Mammography Devices Market Summary
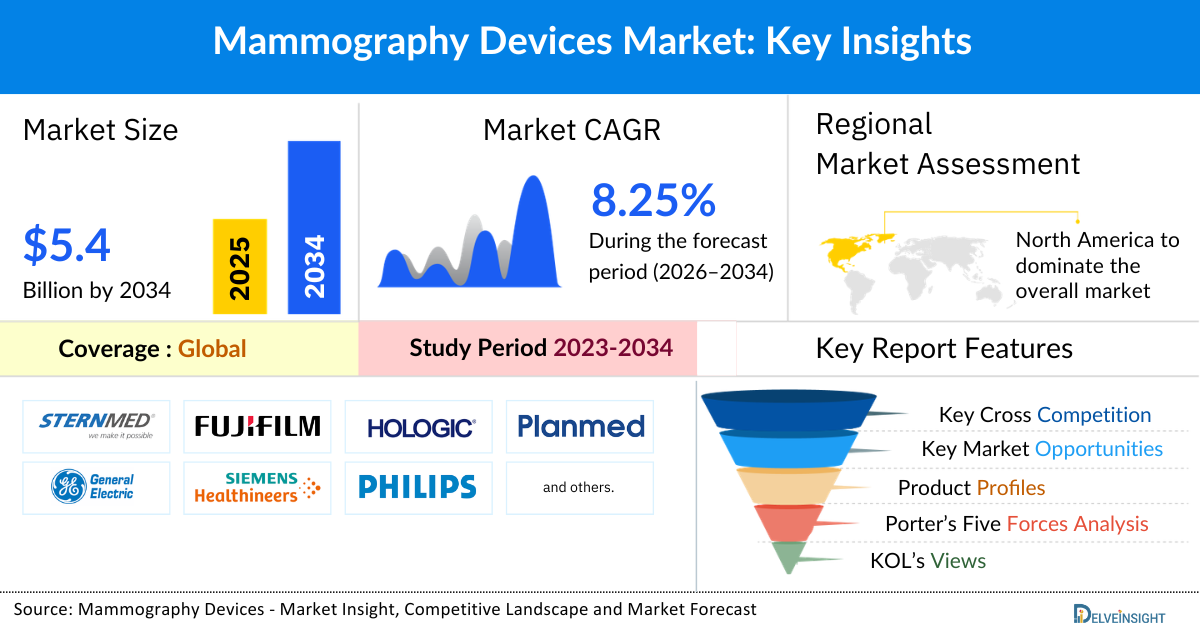
- The global mammography devices market is expected to increase from USD 2,671.77 million in 2025 to USD 5,427.49 million by 2034, reflecting strong and sustained growth.
- The global mammography market is growing at a CAGR of 8.25% during the forecast period from 2026 to 2034.
- The market for mammography devices is primarily driven by the rising incidence of breast cancer, increasing awareness about early detection, and government-led screening programs. Technological advancements such as digital mammography, 3D tomosynthesis, and AI-assisted diagnostics, along with growing healthcare expenditure and aging female populations, further boost adoption. Favorable reimbursement policies and the expansion of private diagnostic centers also contribute to the market’s steady growth.
- The leading companies operating in the mammography devices market include SternMed GmbH, FUJIFILM Corporation, Hologic Inc, Planmed OY, General Electric Company (GE Healthcare), Siemens Healthcare GmbH, Koninklijke Philips NV, DMS Group, IMS Giotto S.P.A, General Medical Merate S.p.A, Konica Minolta Inc, Lanmage, Crestream Health, MEDI-FUTURE Inc, Villa Sistemi Medicali Spa, and others.
- North America is expected to dominate the mammography devices market due to the presence of advanced healthcare infrastructure, high awareness about breast cancer screening, and well-established government screening programs. The widespread adoption of technologically advanced devices such as digital mammography and 3D tomosynthesis, along with strong reimbursement policies and high healthcare spending, further reinforces the region’s leading position in the market.
- In the type of devices segment of the mammography devices market, the digital mammography devices category is estimated to account for the largest market share in 2025.
Request for unlocking the report of the @ Mammography Devices Market
Mammography Devices Market Size and Forecasts
|
Report Metrics |
Details |
|
2025 Market Size |
USD 2,671.77 million |
|
2034 Projected Market Size |
USD 5,427.49 million |
|
Growth Rate (2026-2034) |
8.25% CAGR |
|
Largest Market |
North America |
|
Fastest Growing Market |
Asia-Pacific |
|
Market Structure |
Moderately Concentrated |
Factors Contributing to the Growth of the Mammography Devices Market
- Rising incidence of breast cancer leading to a surge in mammography devices: The rising incidence of breast cancer is driving a surge in demand for mammography devices, as early detection through regular screening becomes increasingly crucial for effective treatment and improved patient outcomes.
- Increasing government initiatives and screening programs are escalating the market of mammography devices: Increasing government initiatives and national breast cancer screening programs are boosting the mammography devices market by promoting early detection and encouraging routine screenings across populations.
- Increase in technological advancement: Advancements in technologies like digital mammography, 3D tomosynthesis, and AI-assisted imaging are boosting the market by improving diagnostic accuracy, efficiency, and patient outcomes.
Mammography Devices Market Report Segmentation
This mammography devices market report offers a comprehensive overview of the global mammography devices market, highlighting key trends, growth drivers, challenges, and opportunities. It covers detailed market segmentation by Type of Devices (Analog, Digital [Full Field Digital Mammography, Breast Tomosynthesis, and Combination Systems]), End User (Hospitals, Specialty Clinics, and Diagnostic Centers), and geography. The report provides valuable insights into the competitive landscape, regulatory environment, and market dynamics across major markets, including North America, Europe, and Asia-Pacific. Featuring in-depth profiles of leading industry players and recent product innovations, this report equips businesses with essential data to identify market potential, develop strategic plans, and capitalize on emerging opportunities in the rapidly growing mammography devices market.
Mammography devices are specialized medical imaging systems designed to capture detailed X-ray images of the breast. They are primarily used for the early detection, diagnosis, and screening of breast cancer and other breast abnormalities. These devices help clinicians identify tumors, cysts, or microcalcifications that may not be palpable during physical examinations. Modern mammography devices include digital mammography, 3D tomosynthesis, and AI-integrated systems, which enhance image clarity, accuracy, and diagnostic efficiency while reducing patient exposure to radiation.
The mammography systems market is primarily driven by the rising incidence of breast cancer, increasing awareness about early detection, and government-led screening programs. Technological advancements such as digital mammography, 3D tomosynthesis, and AI-assisted diagnostics, along with growing healthcare expenditure and aging female populations, further boost adoption. Favorable reimbursement policies and the expansion of private diagnostic centers also contribute to the market’s steady growth.
Get More Insights into the Report @ Mammography Devices Market
What are the latest mammography devices market dynamics and trends?
The rising incidence of breast cancer, coupled with the expansion of organized screening programs, is significantly driving the demand for mammography devices worldwide. According to the International Agency for Research on Cancer (2026), an estimated 2.45 million new cases of breast cancer were reported globally in 2025, with projections indicating a rise to approximately 3.36 million cases by 2045. As one of the most commonly diagnosed cancers among women, breast cancer has prompted a strong global emphasis on early detection and timely diagnosis to improve survival outcomes. Mammography devices remain the primary modality for routine screening, enabling the early identification of tumors, microcalcifications, and other breast abnormalities that may not be detectable through physical examination.
On a global level, initiatives such as the World Health Organization’s Global Breast Cancer Initiative (GBCI) actively promote early detection and structured screening strategies, encouraging countries to implement systematic mammography programs. In the United Arab Emirates, awareness and screening initiatives such as the Pink Caravan campaign, along with government-supported free screening programs, have significantly increased breast cancer screening uptake and the utilization of mammography technologies.
Furthermore, the strong presence and continuous innovation of key market players are accelerating overall market growth. For instance, in November 2025, GE HealthCare announced that it had received FDA Premarket Authorization for Pristina Recon DL, an advanced 3D mammography image reconstruction technology. Powered by deep learning algorithms, Pristina Recon DL enhances image sharpness and definition, setting a new benchmark in diagnostic accuracy for breast imaging.
Collectively, these factors are expected to drive substantial growth in the global mammography devices market during the forecast period from 2026 to 2034.
However, certain challenges may restrain market expansion. Concerns regarding radiation exposure, even at low doses, continue to influence patient perception and screening frequency, potentially limiting device utilization. Additionally, stringent regulatory approval processes, including comprehensive safety and efficacy evaluations by authorities such as the FDA and CE regulatory bodies, can extend time-to-market for new technologies. Together, these factors may moderate the pace of adoption and widespread deployment of advanced mammography systems.
Mammography Devices Market Segment Analysis
Mammography Devices Market Type of Devices (Analog, Digital [Full Field Digital Mammography, Breast Tomosynthesis, and Combination Systems]), End User (Hospitals, Specialty Clinics, And Diagnostic Centers), and Geography (North America, Europe, Asia-Pacific, and Rest of the World)
By Type of Devices: Digital Devices Category Dominates the Market
In the device type segment of the mammography devices market, the digital category accounted for the largest share of approximately 97% in 2025. Within this segment, Full-Field Digital Mammography (FFDM) is estimated to hold the dominant share, driven by its widespread clinical adoption, superior image quality, reduced radiation exposure compared to conventional analog systems, and seamless integration with advanced imaging technologies such as 3D tomosynthesis.
Unlike traditional film-based systems, FFDM captures high-resolution digital images that can be efficiently stored, transmitted, and enhanced for improved visualization. This enables more accurate detection of breast abnormalities, including tumors, cysts, and microcalcifications. The technology also supports computer-aided detection (CAD) and integration with advanced analytics and AI-based tools, enhancing diagnostic precision while reducing false positives and false negatives. Additionally, clinicians can manipulate digital images, such as zooming into specific regions, adjusting contrast, and comparing prior scans, thereby streamlining workflow and facilitating more informed clinical decision-making. Faster image acquisition and processing further improve patient throughput and operational efficiency in hospitals and diagnostic centers.
Ongoing product innovation is also accelerating segment growth. For instance, in April 2024, Siemens Healthineers received FDA 510(k) clearance for its MAMMOMAT B.brilliant mammography system, which includes full-field digital mammography capabilities. This next-generation platform offers enhanced imaging performance along with improved ergonomic design, strengthening the company’s position in the digital mammography space.
Moreover, government initiatives and national breast cancer screening programs are increasingly favoring digital mammography due to its clinical reliability, efficiency, and long-term cost-effectiveness. The growing preference for minimally invasive, precise, and patient-friendly diagnostic solutions, combined with rising breast cancer awareness and expanding healthcare infrastructure across both developed and emerging markets, continues to reinforce FFDM as a key growth driver within the mammography devices market.
By End-Users: Hospitals Category Dominates the Market
In the end-user segment of the mammography equipment medical devices market, the hospitals category is estimated to account for the largest market share in 2025. The hospital segment is a major driver of the mammography devices market, as hospitals represent the primary point of care for breast cancer screening and diagnostics. With the rising incidence of breast cancer and increasing emphasis on early detection, hospitals are investing heavily in advanced imaging technologies such as full-field digital mammography, 3D tomosynthesis, and AI-assisted systems to enhance diagnostic accuracy and patient outcomes. The availability of skilled radiologists, integrated healthcare infrastructure, and high patient footfall allows hospitals to adopt and utilize mammography devices more effectively than standalone diagnostic centers. Moreover, government-supported hospital screening programs and partnerships with private healthcare providers further increase the deployment of mammography equipment in hospital settings. This combination of high demand, technological adoption, and institutional support makes the hospital category a key contributor to the overall growth of the mammography devices market.
Mammography Devices Market Regional Analysis
North America Mammography Devices Market Trends
North America is projected to account for the largest share, approximately 35% of the global mammography devices market in 2025. This dominance is primarily attributed to the region’s advanced healthcare infrastructure, high awareness regarding breast cancer screening, and well-established national screening frameworks. The widespread adoption of technologically advanced systems such as digital mammography and 3D tomosynthesis, supported by favorable reimbursement policies and high per capita healthcare expenditure, further strengthens the region’s leadership position.
According to the International Agency for Research on Cancer (2026), an estimated 323,000 new breast cancer cases were diagnosed in North America in 2025, with projections rising to approximately 384,000 cases by 2045. As breast cancer remains one of the most prevalent cancers among women in the region, there is a strong emphasis on early detection and timely diagnosis to improve survival outcomes. Mammography devices serve as the cornerstone of routine screening programs, enabling early identification of tumors, microcalcifications, and other abnormalities that may not be detectable through physical examination.
In the United States, the United States Preventive Services Task Force updated its breast cancer screening guidelines in April 2024, recommending biennial mammography for women aged 40 to 74 to reduce mortality risk. This policy shift has significantly reinforced clinical demand for mammography technologies. Additionally, federal initiatives such as the National Breast and Cervical Cancer Early Detection Program continue to provide subsidized mammograms and follow-up diagnostic services to low-income and underserved populations, thereby expanding access and increasing screening uptake.
In Canada, the Public Health Agency of Canada, in collaboration with national stakeholders, is actively promoting breast cancer screening equity through awareness campaigns and initiatives aimed at improving access for underserved communities. The Canadian Cancer Society has also intensified nationwide awareness efforts, encouraging eligible women to undergo regular mammography screening and supporting provincial measures to potentially lower the recommended starting age for screening to 40.
Meanwhile, in Mexico, the federal government announced the Universal Care Model for Breast Cancer in October 2025, allocating approximately MX$8 billion toward strengthening early detection, diagnostic, and treatment infrastructure. Biennial mammography screening for women aged 40 and above forms a key component of this initiative, further driving demand for mammography systems across the country.
In addition to policy support, ongoing product innovation continues to propel market growth. For instance, in September 2024, Siemens Healthineers received FDA premarket approval for the tomosynthesis (three-dimensional breast imaging) capability of its Mammomat B.brilliant system, the company’s first fully redesigned mammography platform in over a decade. Such advancements enhance imaging precision, workflow efficiency, and patient comfort, further accelerating technology adoption across North America.
Collectively, these factors, including rising disease burden, supportive screening guidelines, government funding initiatives, and continuous technological advancements, are expected to drive sustained growth of the mammography systems in North America throughout the forecast period.
Europe Mammography Devices Market Trends
Europe is a key growth region in the global mammography devices market, driven by well-established, government-supported breast cancer screening programs, strong awareness regarding early detection, and the rapid adoption of advanced technologies such as digital mammography and 3D tomosynthesis. The region benefits from robust healthcare infrastructure, favorable reimbursement frameworks, and active advocacy efforts by organizations such as Breast Cancer Europe, which promote routine screening and equitable access to diagnostic services. These factors collectively contribute to rising demand for mammography systems across hospitals, specialty clinics, and diagnostic imaging centers.
According to the International Agency for Research on Cancer (2026), approximately 564,000 new breast cancer cases were diagnosed in Europe in 2025, with projections expected to reach nearly 601,000 cases by 2045. The increasing disease burden is significantly intensifying the need for early detection and systematic screening programs. As a result, healthcare providers across the region are expanding their mammography infrastructure and upgrading to advanced digital platforms to enhance diagnostic precision, workflow efficiency, and patient outcomes.
Government-funded population-based screening programs across several European countries play a central role in sustaining market growth. High participation rates, combined with structured recall systems and quality-controlled imaging standards, continue to reinforce the importance of mammography as the primary diagnostic modality for breast cancer screening. Furthermore, hospitals and diagnostic centers are investing in next-generation imaging systems, including AI-integrated solutions, to improve sensitivity and reduce false-positive rates.
In line with ongoing technological advancements, in January 2026, Planmed Oy announced that its Planmed Clarity+ mammography product line had received CE MDR (Medical Device Regulation) certification, enabling commercialization across the European market. Such regulatory approvals strengthen product availability and accelerate the transition toward modern, high-performance mammography platforms in the region.
Overall, the combination of rising breast cancer incidence, structured screening frameworks, supportive regulatory policies, and continuous product innovation is expected to drive sustained growth of the mammography devices market in Europe over the forecast period.
Asia-Pacific Mammography Devices Market Trends
The Asia-Pacific region is emerging as a key growth driver in the mammography devices market due to rising breast cancer incidence, growing awareness of early detection, and expanding government-led screening programs. Rapidly developing healthcare infrastructure, increasing hospital investments in advanced digital and 3D mammography systems, and rising disposable incomes are further boosting adoption, making the region one of the fastest-growing markets globally.
According to statistics published by the International Agency for Research on Cancer (2026), an estimated 1.0 million new cases of breast cancer were diagnosed in Asia in 2025. These cases are projected to increase to approximately 1.38 million by 2045, reflecting a substantial rise in disease burden across the region.
However, the increase in product development activites are further boosting the overall market of mammography devices. For instance, in October 2025, Siemens Healthineers launched the MAMMOMAT B.brilliant 3D mammography system in Thailand, offering advanced breast imaging with high‑resolution and fast scanning to support early detection of breast cancer.
Thus, the factors mentioned are expected to boost the overall market of mammography devices in the Asia-Pacific region during the forecast period.
Who are the major players in the mammography devices market?
The following are the leading companies in the mammography devices market. These companies collectively hold the largest market share and dictate industry trends.
- SternMed GmbH
- FUJIFILM Corporation
- Hologic Inc.
- Planmed OY
- General Electric Company (GE Healthcare)
- Siemens Healthcare GmbH
- Koninklijke Philips NV
- DMS Group
- IMS Giotto S.P.A
- General Medical Merate S.p.A
- Konica Minolta Inc.
- Lanmage
- Crestream Health
- MEDI-FUTURE Inc.
- Villa Sistemi Medicali Spa
- Others
How is the competitive landscape shaping the mammography devices market?
The competitive landscape of the mammography devices market is marked by the presence of a mix of large, well‑established imaging companies and specialized regional players, resulting in a moderately concentrated market at the global level. Major multinational vendors compete based on technological innovation, product portfolio breadth, and strategic partnerships, with a strong focus on digital mammography, 3D tomosynthesis, and AI‑enabled imaging enhancements. These leading companies often secure CE, FDA, and other regional approvals first, reinforcing their market positions and creating higher entry barriers for smaller firms. At the same time, a growing number of local and regional manufacturers, particularly in Asia‑Pacific and emerging markets, are expanding into the space with cost‑effective solutions tailored to local healthcare needs. Competitive strategies such as collaborations with healthcare providers, training programs for radiologists, service contracts, and bundled software offerings further shape differentiation. As a result, the market shows both consolidation around key global players and intensifying competition from agile regional entrants, collectively driving advancements in product features, geographic reach, and customer support services.
Recent Developmental Activities in the Mammography Devices Market
- In January 2026, Planmed Oy announced that its Planmed Clarity+ mammography product line had received CE MDR (Medical Device Regulation) certification, making it available for the European market.
- In November 2025, GE HealthCare announced that it had received FDA Premarket Authorization for Pristina Recon DL, an advanced 3D mammography image reconstruction technology. Powered by deep learning, Pristina Recon DL sets a new standard in image definition and sharpness.
- In October 2025, Siemens Healthineers launched the MAMMOMAT B.brilliant 3D mammography system in Thailand, offering advanced breast imaging with high‑resolution and fast scanning to support early detection of breast cancer.
- In September 2024, Siemens Healthineers announced the Food and Drug Administration’s premarket approval for the tomosynthesis or three-dimensional breast imaging technology of the Mammomat B.brilliant the first completely redesigned Siemens Healthineers mammography platform in over a decade.
- In April 2024, Siemens Healthineers received FDA 510(k) clearance for its MAMMOMAT B.brilliant mammography system, including full‑field digital mammography capabilities. This clearance introduces a next‑generation mammography platform with enhanced imaging and ergonomic features.
|
Report Metrics |
Details |
|
Study Period |
2023 to 2034 |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2034 |
|
Mammography Devices Market CAGR (2026-2034) |
8.25% |
|
Key Companies in the Mammography Devices Market |
SternMed GmbH, FUJIFILM Corporation, Hologic Inc, Planmed OY, General Electric Company (GE Healthcare), Siemens Healthcare GmbH, Koninklijke Philips NV, DMS Group, IMS Giotto S.P.A, General Medical Merate S.p.A, Konica Minolta Inc, Lanmage, Crestream Health, MEDI-FUTURE Inc, Villa Sistemi Medicali Spa, and others. |
|
Mammography Devices Market Segments |
by Type of Devices, by End-Users, and by Geography |
|
Mammography Devices Regional Scope |
North America, Europe, Asia Pacific, the Middle East, Africa, and South America |
|
Mammography Devices Country Scope |
U.S., Canada, Mexico, Germany, United Kingdom, France, Italy, Spain, China, Japan, India, Australia, South Korea, and key Countries |
Mammography Devices Market Segmentation
- Mammography Devices by Type of Device Exposure
- Analog Mammography Devices
- Digital Mammography Devices
- Full Field Digital Mammography Devices
- Breast Tomosynthesis Devices
- Combination Systems
- Mammography Devices End-Users Exposure
- Hospitals
- Specialty Clinics
- Others
- Mammography Devices Geography Exposure
- North America Mammography Devices Market
- United States Mammography Devices Market
- Canada Mammography Devices Market
- Mexico Mammography Devices Market
- Europe Mammography Devices Market
- United Kingdom Mammography Devices Market
- Germany Mammography Devices Market
- France Mammography Devices Market
- Italy Mammography Devices Market
- Spain Mammography Devices Market
- Rest of Europe Mammography Devices Market
- Asia-Pacific Mammography Devices Market
- China Mammography Devices Market
- Japan Mammography Devices Market
- India Mammography Devices Market
- Australia Mammography Devices Market
- South Korea Mammography Devices Market
- Rest of Asia-Pacific Mammography Devices Market
- Rest of the World Mammography Devices Market
- South America Mammography Devices Market
- Middle East Mammography Devices Market
- Africa Mammography Devices Market
- North America Mammography Devices Market
Mammography Devices Market Recent Industry Trends and Milestones (2022-2026)
|
Category |
Key Developments |
|
Mammography Devices Regulatory Approvals |
GE HealthCare - Pristina Recon DL (FDA), Siemens Healthineers - MAMMOMAT B.brilliant mammography system (FDA), Planmed Oy - Planmed Clarity+ mammography CE MDR (Medical Device Regulation) |
|
Company Strategy |
Hologic, Inc. (USA) – Focuses on technological leadership with its advanced 3D breast tomosynthesis and AI‑enabled systems (e.g., 3Dimensions, Selenia Dimensions) to enhance cancer detection accuracy and workflow efficiency. Siemens Healthineers (Germany) – Leverages cutting‑edge imaging technologies, including 3D mammography and contrast‑enhanced mammography, with a focus on AI‑enhanced image processing and workflow optimization. |
|
Emerging Technology |
3D Digital Breast Tomosynthesis (DBT), Contrast‑Enhanced Mammography (CEM), Artificial Intelligence (AI) and Machine Learning, Automated Breast Ultrasound (ABUS) Integration, Photon‑Counting Mammography, and others. |
Impact Analysis
AI-Powered Innovations and Applications:
AI-powered innovations in mammography devices are transforming breast cancer screening and diagnostics by enhancing accuracy, efficiency, and personalization. Advanced AI algorithms, particularly deep learning models, are now integrated into digital mammography and 3D tomosynthesis systems to assist radiologists in detecting subtle lesions, microcalcifications, and early-stage tumors that may be missed during manual review. AI applications include automated image analysis, risk assessment, and predictive modeling to identify high-risk patients and prioritize cases for further examination. These systems can also reduce false positives and unnecessary biopsies, improving patient experience and workflow efficiency in clinical settings. Additionally, AI-driven tools enable continuous learning from large datasets, helping devices adapt to diverse patient populations and breast tissue densities, thereby enhancing diagnostic confidence and overall screening outcomes. Beyond detection, AI integration supports reporting automation, workflow optimization, and remote monitoring, making mammography more precise, accessible, and scalable across healthcare systems.
U.S. Tariff Impact Analysis on Mammography Devices Market:
The U.S. tariff impact on the mammography devices market mirrors broader trends in the medical device sector, where trade policy shifts are reshaping costs, supply chains, and competitive dynamics. Recent U.S. tariff actions on imports of medical equipment and components implemented as part of wider trade and “Section 232” national security reviews have increased the cost of diagnostic imaging technologies that form part of the mammography ecosystem because many devices and parts are sourced globally. Higher duties on imported components such as advanced electronics, sensors, and high‑precision materials can elevate production expenses for mammography manufacturers that rely on cross‑border supply chains, leading to compressed profit margins, potential price hikes for buyers, and slower capital expenditure by healthcare providers. These cost pressures can also slow the adoption of new or upgraded mammography systems as hospitals and imaging centers adjust procurement planning in response to tariff‑induced uncertainty. Moreover, tariff volatility can deter investment in innovation and complicate global manufacturing strategies, prompting some firms to consider relocating or expanding domestic production to mitigate exposure. While domestic manufacturing offers some protection for U.S.-made products, many key components still originate abroad, leaving even established diagnostic imaging segments like mammography susceptible to tariff impacts and supply chain disruptions.
How This Analysis Helps Clients
- Cost Management: By understanding the tariff landscape, clients can anticipate cost increases and adjust pricing strategies accordingly, ensuring profitability.
- Supply Chain Optimization: Clients can identify alternative sourcing options and diversify their supply chains to reduce dependency on high-tariff regions, enhancing resilience.
- Regulatory Navigation: Expert guidance on navigating the evolving regulatory environment helps clients maintain compliance and avoid potential legal challenges.
- Strategic Planning: Insights into tariff impacts enable clients to make informed decisions about manufacturing locations, partnerships, and market entry strategies.
Startup Funding & Investment Trends
|
Company Name |
Total Funding |
Main Products |
Stage of Development |
Core Technology |
|
Clairity, Inc. |
$43 Million |
Clairity Breast |
Series B |
It is the first FDA‑authorized artificial intelligence platform designed to predict a woman’s five‑year breast cancer risk directly from routine mammograms. |
Key takeaways from the Mammography Devices market report study
- Market size analysis for the current mammography devices market size (2025), and market forecast for 8 years (2026 to 2034)
- Top key product/technology developments, mergers, acquisitions, partnerships, and joint ventures happened over the last 3 years.
- Key companies dominating the mammography devices market.
- Various opportunities available for the other competitors in the mammography devices market space.
- What are the top-performing segments in 2025? How these segments will perform in 2034?
- Which are the top-performing regions and countries in the current mammography devices market scenario?
- Which are the regions and countries where companies should have concentrated on opportunities for the mammography devices market growth in the future?