Neuroendocrine Carcinoma Market Summary
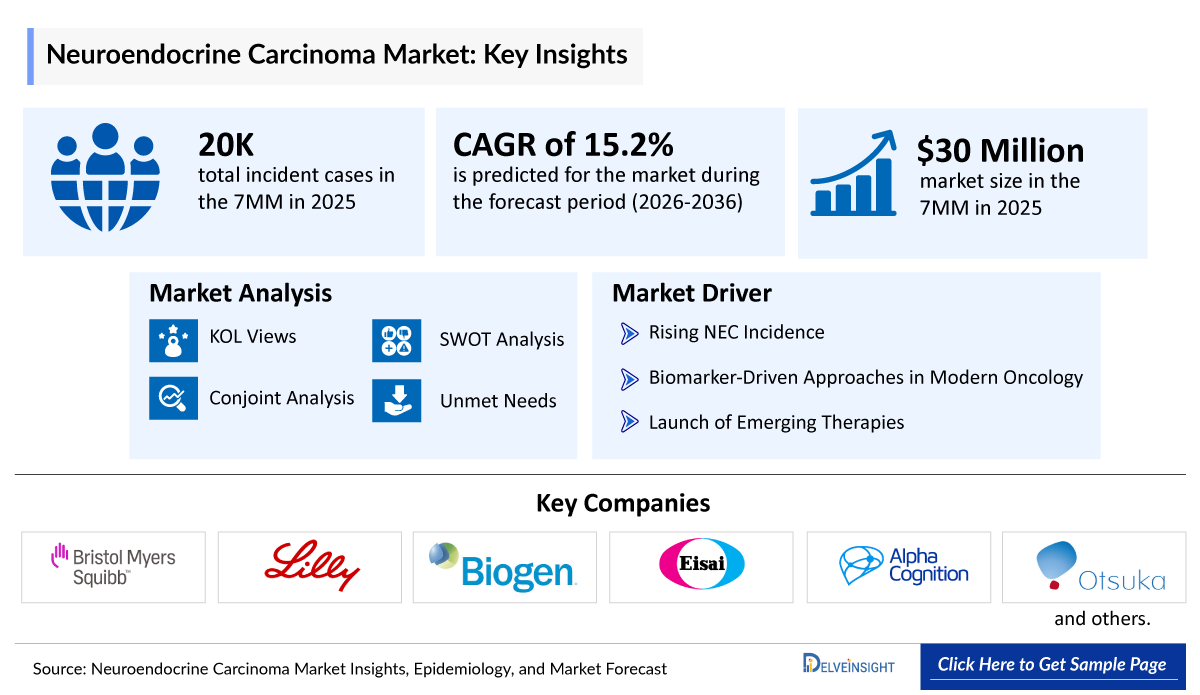
- According to DelveInsight’s analysis, the Neuroendocrine Carcinoma Market Size was nearly USD 30 million in the leading markets (the United States, the EU4 (Germany, France, Italy, and Spain), the United Kingdom, and Japan) in 2025.
- The leading Neuroendocrine Carcinoma Companies such as Boehringer Ingelheim/ Oxford BioTherapeutics, Amgen, Zai Lab/MediLink Therapeutics, Merck/ Daiichi Sankyo, Vyriad and others.
Neuroendocrine Carcinoma Market Insights and Trends
- Neuroendocrine carcinoma is a rare, aggressive malignancy arising from neuroendocrine cells found throughout the body; this report focuses exclusively on extrapulmonary Neuroendocrine Carcinoma, excluding pulmonary forms, to provide a more precise and targeted analysis of disease characteristics, trends, and clinical treatment insights.
- Across the 7MM, the United States represents the largest Neuroendocrine Carcinoma Treatment Market with approximately USD 20 million, accounting for the majority of Neuroendocrine Carcinoma cases in 2025 and expected to maintain its leading position through 2036, supported by higher diagnostic awareness, advanced oncology infrastructure, and greater participation in clinical research programs.
- In 2025, the United States reported approximately 30,000 Neuroendocrine Carcinoma Incident Cases, including around 9,000 cases of Neuroendocrine Carcinoma, reflecting a notable proportion of aggressive malignancies within the broader NEN landscape, with nearly 950 cases attributed to extrapulmonary Neuroendocrine Carcinoma.
- Currently, there are no specifically approved Neuroendocrine Carcinoma Marketed Drugs. Neuroendocrine Carcinoma Treatment strategies largely rely on conventional approaches such as platinum-based chemotherapy regimens, including cisplatin or carboplatin combined with etoposide. In selected cases, surgical resection and radiation therapy may be used to manage localized disease or control tumor growth.
- Wider utilization of advanced imaging modalities and specialized pathology services is improving the detection and clinical characterization of Neuroendocrine Carcinoma, enabling more accurate confirmation of neuroendocrine differentiation and disease staging.
- While several investigational therapies are being explored for extrapulmonary Neuroendocrine Carcinoma, key emerging candidates include obrixtamig (BI 764532), Tarlatamab-dlle (IMDELLTRA), zocilurtatug pelitecan (ZL-1310), gocatamig (MK-6070), voyager-V1 in combination with pembrolizumab, peluntamig (PT217), IDE849, MT-4561, and clesitamig (RO7616789), reflecting a growing and diverse early- to mid-stage pipeline that may contribute to future advancements in extrapulmonary Neuroendocrine Carcinoma Treatment.
- Despite ongoing research efforts, Neuroendocrine Carcinoma remains associated with major clinical challenges, including aggressive tumor biology, frequent late-stage diagnosis, high mortality, and a limited late-stage pipeline, underscoring persistent unmet needs in the field.
Neuroendocrine Carcinoma Market Size and Forecast
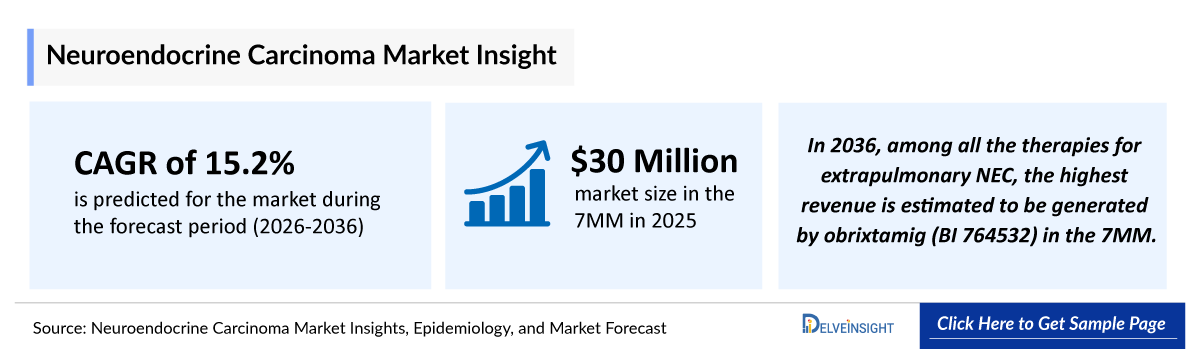
- 2025 Neuroendocrine Carcinoma Market Size in the 7MM: ~USD 30 million
- 2036 Projected Neuroendocrine Carcinoma Market Size: ~USD 120 million
- Neuroendocrine Carcinoma Growth Rate (2026–2036): 15.2% CAGR (Compound Annual Growth Rate)
Request for Unlocking the Sample Page of the "Neuroendocrine Carcinoma Treatment Market"
Key Factors Driving the Neuroendocrine Carcinoma Market Growth
-
Increasing Incidence of Neuroendocrine Malignancies
Rising cases of aggressive neuroendocrine tumors and carcinomas are significantly contributing to market growth worldwide.
-
Advancements in Diagnostic Technologies
Improved imaging techniques, biomarker testing, PET scans, and molecular diagnostics are enabling earlier and more accurate diagnosis.
-
Growing Adoption of Targeted Therapies
Increasing use of targeted therapies and precision medicine approaches is improving treatment outcomes for patients with neuroendocrine carcinoma.
-
Expansion of Immunotherapy Applications
The growing role of immune checkpoint inhibitors and combination immunotherapies is creating new treatment opportunities in the market.
-
Strong Pipeline of Emerging Therapies
Pharmaceutical and biotechnology companies are actively developing innovative therapies targeting tumor growth pathways and molecular alterations.
-
Increasing Research and Development Investments
Rising investments in oncology research and rare cancer therapeutics are accelerating clinical development activities.
-
Growing Awareness and Early Detection
Increased awareness among healthcare professionals and patients is supporting timely diagnosis and treatment initiation.
-
Rising Demand for Personalized Medicine
Biomarker-driven therapies and individualized treatment approaches are enhancing therapeutic effectiveness and patient management.
DelveInsight's ‘Neuroendocrine Carcinoma Market Insights, Epidemiology and Market Forecast – 2036’ report delivers an in-depth understanding of Neuroendocrine Carcinoma, historical and forecasted epidemiology, as well as the Neuroendocrine Carcinoma market trends in the United States, EU4 (Germany, Spain, Italy, and France), the United Kingdom, and Japan.
The Neuroendocrine Carcinoma Treatment Market Report delivers a comprehensive analysis of the current treatment landscape, including standards of care, clinical practices, and evolving therapeutic algorithms. It evaluates, Neuroendocrine Carcinoma patient burden trends, revenue & market share dynamics, peak patient share & therapy uptake analysis, and provides an in-depth market size assessment, and growth rate projections (Historical & Forecast 2022–2036) across the 7MM regions. The report highlights key unmet medical needs in Neuroendocrine Carcinoma and maps the competitive and clinical landscape to uncover high-value opportunities, providing a clear outlook on future market growth potential.
Scope of the Neuroendocrine Carcinoma Market Report | |
|
Study Period |
2022–2036 |
|
Historical Year |
2022–2025 |
|
Forecast Period |
2026–2036 |
|
Base Year |
2025 |
|
Geographies Covered |
|
|
Neuroendocrine Carcinoma Market CAGR | |
|
Neuroendocrine Carcinoma Epidemiology Segmentation Analysis |
Patient Burden Assessment
|
|
Neuroendocrine Carcinoma Companies |
|
|
Neuroendocrine Carcinoma Emerging Therapies |
|
|
Neuroendocrine Carcinoma Market |
Segmented by
|
|
Analysis |
|
Neuroendocrine Carcinoma Understanding and Treatment Algorithm
Neuroendocrine Carcinoma a rare and highly aggressive malignancy that arises from neuroendocrine cells, which are specialized cells possessing characteristics of both nerve cells and hormone-producing endocrine cells. These cells are widely distributed throughout the body, particularly in organs such as the lungs, gastrointestinal tract, pancreas, and other endocrine tissues. Genetic and molecular alterations within neuroendocrine cells may disrupt normal cellular regulation, resulting in uncontrolled proliferation, tumor formation, and the potential for local invasion and distant metastasis.
Neuroendocrine Carcinoma Diagnosis involves a comprehensive and multidisciplinary evaluation that combines clinical assessment, radiologic imaging, and histopathological confirmation. Because Neuroendocrine Carcinoma often presents with non-specific clinical manifestations and can arise in several organs such as the lung, gastrointestinal tract, pancreas, and other neuroendocrine tissues, establishing an accurate diagnosis requires careful integration of clinical findings, imaging results, and tissue-based pathological examination. The aggressive nature of Neuroendocrine Carcinoma, which is characterized by rapid tumor growth, early metastatic spread, and high proliferative activity, makes early and accurate identification particularly important for understanding disease progression and biological behavior.
Current Neuroendocrine Carcinoma Treatment Landscape
The therapeutic management of Neuroendocrine Carcinoma often involves a combination of systemic and local treatment approaches depending on tumor characteristics and disease stage. The integration of chemotherapy, surgery in selected cases, radiation therapy, and emerging immunotherapeutic strategies reflects the complexity of managing this aggressive malignancy. Neuroendocrine Carcinoma treatment landscape is largely dominated by off-label systemic therapies, with etoposide plus cisplatin or carboplatin chemotherapy forming the backbone, alongside somatostatin analogs, everolimus, immunotherapy, and other chemotherapy regimens, reflecting the absence of disease-specific approved treatments.
Neuroendocrine Carcinoma Unmet Needs
The section “unmet needs of Neuroendocrine Carcinoma” outlines the critical gaps between the current state of patient care, diagnosis, and the ideal & effective management of the disease. It highlights the obstacles experienced by patients, clinicians, and researchers and identifies potential solutions for future progress.
- Delayed and Complex Diagnosis Continues to Obscure Early Identification of Neuroendocrine Carcinoma
- Limited Therapeutic Effectiveness Reflects the Biological Complexity of Neuroendocrine Carcinoma
- Scarcity of Targeted Therapies Highlights a Significant Innovation Gap in Neuroendocrine Carcinoma
Neuroendocrine Carcinoma Epidemiology Insights in the 7MM
Key Findings from Neuroendocrine Carcinoma Epidemiological Analysis and Forecast
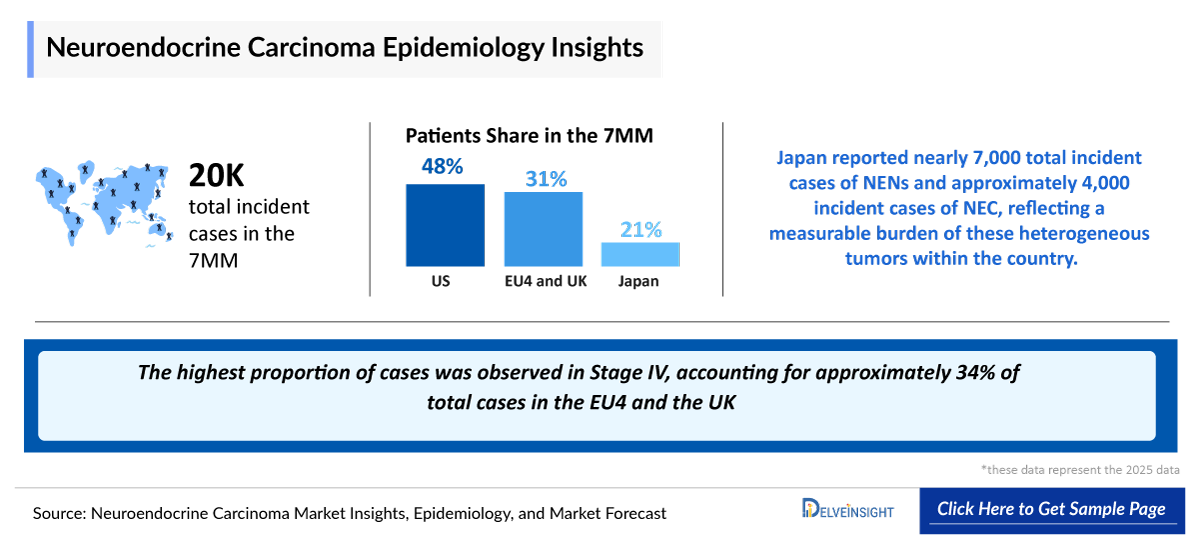
- In 2025, the total number of Neuroendocrine Carcinoma Incident Cases across the 7MM was approximately 20,000 cases and it is projected to rise due to improved pathological evaluation and broader clinical awareness of high-grade neuroendocrine malignancies.
- In 2025, Japan reported nearly 7,000 total Neuroendocrine Carcinoma Incident Cases and approximately 4,000 incident cases of Neuroendocrine Carcinoma, reflecting a measurable burden of these heterogeneous tumors within the country.
- In 2025, the total number of Neuroendocrine Carcinoma Incident Cases in the United States was approximately 950 cases and is projected to increase by 2036, reflecting gradual changes in disease detection and reporting.
- In EU4 and the UK, the extrapulmonary Neuroendocrine Carcinoma burden in 2025 was supported by a considerable number of cases, driven by improved diagnostic capabilities, increased awareness of rare neuroendocrine malignancies, and broader access to specialized oncology services. The highest proportion of cases was observed in Stage IV, accounting for approximately 34% of total cases in 2025.
- In Japan, the extrapulmonary Neuroendocrine Carcinoma burden in 2025 showed a higher occurrence of small-cell histology with approximately 280 cases compared with large-cell Neuroendocrine Carcinoma with around 130 cases. This distribution highlights the predominance of the small-cell subtype within the Neuroendocrine Carcinoma histological spectrum in the region.
- In 2025, the number of incident cases of extrapulmonary Neuroendocrine Carcinoma across EU4 and the UK was approximately 620, with UK accounting for the largest share of approximately 250 cases.
Neuroendocrine Carcinoma Epidemiology Segmentation in the 7MM
- Total Neuroendocrine Carcinoma Incident Cases
- Total Incident Cases of Extrapulmonary
- Neuroendocrine Carcinoma Stage-specific Cases
- Neuroendocrine Carcinoma Site-specific Cases
- Neuroendocrine Carcinoma Gender-specific Cases
- Neuroendocrine Carcinoma Histology-specific Cases
- Total Neuroendocrine Carcinoma Treated Cases
Neuroendocrine Carcinoma Drug Analysis & Competitive Landscape
The Neuroendocrine Carcinoma drug chapter provides a detailed, market-focused review of the emerging pipeline across Phase I-II Neuroendocrine Carcinoma Clinical Trials. It covers Neuroendocrine Carcinoma Mechanism of Action, clinical trial data, regulatory approvals, patents, collaborations, strategic partnerships upcoming key catalyst for each therapy, along with their advantages, limitations, and recent developments. This section offers critical insights into the Neuroendocrine Carcinoma treatment landscape, supporting market assessment, competitive analysis, and growth forecasting for the Neuroendocrine Carcinoma therapeutics market.
Neuroendocrine Carcinoma Pipeline Analysis
-
Obrixtamig (BI 764532): Boehringer Ingelheim/Oxford BioTherapeutics
Obrixtamig (BI 764532, OBT620) is an investigational bispecific T-cell engager immunotherapy developed by Boehringer Ingelheim for Small Cell Lung Cancer, an aggressive subtype frequently associated with Neuroendocrine Carcinoma of the lung. This dual-targeting mechanism aims to enhance immune-mediated tumor cell elimination and represents a targeted therapeutic approach for patients with DLL3-expressing small cell lung cancer, where effective options remain limited.
-
- In July 2025, Phase I dose-escalation results for obrixtamig (BI 764532), a DLL3/CD3 IgG-like T-cell engager, were reported in patients with DLL3-positive SCLC or Neuroendocrine Carcinoma.
Competitive Landscape of Neuroendocrine Carcinoma Pipeline Drugs | ||||||
|
Drug Name |
Company |
Highest Phase |
Indication |
RoA |
MoA |
Anticipated Launch in the US |
|
Obrixtamig (BI 764532) |
Boehringer Ingelheim/Oxford BioTherapeutics |
II |
Extra-pulmonary Neuroendocrine Carcinoma |
IV infusion |
DLL3 × CD3 bispecific antibody |
2030 |
|
Tarlatamab (IMDELLTRA) |
Amgen |
II |
NEPC; GEP-Neuroendocrine Carcinoma |
IV infusion |
DLL3 × CD3 bispecific antibody | Information is available in the full report |
|
Zocilurtatug pelitecan (ZL-1310) |
Zai Lab/MediLink Therapeutics |
I/II |
Neuroendocrine Carcinoma |
IV infusion |
DLL3-targeted ADC |
Information is available in the full report |
|
Gocatamig (MK-6070) |
Merck/ Daiichi Sankyo |
I/II |
NEPC |
IV infusion |
DLL3-targeted T cell activation |
Information is available in the full report |
|
Note: Launch insights are provisional and may change with future report updates or the occurrence of major key catalysts. | ||||||
Neuroendocrine Carcinoma Companies, Market Leaders and Emerging Companies
- Boehringer Ingelheim /Oxford BioTherapeutics
- Amgen
- Zai Lab/MediLink Therapeutics
- Merck/ Daiichi Sankyo
- Phanes Therapeutics
Neuroendocrine Carcinoma Drug Updates
- In February 2026, Merck presented data at the 2026 American Society of Clinical Oncology (ASCO) GU Symposium, including Phase II efficacy and safety results of the DLL3 T-cell engager gocatamig in NEPC and other neuroendocrine neoplasms, highlighting its oncology pipeline advancement.
- In October 2025, Zai Lab reported that its Neuroendocrine Carcinoma programs are advancing toward the registrational Phase in 2026, reflecting continued clinical progress in DLL3-targeted therapies for high-grade neuroendocrine malignancies.
- In July 2025, Phase I dose-escalation results for obrixtamig (BI 764532), a DLL3/CD3 IgG-like T-cell engager, were reported in patients with DLL3-positive SCLC and Neuroendocrine Carcinoma.
Neuroendocrine Carcinoma Market Outlook
In the modern Neuroendocrine Carcinoma ecosystem, there are no specifically approved therapies for Neuroendocrine Carcinoma, and clinical practice largely relies on off-label systemic approaches. Current strategies commonly include chemotherapy regimens such as etoposide combined with cisplatin or carboplatin, along with somatostatin analogs, mTOR pathway inhibitors such as everolimus, and immunotherapy-based approaches, alongside other supportive systemic options. This reliance on off-label modalities highlights the significant unmet need for disease-specific therapeutic innovations in Neuroendocrine Carcinoma. Together, these off-label systemic approaches, including chemotherapy, somatostatin analogs, mTOR inhibitors, and immunotherapy-based strategies, constitute the current clinical landscape for Neuroendocrine Carcinoma, reflecting the absence of disease-specific approved therapies.
- In 2025, the extrapulmonary Neuroendocrine Carcinoma Treatment Market across the 7MM was approximately USD 30 million. The US accounted for about 70% of total revenues, followed by EU4 and the UK.
- In 2036, among all the therapies for extrapulmonary Neuroendocrine Carcinoma, the highest revenue is estimated to be generated by obrixtamig (BI 764532) in the 7MM.
- Japan’s extrapulmonary Neuroendocrine Carcinoma Treatment Market is demonstrating steady growth, supported by increasing clinical awareness, improving diagnostic capabilities, and growing recognition of high-grade neuroendocrine malignancies.
- In EU4 and the UK, the extrapulmonary Neuroendocrine Carcinoma market surpassed USD 5 million in 2025, reflecting a modest but notable presence, and is anticipated to experience steady growth, with values expected to increase further by 2036.
Neuroendocrine Carcinoma Drug Class/Insights into Leading Emerging Therapies (2022–2036 Forecast)
Neuroendocrine Carcinoma management is evolving beyond conventional cytotoxic chemotherapy toward a broader platform integrating platinum-based regimens, emerging immunotherapies, and targeted approaches aimed at improving tumor control, expanding therapeutic options, and addressing the aggressive biology and limited durability of responses. Cisplatin + Etoposide: This platinum-based chemotherapy combination is widely used as a first-line systemic regimen for advanced Neuroendocrine Carcinoma due to its ability to induce rapid tumor responses in highly proliferative tumors. However, responses are often short-lived, with many patients experiencing relapse within months, highlighting the need for more durable treatment strategies.
Carboplatin + Etoposide: Frequently used as an alternative platinum-based regimen, particularly in patients who may not tolerate cisplatin. This combination provides comparable tumor control while offering a more manageable safety profile in certain patient populations with advanced Neuroendocrine Carcinoma. Irinotecan + Cisplatin: A chemotherapy combination sometimes used in selected Neuroendocrine Carcinoma cases, particularly in high-grade disease. This regimen has demonstrated activity in rapidly proliferating neuroendocrine tumors but remains associated with substantial toxicity and variable response durability.
Temozolomide + Capecitabine: This oral chemotherapy combination is occasionally used in later-line settings for neuroendocrine malignancies, including selected Neuroendocrine Carcinoma cases. It offers a convenient administration route and has demonstrated antitumor activity in certain neuroendocrine tumor subtypes. Topotecan: Originally developed for small-cell lung cancer, this topoisomerase I inhibitor is sometimes used as a subsequent-line systemic therapy in Neuroendocrine Carcinoma after platinum-based chemotherapy failure, although clinical benefit is generally limited.
Nivolumab: An immune checkpoint inhibitor targeting PD-1 that has been explored off-label in Neuroendocrine Carcinoma. While immunotherapy has shown activity in certain neuroendocrine malignancies, responses in Neuroendocrine Carcinoma remain variable, and patient selection remains challenging. Pembrolizumab: Another PD-1–targeting immune checkpoint inhibitor investigated in selected Neuroendocrine Carcinoma patients, particularly in tumors with specific biomarkers such as high tumor mutational burden or microsatellite instability. Its role in Neuroendocrine Carcinoma remains under investigation in clinical studies.
Neuroendocrine Carcinoma Drug Uptake
This section focuses on the uptake rate of potential Neuroendocrine Carcinoma drugs expected to be launched in the market during the forecast period (2026–2036). The analysis covers the Neuroendocrine Carcinoma drug’s uptake, performance at peak, factors affecting performance during prime years of growth, patient uptake by therapy, and anticipated sales generated by each drug. The Neuroendocrine Carcinoma landscape is largely dominated by off-label systemic therapies, with etoposide plus cisplatin or carboplatin chemotherapy forming the backbone, alongside somatostatin analogs, everolimus, immunotherapy, and other chemotherapy regimens, reflecting the absence of disease-specific approved treatments.
Tarlatamab-dlle (IMDELLTRA) – Developed by Amgen, this Phase II DLL3 × CD3 bispecific antibody targets NEPC and GEP-Neuroendocrine Carcinoma. Administered via IV infusion, it redirects T cells toward DLL3-expressing tumor cells, enabling immune-mediated cytotoxicity, with early data indicating manageable safety and improved responses in DLL3-positive patients. IMDELLTRA, the first and only DLL3-targeting BiTE therapy, is anticipated to enter the US market by medium update, with an estimated annual treatment cost of approximately USD 230,000, reflecting its positioning as a high-cost, targeted therapeutic option.
Neuroendocrine Carcinoma Market Access and Reimbursement of Therapies
The report further provides detailed insights on the country-wise accessibility and reimbursement scenarios, cost-effectiveness scenario of therapies, programs making accessibility easier and out-of-pocket costs more affordable, insights on patients insured under federal or state government prescription drug programs, etc. Reimbursement is a crucial factor that affects the drug’s access to the market. Often, the decision to reimburse comes down to the price of the drug relative to the benefit it produces in treated patients. To reduce the healthcare burden of these high-cost therapies, many payment models are being considered by payers and other industry insiders.
Neuroendocrine Carcinoma Standard of Care
Treatment of Neuroendocrine Carcinoma generally involves multiple therapeutic strategies because the disease is typically aggressive, rapidly proliferating, and frequently diagnosed at an advanced stage. The selection of therapeutic approaches depends on several factors, including the primary tumor location, stage of the disease, extent of metastasis, and the overall clinical condition of the patient. A combination of different treatment approaches is therefore commonly used to control tumor growth and limit disease progression.
Chemotherapy: Systemic chemotherapy represents one of the primary therapeutic approaches used in Neuroendocrine Carcinoma, particularly in patients with advanced or metastatic disease. High-grade Neuroendocrine Carcinoma tumors are characterized by rapid cellular proliferation, making them more responsive to cytotoxic chemotherapy agents that target dividing cells. Platinum-based chemotherapy regimens are most frequently used, typically involving cisplatin or carboplatin in combination with etoposide.
Surgery: Surgical resection may be considered in certain patients when the tumor is localized and can be completely removed. The goal of surgery is to excise the primary tumor along with surrounding affected tissues in order to achieve local disease control and prevent further tumor spread. In some situations, surgery may also involve removal of nearby lymph nodes if regional lymphatic involvement is suspected.
Radiation therapy: It is another therapeutic modality that may be used in the management of Neuroendocrine Carcinoma. This treatment uses high-energy radiation to damage tumor DNA and destroy cancer cells. Radiation therapy may be directed toward the primary tumor site or toward metastatic lesions to help control tumor growth.
Immunotherapy: In recent years, immunotherapy has emerged as an additional therapeutic strategy being explored in Neuroendocrine Carcinoma. Immunotherapeutic approaches involve agents that stimulate the immune system to recognize and attack tumor cells. Immune checkpoint inhibitors are among the most widely studied agents in this category and are currently being investigated in clinical studies involving high-grade neuroendocrine carcinomas.
Industry Experts and Physician Views for Neuroendocrine Carcinoma
To keep up with Neuroendocrine Carcinoma market trends, we take Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) opinions working in the domain through primary research to fill the data gaps and validate our secondary research. Industry Experts were contacted for insights on the Neuroendocrine Carcinoma emerging therapies, evolving treatment landscape, patient adherence to conventional therapies, therapy switching trends, drug adoption and uptake, accessibility challenges, and epidemiology and real-world prescription patterns in Neuroendocrine Carcinoma, including MD, PhD, Instructor, Postdoctoral Researcher, Professor, Researcher, and others.
DelveInsight’s analysts conNeuroendocrine Carcinomated with 8+ KOLs to gather insights at country level. Centers such as the Emory University, Case Western Reserve University, Stanford University, University Hospital of Giessen etc. were contacted.
Their opinion helps understand and validate current and emerging Neuroendocrine Carcinoma therapies, highlight unmet medical needs, provide epidemiological context, and support strategic decisions for market access, therapy adoption, and pipeline prioritization in Neuroendocrine Carcinoma.
Region |
Key Opinion Leaders (KOLs) and Subject Matter Experts (SMEs) |
|
United States |
“The therapeutic landscape of Neuroendocrine Carcinoma is entering a phase of innovation, with novel modalities such as ADC, bispecific antibodies, and cellular therapies showing potential to address the historically limited options for these aggressive tumors.” |
|
Germany |
“Despite the aggressive biology of Neuroendocrine Carcinoma, early clinical data from next-generation immunotherapies suggest that durable responses may be achievable in a subset of patients when the immune system is effectively engaged.” |
Neuroendocrine Carcinoma Qualitative Analysis: SWOT and Conjoint Analysis
We perform qualitative and market Intelligence analysis using various approaches, such as SWOT analysis and conjoint analysis. In the SWOT analysis of Neuroendocrine Carcinoma, strengths, weaknesses, opportunities, and threats in terms of disease diagnosis, patient awareness, patient burden, competitive landscape, cost-effectiveness, and geographical accessibility of therapies are provided. Conjoint analysis analyzes emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. Scoring is given based on these parameters to analyze the effectiveness of therapy.
The team of analysts analyzes promising emerging therapies based on relevant attributes such as safety, efficacy, frequency of administration, route of administration, and order of entry. In efficacy, the trial’s primary and secondary outcome measures are evaluated, whereas the therapies’ safety is evaluated, wherein the acceptability, tolerability, and adverse events are majorly observed. In addition, the scoring is also based on the route of administration, order of entry, probability of success, and the addressable patient pool for each therapy. According to these parameters, the final weightage score and the ranking of the emerging therapies are decided.
Scope of the Neuroendocrine Carcinoma Market Report
- The Neuroendocrine Carcinoma Treatment Market Report covers a segment of key events, an executive summary, a descriptive overview of Neuroendocrine Carcinoma, explaining their causes, signs and symptoms, pathogenesis, and currently available treatments.
- Comprehensive insight has been provided into the epidemiology segments and forecasts, the future growth potential of the diagnosis rate, and disease progression along treatment guidelines.
- Additionally, an all-inclusive account of both the current and emerging treatments, along with the elaborative profiles of late-stage and prominent therapies, will have an impact on the current Neuroendocrine Carcinoma Treatment landscape.
- A detailed review of the Neuroendocrine Carcinoma Treatment Market, historical and forecasted Neuroendocrine Carcinoma Market size, Neuroendocrine Carcinoma Market Share by therapies, detailed assumptions, and rationale behind our approach is included in the report, covering the 7MM drug outreach.
- The Neuroendocrine Carcinoma Treatment Market Report provides an edge while developing business strategies by understanding trends through SWOT analysis and expert insights/KOL views, patient journey, and treatment preferences that help in shaping and driving the 7MM Neuroendocrine Carcinoma Drugs Market.
Neuroendocrine Carcinoma Market Report Insights
- Neuroendocrine Carcinoma Patient Population Forecast
- Neuroendocrine Carcinoma Therapeutics Market Size
- Neuroendocrine Carcinoma Pipeline Analysis
- Neuroendocrine Carcinoma Market Size and Trends
- Neuroendocrine Carcinoma Market Opportunity (Current and Forecasted)
Neuroendocrine Carcinoma Market Report Key Strengths
- Epidemiology-based (Epi-based) bottom-up forecasting
- Artificial Intelligence (AI)-enabled market research report
- 11-year forecast
- Neuroendocrine Carcinoma Market Outlook (North America, Europe, Asia-Pacific)
- Patient Burden Trends (by geography)
- Neuroendocrine Carcinoma Treatment Addressable Market (TAM)
- Neuroendocrine Carcinoma Competitive Landscape
- Neuroendocrine Carcinoma Major Companies Insights
- Neuroendocrine Carcinoma Price Trends and Analogue Assessment
- Neuroendocrine Carcinoma Therapies Drug Adoption/Uptake
- Neuroendocrine Carcinoma Therapies Peak Patient Share analysis
Neuroendocrine Carcinoma Market Report Assessment
- Neuroendocrine Carcinoma Current Treatment Practices
- Neuroendocrine Carcinoma Unmet Needs
- Neuroendocrine Carcinoma Clinical Development Analysis
- Neuroendocrine Carcinoma Emerging Drugs Product Profiles
- Neuroendocrine Carcinoma Market Attractiveness
- Neuroendocrine Carcinoma Qualitative Analysis (SWOT and conjoint analysis)
Key Questions Answered in the Neuroendocrine Carcinoma Market Report
Neuroendocrine Carcinoma Treatment Market Insights
- What was the Neuroendocrine Carcinoma Treatment Market Size, the Neuroendocrine Carcinoma Market Size by therapies, Neuroendocrine Carcinoma Drugs Market Share (%) distribution in 2025, and what would it look like by 2036? What are the contributing factors for this growth?
- What are the anticipated pricing variations among different geographies for the emerging therapies in the future?
- What can be the future treatment paradigm of Neuroendocrine Carcinoma?
- What are the disease risks, burdens, and unmet needs of Neuroendocrine Carcinoma? What will be the growth opportunities across the 7MM concerning the patient population with Neuroendocrine Carcinoma?
- Who is the major future competitor in the Neuroendocrine Carcinoma Drugs Market, and how will the competitors affect their market share?
- What are the current options for the treatment of Neuroendocrine Carcinoma? What are the current guidelines for treating Neuroendocrine Carcinoma in the US, Europe, and Japan?
Reasons to Buy the Neuroendocrine Carcinoma Market Report
- The Neuroendocrine Carcinoma Therapeutics Market Report will help in developing business strategies by understanding the latest trends and changing treatment dynamics driving the Neuroendocrine Carcinoma Drugs Market.
- Bottom up forecasting builds from the affected population to product forecasts, delivering a robust, data driven approach ideal for new therapies and novel classes.
- Insights on patient burden/disease Neuroendocrine Carcinoma Prevalence, evolution in diagnosis, and factors contributing to the change in the epidemiology of the disease during the forecast years.
- Understand the existing market opportunities in varying geographies and the growth potential over the coming years.
- Identifying strong upcoming players in the Neuroendocrine Carcinoma Drugs Market will help devise strategies to help get ahead of competitors.
- Detailed analysis and ranking of class-wise potential current and emerging therapies under the conjoint analysis section to provide visibility around leading classes.
- To understand KOLs’ perspectives on the accessibility, acceptability, and compliance-related challenges of existing treatment to overcome barriers in the future.
- Detailed insights on the unmet needs of the existing Neuroendocrine Carcinoma Drugs Market so that the upcoming players can strengthen their development and launch strategy.
- This Artificial Intelligence (AI) enabled report summarize and simplify complex datasets within the report into clear, actionable insights for stakeholders, investors, and healthcare providers, enabling faster, data driven decisions.
Stay updated with us for Recent Articles