis (gMG)? What will be the growth opportunities across the 7MM concerning the Generalized Myasthenia Gravis patient population?
- Who is the major future competitor in the market, and how will the competitors affect their market share?
- What are the current options for the treatment of Generalized Myasthenia Gravis (gMG)? What are the current guidelines for treating Generalized Myasthenia Gravis (gMG) in the US, Europe, and Japan?
Reasons to Buy Generalized Myasthenia Gravis Market Forecast Report
- The report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Generalized Myasthenia Gravis (gMG) market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the attribute analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.

-pipeline.png&w=256&q=75)



