human papillomavirus 16 positive hpv 16 cancers epidemiology forecast
HPV16+ Cancers Insights and Trends
- HPV 16 remains the predominant oncogenic type (≥60% in anogenital cancer sites and 44–91% in head and neck cancers). The inclusion of additional markers increases the attributable fraction of HPV 16 in vulvar, penile, oral cavity, and oropharyngeal cancers, and also confirms the causal role of other, less common mucosal HPV types.
- HPV16 status is primarily confirmed via molecular or immunohistochemical testing. p16 overexpression is widely used as a practical screening tool, especially in oropharyngeal cancers, but requires confirmation with HPV-specific assays.
- According to the Centre for Disease Control and Prevention, among all the cases of anal cancer in the United States, about 91% are thought to be attributable to be caused by HPV infection.
- According to the Centre for Disease Control and Prevention, among all the cases of anal cancer in the United States, about 91% are thought to be attributable to be caused by HPV infection.
- Most cases of cervical cancer occur as a result of infection with HPV16 and 18. Type 16 is the cause of approximately 50% of cervical cancers worldwide.
- Approximately 90% of head and neck cancers are accounted for by HNSCC, and its incidence is steadily increasing, largely due to the growing burden of HPV-positive OPSCC in developed countries. At the same time, in developing regions, continued tobacco and lifestyle exposures remain the main cause.
HPV16+ Cancers Epidemiology Forecast in the 7MM
- 2025 HPV16+ Cancers Incident Cases: XXX
- 2036 Projected HPV16+ Cancers Incident Cases: XXX
- HPV16+ Cancers Growth Rate (2026–2036): XXX
DelveInsight's ‘HPV16+ Cancers – Epidemiology Forecast – 2036’ report delivers an in-depth understanding of the HPV16+ Cancers, historical and forecasted epidemiology, in the United States, EU4 (Germany, Spain, Italy, and France) and the United Kingdom, and Japan.
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2026 |
|
Geographies Covered |
|
|
HPV16+ Cancers Epidemiology CAGR (Study period/Forecast period) |
XX% (2026–2036) |
|
HPV16+ Cancers Epidemiology Segmentation Analysis |
Patient Burden Assessment
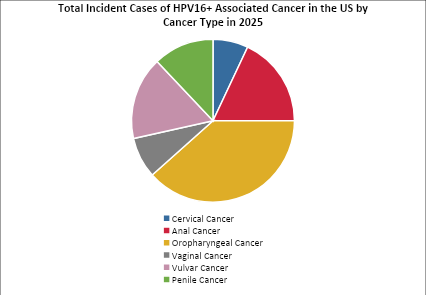
*(Selected indications Cervical Cancer, Anal Cancer, Oropharyngeal Cancer, Vaginal Cancer, Vulvar Cancer, Penile Cancer, and others…) |
HPV16+ Cancers Understanding and Diagnosis Algorithm
HPV16+ Cancers Overview and Diagnosis
HPV16+ cancers are virus-driven epithelial malignancies primarily caused by persistent infection with human papillomavirus type 16, one of the most oncogenic HPV strains responsible for a significant proportion of cervical cancers and head & neck squamous cell carcinomas, particularly oropharyngeal tumors. These cancers arise when viral oncoproteins E6 and E7 disrupt key tumor suppressor pathways, leading to uncontrolled cellular proliferation, genomic instability, and malignant transformation of epithelial cells.
Further details are provided in the report.
HPV16+ Cancers Diagnosis
Diagnosis begins with clinical suspicion based on symptoms or screening findings, followed by confirmation through biopsy and histopathological examination of the suspected lesion, which typically reveals squamous cell carcinoma. Once malignancy is established, HPV16 status is determined using molecular and immunohistochemical methods, most commonly HPV DNA genotyping to directly detect viral genetic material and p16 immunohistochemistry. In some settings, HPV E6/E7 mRNA testing may also be used for more definitive evidence of active viral oncogene expression.
Further details are provided in the report.
HPV16+ Cancers Epidemiology
Key Findings from HPV16+ Cancers Epidemiological Analysis and Forecast
- An estimated 26 million Americans have an oral HPV infection at any given time, of whom approximately 2,600 are infected with HPV16.
- The total incident cases of HPV16+ Oropharyngeal Squamous Cell Carcinoma (OPSCC) in Japan were nearly 3,900 in 2025.
- High-risk HPV types account for nearly 5% of cancers worldwide and are predominantly associated with cervical, anogenital, and oropharyngeal malignancies. Outside these regions, HPV-associated tumors, largely driven by HPV16, remain rare and are typically reported at acral sites such as the hands and feet.
- Most cases of cervical cancer occur as a result of infection with HPV16 and 18. High-risk types, especially HPV16, are found to be highly prevalent in human populations. Type 16 is the cause of approximately 50% of cervical cancers worldwide. The infection is usually transmitted by sexual contact, causing squamous intraepithelial lesions.
Scope of the Report
- The report covers a segment of a descriptive overview of HPV16+ cancers, explaining its causes, signs and symptoms, and pathogenesis.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression.
Report Insights
HPV16+ Cancers Patient Population Forecast
Report Key Strengths
- Epidemiology‑based (Epi‑based) Bottom‑up Forecasting
- 11-year Forecast
- Patient Burden Trends (by geography)
FAQs
- What are the disease risks, burdens, and unmet needs of HPV16+ cancers? What will be the growth opportunities across the 7MM concerning the patient population with HPV16+ Cancers?
- What is the historical and forecasted HPV16+ cancers patient pool in the US, EU4 (Germany, France, Italy, and Spain), the UK, and Japan?
Reasons to Buy
- Insights on patient burden/disease prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- To understand key opinion leaders’ perspectives around the diagnostic challenges to overcome barriers in the future.
- Detailed insights on various factors hampering disease diagnosis and other existing diagnostic challenges.



-cancers-pipeline.png&w=256&q=75)

